Revolutionizing Polyimide Monomer Production: A Deep Dive into High-Efficiency ODPA Synthesis
Revolutionizing Polyimide Monomer Production: A Deep Dive into High-Efficiency ODPA Synthesis
The landscape of advanced material synthesis is undergoing a significant transformation, driven by the urgent demand for high-performance polyimide precursors that meet stringent electronic-grade specifications. Central to this evolution is the production of 4,4'-diphenyl ether dianhydride (ODPA), a critical monomer that imparts exceptional flexibility and thermal stability to polyimide molecular chains. As detailed in the groundbreaking patent CN111187240B, a novel preparation method has emerged that fundamentally alters the economic and technical feasibility of ODPA manufacturing. This proprietary technology leverages a sophisticated one-step dehydration coupling reaction, bypassing the cumbersome multi-stage protocols of the past to deliver a product with unparalleled purity and yield. For R&D directors and procurement strategists alike, understanding the nuances of this synthesis route is paramount, as it represents a shift towards more sustainable, cost-effective, and scalable chemical manufacturing processes that define the next generation of reliable electronic material suppliers.
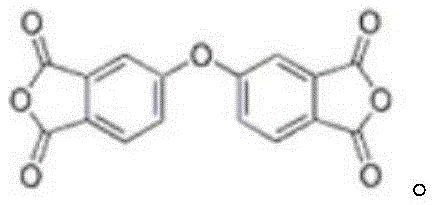
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of 4,4'-diphenyl ether dianhydride has been plagued by inherent inefficiencies that compromise both economic viability and environmental compliance. Traditional pathways often rely on complex sequences such as the condensation of 3,4-dimethoxyphenol with 4-bromo-o-xylene followed by aggressive oxidation using potassium permanganate in pyridine media. Alternatively, other legacy routes involve the methylation and nitration of phthalic anhydride to form intermediates like 4-nitro-N-methylphthalimide, necessitating subsequent bimolecular condensation, hydrolysis, and acidification steps. These antiquated processes are characterized by poor reaction selectivity, leading to the generation of substantial quantities of difficult-to-remove byproducts. Consequently, the overall yield remains stubbornly low, and the downstream purification burden is immense, requiring extensive resource allocation for waste treatment and product refinement. Furthermore, the use of harsh oxidizing agents and the generation of ammonia nitrogen emissions pose significant environmental liabilities, making these conventional methods increasingly untenable in a regulatory environment that prioritizes green chemistry and sustainability.
The Novel Approach
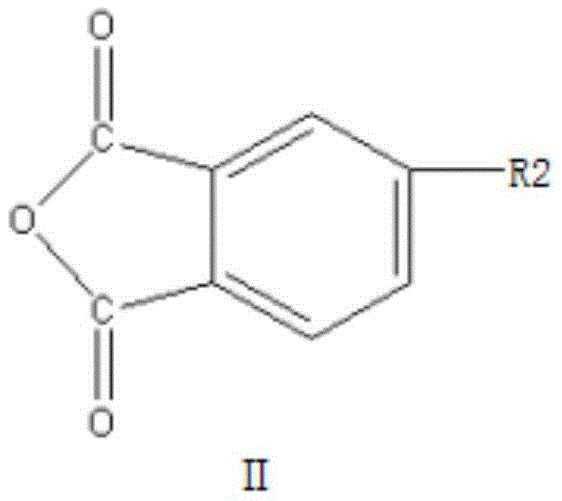
In stark contrast to the convoluted legacy pathways, the innovative method disclosed in patent CN111187240B introduces a streamlined, one-step dehydration coupling strategy that dramatically simplifies the production workflow. By strategically combining a specific alkali metal salt derivative (Compound I) with a 4-halogenated phthalic anhydride (Compound II) in the presence of a specialized solvent system and a water-carrying agent, the reaction proceeds with remarkable efficiency at temperatures ranging from 110°C to 155°C. This direct coupling approach eliminates the need for intermediate isolation and harsh oxidative steps, thereby minimizing the formation of side products and maximizing atom economy. The result is a robust process capable of achieving reaction yields exceeding 97 percent, with the final crystalline product demonstrating exceptional purity levels suitable for high-end applications. Moreover, the ability to recover and reuse the solvent and water-carrying agents post-reaction significantly lowers the operational expenditure and environmental footprint, establishing a new benchmark for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Dehydration Coupling Reaction
The core of this technological breakthrough lies in the precise mechanistic interaction between the nucleophilic oxygen species of Compound I and the electrophilic carbon center of Compound II. Compound I, typically an alkali metal salt such as sodium or potassium 4-oxyphthalic anhydride, acts as a potent nucleophile due to the electron-rich nature of the deprotonated phenolic oxygen. When introduced to Compound II, which features a highly reactive halogen substituent (fluorine, chlorine, bromine, or iodine) at the 4-position of the phthalic anhydride ring, a nucleophilic aromatic substitution (SnAr) mechanism is initiated. The reaction is facilitated by polar aprotic solvents like N,N-dimethylformamide or dimethylsulfoxide, which stabilize the transition state and enhance the solubility of the ionic species. The presence of a water-carrying agent such as toluene or xylene is critical, as it continuously removes the water molecules generated during the coupling process, driving the equilibrium towards the formation of the ether bond and preventing the hydrolysis of the sensitive anhydride rings. This delicate balance ensures that the molecular integrity of the dianhydride structure is preserved throughout the synthesis.
