Revolutionizing ODPA Production: A Scalable Route for High-Purity Polyimide Monomers
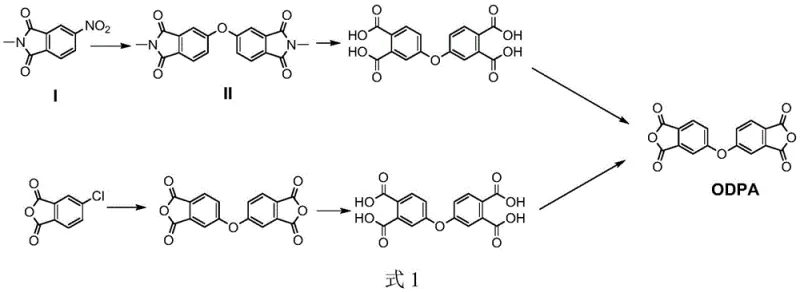
The global demand for high-performance polyimide (PI) materials has surged dramatically, driven by their critical role in flexible displays, aerospace components, and advanced microelectronics. At the heart of this supply chain lies 3,3',4,4'-diphenyl ether dianhydride (ODPA), a key monomer that dictates the thermal and mechanical properties of the final polymer. However, traditional manufacturing routes for ODPA have long struggled with inconsistent yields and significant environmental burdens, particularly when scaling from laboratory to industrial production. A groundbreaking technical solution detailed in patent CN112724111B addresses these bottlenecks by introducing a novel two-stage condensation protocol utilizing a robust solid base catalyst system. This innovation not only stabilizes the reaction kinetics against moisture interference but also significantly enhances the overall atom economy of the process. For industry leaders seeking a reliable ODPA supplier, understanding the mechanistic advantages of this patented approach is essential for securing a stable supply of high-purity electronic chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of the critical intermediate oxybis-(N-methylphthalimide) has relied on homogeneous alkali metal salt catalysts such as potassium carbonate, sodium nitrite, or simple potassium fluoride in polar aprotic solvents. While these methods function adequately on a gram scale, they exhibit severe deficiencies when translated to commercial manufacturing environments. The primary failure point is the extreme sensitivity of these soluble catalysts to trace moisture; even minor fluctuations in the water content of raw materials or solvents can destabilize the condensation reaction, leading to unpredictable conversion rates. Furthermore, conventional protocols typically necessitate quenching the reaction mixture by pouring it into vast quantities of dilute hydrochloric acid or water to precipitate the product. This archaic workup procedure generates enormous volumes of high-salt, high-chemical oxygen demand (COD) wastewater, creating a substantial environmental liability and increasing disposal costs. Additionally, scale-up data indicates that yields often plummet from acceptable laboratory levels of roughly 70% down to approximately 50% in larger reactors, rendering the process economically unviable for mass production of high-purity OLED materials.
The Novel Approach
In stark contrast, the methodology disclosed in the patent employs a heterogeneous KF/Al2O3 solid base catalyst that fundamentally alters the reaction landscape. By immobilizing the active fluoride species on an alumina support, the catalyst becomes insoluble in the reaction medium, thereby eliminating the moisture sensitivity that plagues traditional homogeneous systems. The process utilizes a sophisticated two-stage reaction sequence within a mixed solvent system comprising an aprotic polar solvent and a water-carrying agent like toluene. This setup allows for the continuous removal of reaction-generated water via azeotropic distillation, driving the equilibrium forward without deactivating the catalyst. The result is a highly stable condensation process that maintains high efficiency regardless of minor variations in feedstock quality. Moreover, the workup involves simple hot filtration to recover the solid catalyst, followed by controlled cooling to crystallize the product, completely avoiding the need for massive aqueous quenching steps. This strategic shift not only boosts the total yield of the condensation step to over 80% but also aligns perfectly with modern green chemistry principles required for sustainable electronic chemical manufacturing.
Mechanistic Insights into KF/Al2O3 Solid Base Catalysis
The core innovation of this synthesis route lies in the unique physicochemical properties of the KF/Al2O3 solid base catalyst. Unlike soluble fluoride salts that dissociate freely and interact unpredictably with trace protons in the solvent, the fluoride ions on the alumina surface act as localized basic sites that facilitate the nucleophilic attack of the phthalimide nitrogen on the nitro-substituted aromatic ring. This heterogeneous mechanism ensures that the catalytic activity remains consistent throughout the reaction duration, as the active sites are protected within the solid matrix. The use of a mixed solvent system further enhances this mechanism; the water-carrying agent forms an azeotrope with the reaction byproduct (water), allowing it to be continuously distilled off at temperatures between 150°C and 160°C. This continuous dehydration prevents the reverse hydrolysis reaction and maintains a driving force for the condensation, which is critical for achieving high conversion rates in such equilibrium-limited reactions. Consequently, the reaction proceeds with remarkable stability, unaffected by the hygroscopic nature of the starting materials that would typically derail a standard alkali-catalyzed process.
Impurity control is another critical aspect where this mechanistic approach excels, directly impacting the quality of the final high-purity ODPA. In conventional routes, the presence of soluble metal salts often leads to complex side reactions or the formation of difficult-to-remove metal-organic complexes that discolor the product. The solid nature of the KF/Al2O3 catalyst allows for its complete physical removal via hot filtration before the product crystallization begins. This physical separation ensures that the first fraction of the condensation product precipitates with a purity exceeding 98.5% and a desirable light yellow color, indicating minimal degradation or side-product formation. The second fraction, obtained after adding fresh catalyst to the mother liquor, also maintains high quality. By preventing the accumulation of soluble metal impurities in the product stream, this method significantly reduces the burden on downstream purification steps, ensuring that the final dianhydride monomer meets the rigorous specifications demanded by the semiconductor and display industries.
