Scalable Synthesis of Benzimidazole Derivatives: A Novel Low-Cost Industrial Route
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and cost-effective routes for critical heterocyclic scaffolds. Patent CN116354889A introduces a groundbreaking process for the preparation of benzimidazole derivatives, addressing long-standing challenges in synthetic efficiency and purification. Benzimidazoles are recognized as vital pharmacophores with diverse biological activities, including antimicrobial, antiviral, and anti-ulcer properties. However, traditional synthesis methods often suffer from high costs, safety risks associated with hazardous reagents, and difficult purification protocols. This new methodology leverages a copper-catalyzed hydroxylation strategy followed by reductive cyclization, offering a robust alternative for producing high-purity intermediates. The core innovation lies in the ability to utilize commercially available reagents and solvents while achieving demonstrated yields exceeding 90% in key steps, thereby streamlining the supply chain for complex organic synthesis.
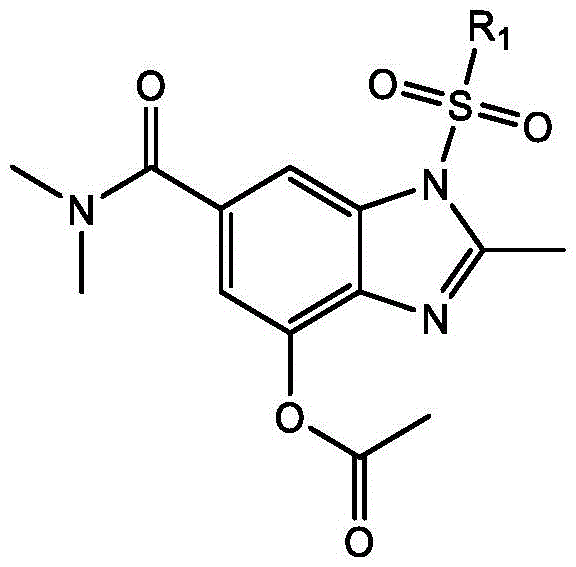
For R&D directors and process chemists, the structural integrity and purity of the final compound are paramount. The target molecules, represented generally by Formula 1, feature a substituted benzimidazole core with specific functional groups such as sulfonyl and acetate moieties. These structural features are crucial for downstream biological activity and formulation stability. The patent details a versatile synthetic route where substituents R1 can vary from methyl to trifluoromethyl or tolyl groups, allowing for the generation of a diverse library of analogs. This flexibility is essential for medicinal chemistry campaigns aiming to optimize potency and pharmacokinetic profiles. By establishing a reliable pathway to these scaffolds, manufacturers can accelerate the development of new therapeutic agents and agrochemical solutions, ensuring a steady supply of high-quality building blocks for drug discovery.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzimidazole derivatives has relied on methodologies that present significant bottlenecks for industrial application. Prior art, such as International Patent Publication WO 2004/054984, describes routes utilizing 2-amino-3-nitrophenol. While chemically feasible, these conventional approaches often necessitate the use of expensive intermediates and reagents during the amidation reaction stages. A critical drawback is the reliance on metal catalysts that are notoriously difficult to remove from the final product, leading to potential contamination issues that require rigorous and costly purification steps. Furthermore, the use of silica gel for separating intermediates is not only labor-intensive but also economically unviable for large-scale production due to the high volume of solid waste generated. Additionally, some traditional methods involve metal catalysts with fire or explosion hazards, necessitating specialized equipment and strict safety protocols that increase capital expenditure and operational complexity.
The Novel Approach
In stark contrast, the process disclosed in CN116354889A offers a streamlined solution that mitigates these risks and costs. The novel approach employs a copper-catalyzed hydroxylation reaction that proceeds under mild conditions, typically between 37°C and 60°C, using water and dimethyl sulfoxide as solvents. This shift away from hazardous conditions enhances operational safety and reduces the need for specialized containment infrastructure. Crucially, the new method eliminates the requirement for chromatographic purification, a major cost driver in fine chemical manufacturing. Instead, the process relies on crystallization and filtration, which are inherently more scalable and solvent-efficient. The use of commercially available reagents, such as cuprous bromide and common diamine ligands, ensures supply chain stability and reduces raw material costs. By avoiding high-risk reagents and simplifying the workup procedure, this methodology facilitates mass production with high yields, making it a superior choice for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Cu-Catalyzed Hydroxylation and Cyclization
The heart of this synthetic strategy is the initial copper-catalyzed hydroxylation step, which transforms a bromo-substituted nitrobenzamide into a hydroxy-intermediate. This reaction utilizes a monovalent copper catalyst, specifically selected from cuprous chloride, cuprous bromide, cuprous iodide, or cuprous oxide, with cuprous bromide being a preferred embodiment. The catalytic cycle is supported by a bidentate ligand, such as trans-N,N'-dimethylcyclohexane-1,2-diamine, which stabilizes the copper center and facilitates the substitution of the bromine atom with a hydroxyl group. The reaction is conducted in a mixed solvent system of water and DMSO, with a base like potassium carbonate present to neutralize the acid byproduct. The molar ratio of the substrate to catalyst to ligand is carefully optimized, typically ranging from 10:2:4 to 10:0.5:0.5, ensuring high conversion rates while minimizing catalyst loading. This step is critical as it installs the phenolic hydroxyl group necessary for the subsequent ring closure.