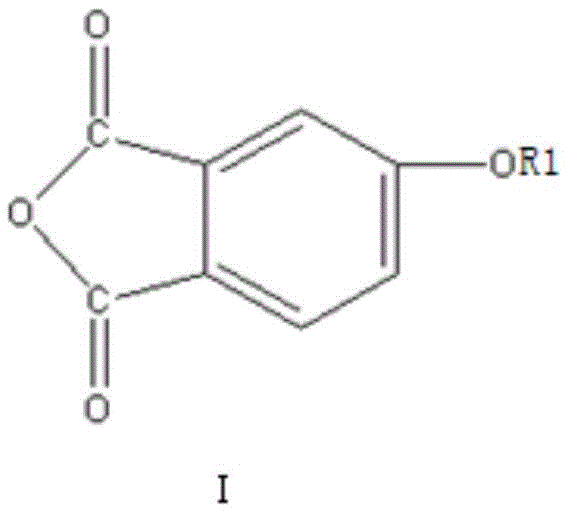
Furthermore, the control of impurities is meticulously managed through the specific selection of reactants and reaction conditions. The use of Compound I greatly improves reaction selectivity, effectively suppressing the formation of homocoupling byproducts that often occur when using Compound II alone. The patent specifies that the water content of the solvent must be strictly maintained below 500 ppm to ensure that the 4,4'-diphenyl ether dianhydride crystallizes in an orderly and thorough manner during the cooling phase. This rigorous control over moisture levels prevents the degradation of the anhydride functionality into dicarboxylic acids, which would otherwise contaminate the final product and lower its thermal performance. The subsequent refining step, involving recrystallization in fresh, low-water solvent, further purges trace impurities, elevating the purity of the ODPA crystals to 99.95 percent or higher. This level of purity is essential for ensuring the consistent mechanical and electrical properties of the resulting polyimide films, particularly in demanding applications such as flexible displays and semiconductor packaging.
How to Synthesize 4,4'-Diphenyl Ether Dianhydride Efficiently
The practical implementation of this synthesis route requires strict adherence to the optimized parameters defined in the patent to guarantee reproducibility and high quality. The process begins with the precise mixing of Compound I and Compound II in a molar ratio of 1:1 to 1:1.2, combined with a solvent and a water-carrying agent in ratios of 0.5-6 and 0.5-1.5 respectively. The reaction mixture is then heated to a temperature window of 110-155°C and maintained for a duration of 6 to 10 hours to ensure complete conversion of the starting materials. Following the reaction, a controlled cooling protocol is executed, reducing the temperature at a rate of 6-8°C per hour. A critical hot filtration step is performed when the temperature reaches 80-100°C to remove insoluble solid byproducts before the final crystallization occurs upon further cooling to 20-25°C. For those seeking the detailed standardized operating procedures and specific safety protocols for scaling this reaction, the comprehensive guide is provided below.
- Mix Compound I (alkali metal salt), Compound II (4-halophthalic anhydride), a polar aprotic solvent, and a water-carrying agent in specific molar ratios.
- Perform a dehydration coupling reaction at temperatures between 110°C and 155°C for 6 to 10 hours to ensure complete conversion.