How to Synthesize Oxybis-(N-methylphthalimide) Efficiently
The operational protocol for this synthesis is designed to maximize throughput while minimizing waste generation, making it ideal for commercial scale-up of complex polymer additives. The process begins by charging N-methyl-4-nitrophthalimide and the KF/Al2O3 catalyst into a reactor containing the optimized mixed solvent blend. The mixture is heated to reflux conditions to initiate the first-stage condensation, during which water is continuously separated. Upon completion, the catalyst is removed via hot filtration, and the filtrate is cooled to isolate the primary crop of the intermediate. The mother liquor is not discarded; instead, it is recycled into the reactor with a fresh dose of catalyst for a second reaction cycle, extracting additional value from the unreacted starting materials. This iterative approach ensures that the overall material efficiency is maximized. For a detailed breakdown of the specific temperatures, catalyst loading ratios, and isolation procedures, please refer to the standardized synthesis guide below.
- Conduct a first-stage condensation reaction using N-methyl-4-nitrophthalimide and KF/Al2O3 catalyst in a mixed solvent system at temperatures exceeding 120°C.
- Perform hot filtration to remove the solid catalyst, then cool the filtrate to precipitate the first portion of high-purity oxybis-(N-methylphthalimide).
- Add fresh catalyst to the remaining liquid for a second-stage reaction, followed by cooling and water addition to isolate the second portion of the condensation product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this patented synthesis route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The elimination of moisture sensitivity in the catalytic system translates directly to reduced batch failure rates and more predictable production schedules, which is crucial for maintaining continuity in the supply of critical electronic materials. Furthermore, the ability to recycle the mixed solvent system and regenerate the solid catalyst creates a closed-loop process that drastically lowers the consumption of raw materials. This efficiency gain allows manufacturers to offer more competitive pricing structures without compromising on the stringent quality standards required for polyimide precursors. By adopting this technology, partners can secure a supply chain that is both cost-effective and resilient against the volatility of raw material markets.
- Cost Reduction in Manufacturing: The implementation of the KF/Al2O3 catalyst system eliminates the need for expensive phase transfer agents and reduces the reliance on high-purity anhydrous solvents that are costly to maintain. Since the catalyst is heterogeneous, it can be easily separated and regenerated through simple washing and drying processes, significantly lowering the recurring cost of catalytic materials. Additionally, the drastic reduction in wastewater volume means that facilities can operate with smaller effluent treatment capacities, leading to substantial savings in utility and waste disposal expenses. These cumulative efficiencies result in a leaner manufacturing cost structure, allowing for significant cost reduction in electronic chemical manufacturing.
- Enhanced Supply Chain Reliability: One of the most significant risks in fine chemical supply chains is batch-to-batch variability caused by environmental factors like humidity. Because this solid base catalyst is insensitive to moisture, the process is inherently more robust, ensuring consistent output quality regardless of seasonal weather changes or storage conditions of the raw materials. This reliability minimizes the need for reprocessing or rejecting off-spec batches, thereby stabilizing lead times for high-purity ODPA deliveries. Suppliers utilizing this method can guarantee a steady flow of material, which is vital for downstream customers who rely on just-in-time inventory models for their own polymer production lines.
- Scalability and Environmental Compliance: The process design inherently supports seamless scale-up from pilot plants to multi-ton annual production capacities without the yield penalties observed in traditional methods. The simplified workup procedure, which avoids massive aqueous quenching, aligns perfectly with increasingly strict global environmental regulations regarding industrial discharge. By generating significantly less hazardous waste and enabling solvent recovery, this method positions manufacturers as leaders in sustainable chemistry. This environmental compliance not only mitigates regulatory risk but also enhances the brand value of the supply chain, appealing to end-users who prioritize green sourcing in their procurement strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced ODPA synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear picture of the operational advantages. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing supply networks.
Q: Why is the KF/Al2O3 solid base catalyst superior to traditional alkali metal salts?
A: Traditional catalysts like K2CO3 or NaNO2 are highly sensitive to moisture, leading to unstable reaction rates and lower yields upon scale-up. The KF/Al2O3 solid base catalyst is water-insoluble and maintains consistent catalytic activity regardless of trace water content in solvents or raw materials, ensuring process stability.
Q: How does this new method reduce environmental waste compared to conventional processes?
A: Conventional methods require pouring reaction mixtures into large volumes of dilute acid or water to precipitate products, generating massive amounts of high-salt, high-COD wastewater. This patented method utilizes a two-stage precipitation strategy with minimal water addition only in the second stage, drastically reducing wastewater volume and simplifying treatment.
Q: What is the expected purity profile of the ODPA produced via this route?
A: The first-stage condensation product achieves a purity of not less than 98.5% with a light yellow color, while the total yield of the condensation step reaches 80-85%. This high-quality intermediate ensures that the final ODPA monomer meets the stringent specifications required for advanced polyimide applications in electronics.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable ODPA Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies requires a partner with deep technical expertise and proven industrial capability. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN112724111B are fully realized in practice. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of ODPA meets the exacting standards required for aerospace and electronic applications. We are committed to delivering not just a chemical product, but a comprehensive solution that enhances your downstream polymer performance.
We invite you to collaborate with us to optimize your supply chain for polyimide monomers. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this efficient synthesis route can improve your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and high-quality source of ODPA for your next generation of materials.