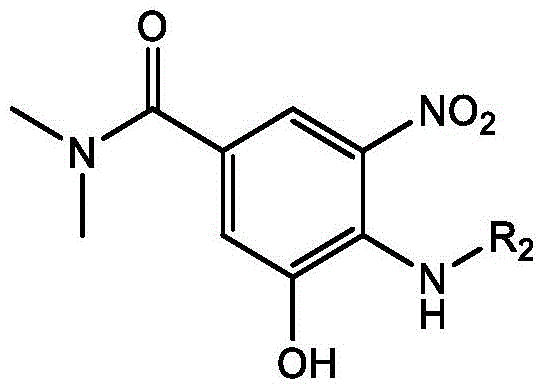
Following hydroxylation, the process proceeds through a reductive cyclization to form the benzimidazole core. This transformation involves the reduction of the nitro group to an amine, which then condenses with an acetyl source, such as triethyl orthoacetate, to close the imidazole ring. The reduction can be achieved using various agents, including hydrogen gas over palladium on carbon, reduced iron powder, or sodium dithionite, providing flexibility based on facility capabilities. For instance, hydrogenation at 0.2 MPa pressure or chemical reduction with iron in acetic acid effectively generates the reactive amine in situ. The subsequent reaction with triethyl orthoacetate drives the cyclization forward, yielding the 2-methylbenzimidazole scaffold. This tandem reduction-cyclization sequence is highly efficient, avoiding the isolation of unstable amino-intermediates. Finally, the phenolic hydroxyl group is protected or functionalized via acetylation and sulfonylation using acetyl chloride and sulfonyl chlorides in the presence of bases like pyridine or potassium carbonate, completing the synthesis of the target derivatives with high regioselectivity and purity.
How to Synthesize Benzimidazole Derivatives Efficiently
The synthesis of these valuable benzimidazole derivatives is achieved through a logical sequence of four main operational steps, each optimized for yield and scalability. The process begins with the copper-catalyzed substitution to install the hydroxyl group, followed by the reductive cyclization to build the heterocyclic core. The third stage involves the introduction of the sulfonyl and acetyl groups to finalize the molecular architecture, and the final step adjusts the pH to isolate the product in its desired form. Detailed standard operating procedures for each stage, including specific temperature controls, stirring times, and quenching protocols, are essential for reproducibility. The following guide outlines the standardized synthesis steps derived from the patent embodiments to ensure consistent quality and performance.
- Perform Cu-catalyzed hydroxylation of bromo-nitrobenzamides using CuBr and diamine ligands in aqueous DMSO at 37-60°C.
- Execute reductive cyclization using hydrogen/palladium or iron reduction followed by reaction with triethyl orthoacetate.
- Conduct sulfonylation and acetylation using sulfonyl chlorides and acetyl chloride in the presence of base.
- Finalize by adjusting pH to 8-12 using ammonia water to isolate the hydroxy-benzimidazole product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere technical feasibility. The primary advantage lies in the substantial cost savings driven by the elimination of chromatographic purification. Traditional methods often require large quantities of silica gel and solvents for column chromatography, which not only increases direct material costs but also creates significant waste disposal liabilities. By switching to a crystallization-based purification workflow, manufacturers can drastically reduce solvent consumption and waste treatment expenses. Furthermore, the use of commercially available and inexpensive reagents, such as cuprous bromide and common organic bases, stabilizes raw material costs and reduces dependency on specialty suppliers. This economic efficiency translates directly into improved margins and competitive pricing for the final active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The process achieves significant cost optimization by removing the need for expensive metal catalysts that are difficult to remove and eliminating silica gel chromatography. The reliance on crystallization for purification reduces solvent usage and waste disposal costs, while the use of commodity chemicals like acetyl chloride and potassium carbonate ensures stable and low raw material pricing. This streamlined approach minimizes unit production costs, allowing for more competitive bidding in global tenders.
- Enhanced Supply Chain Reliability: By utilizing reagents that are readily available in the global chemical market, the risk of supply disruption is significantly mitigated. The process does not depend on exotic or custom-synthesized catalysts that may have long lead times. Additionally, the moderate reaction conditions (37°C to 60°C) reduce energy consumption compared to high-temperature or high-pressure alternatives, further insulating the production schedule from energy price volatility and ensuring consistent delivery timelines for downstream customers.
- Scalability and Environmental Compliance: The avoidance of hazardous reagents and the reduction of heavy metal waste align with increasingly stringent environmental regulations. The process generates less hazardous waste, simplifying compliance reporting and reducing the burden on wastewater treatment facilities. The demonstrated scalability in the patent examples, with reactions performed on hundred-gram scales with high yields, indicates a smooth path to ton-scale production without the need for extensive process re-engineering, ensuring long-term supply continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this benzimidazole synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on process parameters, safety considerations, and product specifications. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: What are the key advantages of this benzimidazole synthesis method?
A: The process eliminates the need for expensive metal catalysts that are difficult to remove and avoids chromatographic purification, significantly reducing production costs and facilitating large-scale manufacturing.
Q: What catalysts are used in the hydroxylation step?
A: The method utilizes monovalent copper catalysts such as cuprous bromide (CuBr) in combination with diamine ligands like trans-N,N'-dimethylcyclohexane-1,2-diamine.
Q: Is this process suitable for industrial scale-up?
A: Yes, the process uses commercially available reagents and solvents, operates at moderate temperatures (37-60°C), and achieves high yields (up to 95%) without complex purification steps, making it ideal for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzimidazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the laboratory to the marketplace. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards. We are fully equipped to implement the advanced copper-catalyzed processes described in CN116354889A, leveraging our state-of-the-art facilities to deliver benzimidazole derivatives with the consistency and reliability your supply chain demands.
We invite you to collaborate with us to optimize your sourcing strategy and achieve your cost targets. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a sustainable and efficient supply of these essential pharmaceutical intermediates, driving your projects forward with confidence and precision.