- Cool the reaction mixture gradually (6-8°C/h), filter hot to remove byproducts, crystallize the filtrate, and refine via recrystallization in low-water content solvents.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this novel synthesis methodology offers profound advantages that extend far beyond simple chemical yield improvements. The simplification of the process flow from a multi-step sequence to a single coupling reaction inherently reduces the capital expenditure required for reactor trains and associated processing equipment. This consolidation of steps translates directly into a significant reduction in operational complexity, lowering the labor and energy costs associated with heating, cooling, and transferring materials between multiple unit operations. For supply chain managers, the robustness of this method means a more predictable production schedule with fewer interruptions caused by purification bottlenecks or off-spec batches. The ability to recycle solvents and water-carrying agents further insulates the manufacturing process from volatility in raw material pricing, providing a stable cost structure that is highly attractive for long-term supply agreements. Additionally, the elimination of hazardous reagents like potassium permanganate and the reduction of ammonia nitrogen emissions simplify waste management compliance, reducing the regulatory burden and potential liability for the manufacturing facility.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the drastic simplification of the synthetic route, which eliminates the need for expensive oxidation reagents and complex intermediate isolation steps. By utilizing readily available and low-cost raw materials such as alkali metal salts and halogenated anhydrides, the direct material cost is substantially minimized. The high reaction selectivity ensures that the majority of the input mass is converted into the desired product, thereby reducing the loss of valuable feedstock to waste streams. Furthermore, the recovery and reuse of high-boiling polar solvents and water-carrying agents create a closed-loop system that drastically cuts down on solvent consumption costs, which are typically a major expense in fine chemical synthesis. This holistic approach to cost optimization ensures that the final product is competitively priced without compromising on quality or performance standards.
- Enhanced Supply Chain Reliability: The reliance on common, commercially available starting materials mitigates the risk of supply disruptions that often plague specialty chemical manufacturing. Unlike processes that depend on custom-synthesized intermediates with long lead times, the precursors for this ODPA method are commodity chemicals with established global supply chains. The robustness of the reaction conditions, which tolerate a range of solvents and halogen substituents, provides flexibility in sourcing; if one specific halogenated anhydride becomes scarce, alternatives such as switching from chloro- to fluoro- derivatives can be accommodated within the same process framework. This adaptability ensures continuous production capability even in the face of market fluctuations, securing the supply continuity that is critical for downstream polyimide manufacturers who operate on just-in-time inventory models.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the straightforward nature of the dehydration coupling reaction, which does not require exotic catalysts or extreme pressure conditions. The exothermic nature of the reaction is manageable within standard jacketed reactors, allowing for safe scale-up to multi-ton capacities. From an environmental standpoint, the process aligns with modern green chemistry principles by minimizing waste generation and avoiding the use of heavy metal oxidants. The absence of ammonia nitrogen emissions simplifies wastewater treatment requirements, reducing the load on effluent treatment plants and lowering the overall environmental compliance costs. This eco-friendly profile not only meets current regulatory standards but also future-proofs the manufacturing operation against tightening environmental legislation, making it a sustainable choice for long-term industrial investment.
Frequently Asked Questions (FAQ)
To address the specific technical and commercial inquiries often raised by industry stakeholders regarding this advanced synthesis technology, we have compiled a set of answers based on the detailed disclosures within the patent literature. These responses clarify the operational parameters, purity capabilities, and environmental benefits that distinguish this method from traditional approaches. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this material into their existing formulations or for procurement officers assessing the total cost of ownership. The following insights provide a transparent view into the capabilities and limitations of the process, ensuring that all partners have a clear and accurate understanding of the value proposition offered by this innovative manufacturing route.
Q: What are the primary advantages of this new ODPA synthesis method over conventional routes?
A: Unlike traditional multi-step methods involving harsh oxidation or nitration which suffer from low selectivity and difficult purification, this novel one-step dehydration coupling route offers significantly higher reaction efficiency, fewer byproducts, and yields exceeding 97% with purity up to 99.99%.
Q: How does the process control impurities to achieve electronic-grade purity?
A: The process utilizes a controlled cooling crystallization strategy where the solution is filtered at 80-100°C to remove initial solid impurities, followed by slow cooling to 20-25°C. A subsequent refining step using solvents with water content below 500ppm ensures the removal of trace contaminants, achieving purity levels suitable for advanced electronic applications.
Q: Is the solvent system used in this synthesis environmentally sustainable?
A: Yes, the method allows for the recovery and repeated use of both the reaction solvent and the water-carrying agent after product separation. This recyclability drastically reduces organic waste emission, specifically avoiding the release of harmful substances like ammonia nitrogen, aligning with green chemistry principles.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Diphenyl Ether Dianhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to industrial reality requires a partner with deep technical expertise and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising metrics of patent CN111187240B are faithfully translated into bulk manufacturing reality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of 4,4'-diphenyl ether dianhydride meets the exacting standards required for high-performance polyimide applications. Our dedication to process optimization allows us to consistently deliver materials that empower our clients to push the boundaries of material science, whether in flexible electronics, aerospace composites, or advanced insulation systems.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and purity requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits of switching to this superior supply source. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and innovation in your supply chain. Let us collaborate to secure a reliable, high-quality supply of 4,4'-diphenyl ether dianhydride that supports your long-term growth and technological leadership.
