Scalable Production of 2,3,5-Trimethylhydroquinone Diester via Advanced Acidic Ionic Liquid Catalysis
The pharmaceutical and fine chemical industries are constantly seeking more sustainable and efficient pathways for producing critical vitamin precursors. Patent CN101607896B introduces a transformative methodology for the preparation of 2,3,5-trimethylhydroquinone diester (DA-TMHQ), a pivotal intermediate in the synthesis of Vitamin E. This innovation shifts the paradigm from traditional, corrosive acid catalysis to the utilization of advanced acidic ionic liquids. By leveraging the unique physicochemical properties of these designer solvents, the process achieves a delicate balance between high catalytic activity and environmental stewardship. The technology addresses long-standing challenges in the oxidation and esterification of oxyisophorone (KIP), offering a route that minimizes hazardous waste generation while maintaining robust reaction kinetics. For global supply chains, this represents a significant step towards greener manufacturing protocols that do not compromise on yield or purity standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the conversion of 3,5,5-trimethyl-cyclohex-2-ene-1,4-dione (oxyisophorone) into DA-TMHQ has relied heavily on the use of strong protonic acids such as sulfuric acid, nitric acid, or hydrochloric acid. These conventional catalytic systems present severe operational drawbacks, primarily stemming from their high corrosivity which necessitates expensive, specialized reactor linings and increases maintenance downtime. Furthermore, these methods often require stoichiometric or near-stoichiometric amounts of acid, leading to massive volumes of acidic wastewater that require complex and costly neutralization treatments before disposal. Alternative methods utilizing Lewis acids or expensive metal salts like indium trichloride have been proposed to mitigate some issues, but they introduce new problems regarding catalyst recovery and the risk of heavy metal contamination in the final pharmaceutical grade product. The volatility of some traditional catalysts also poses safety risks and leads to material loss during high-temperature processing stages.
The Novel Approach
The methodology disclosed in the patent data revolutionizes this synthesis by employing acidic ionic liquids as the primary catalyst system. These ionic liquids, characterized by their negligible vapor pressure and high thermal stability, function effectively at moderate temperatures ranging from -20°C to 130°C. Unlike traditional acids, these catalysts do not produce acid mists, thereby protecting equipment integrity and ensuring a safer working environment for plant operators. A defining feature of this approach is the tunability of the ionic liquid structure; by modifying the cation (such as imidazolium or pyridinium derivatives) and the anion (such as bisulfate or dihydrogen phosphate), chemists can precisely optimize the acidity to match the specific requirements of the rearrangement and esterification steps. This flexibility allows for a dramatic reduction in the environmental footprint of the process, aligning perfectly with modern green chemistry principles while delivering consistent product quality.
Mechanistic Insights into Acidic Ionic Liquid-Catalyzed Rearrangement
The core of this technological advancement lies in the dual functionality of the acidic ionic liquid, which acts both as a source of protons to activate the carbonyl groups of the acid anhydride and as a stabilizing medium for the reaction intermediates. The mechanism involves the protonation of the acid anhydride by the acidic proton located on the ionic liquid cation or anion, generating a highly reactive acylium ion species. This electrophile then attacks the enolic form of the oxyisophorone, facilitating a complex rearrangement that ultimately yields the 2,3,5-trimethylhydroquinone diester structure. The ionic environment provided by the molten salt helps to stabilize charged transition states, lowering the activation energy required for the reaction compared to non-polar solvent systems. This results in improved selectivity towards the desired diester product, minimizing the formation of polymeric byproducts or over-oxidized impurities that often plague traditional acid-catalyzed routes.
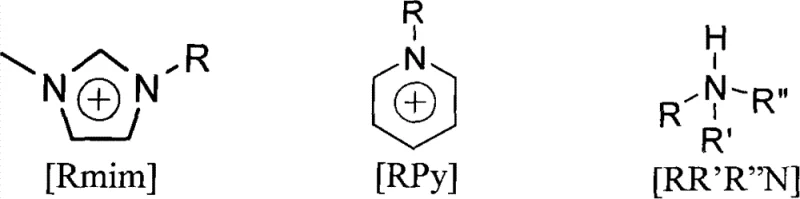
Crucially, the structural diversity of the ionic liquids allows for precise control over the reaction pathway. As illustrated in the catalyst structures, variations in the alkyl chain length (R groups) on the nitrogen-containing heterocycles influence the lipophilicity and solubility profile of the catalyst. This structural tuning is essential for the downstream purification strategy. Because the selected ionic liquids possess high water solubility, they can be effortlessly separated from the organic product phase simply by washing with water. This physical property is exploited to create a closed-loop system where the catalyst remains in the aqueous phase, ready for regeneration and reuse, while the organic product is isolated in high purity. This mechanism effectively eliminates the need for energy-intensive distillation steps solely for catalyst removal, which is a common bottleneck in processes using volatile organic catalysts.
How to Synthesize 2,3,5-Trimethylhydroquinone Diester Efficiently
The synthesis protocol outlined in the patent provides a robust framework for industrial implementation, emphasizing simplicity and scalability. The process begins with the precise metering of oxyisophorone and the chosen acid anhydride into a reactor containing the pre-heated acidic ionic liquid catalyst. Maintaining strict control over the molar ratios, typically keeping the oxyisophorone to catalyst ratio between 500:1 and 10:1, is critical for maximizing turnover numbers. The reaction mixture is then agitated under controlled thermal conditions, allowing the rearrangement and esterification to proceed to completion. Following the reaction, the workup procedure is notably straightforward, relying on vacuum distillation to recover unreacted starting materials followed by a liquid-liquid extraction to isolate the product. For detailed operational parameters and specific step-by-step instructions, please refer to the standardized guide below.
- Mix oxyisophorone (KIP) with an acid anhydride and an acidic ionic liquid catalyst (e.g., [Hmim][HSO4]) in a reactor.
- Heat the mixture to a temperature between -20°C and 130°C (preferably 0°C to 100°C) and stir for the required reaction time to facilitate rearrangement and esterification.
- Recover unreacted materials via vacuum distillation, then separate the product from the catalyst using water extraction due to the catalyst's high water solubility.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this ionic liquid-based technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift away from corrosive mineral acids and expensive transition metal catalysts fundamentally alters the cost structure of manufacturing DA-TMHQ. By eliminating the need for specialized corrosion-resistant alloys in reactors and reducing the volume of hazardous waste requiring disposal, the overall operational expenditure is significantly optimized. Furthermore, the ability to recycle the catalyst multiple times without significant loss of activity translates directly into reduced raw material consumption per kilogram of finished product. This efficiency gain provides a buffer against fluctuating raw material prices, ensuring more stable long-term costing models for downstream vitamin manufacturers.
- Cost Reduction in Manufacturing: The implementation of acidic ionic liquids removes the dependency on stoichiometric amounts of strong mineral acids, which traditionally incur high costs related to neutralization agents and waste treatment fees. Additionally, the avoidance of precious metal catalysts like indium salts eliminates a major variable cost driver, as these metals are subject to volatile market pricing and supply constraints. The simplified purification process, which relies on water extraction rather than complex chromatographic separations or multiple distillation cuts, further reduces energy consumption and labor hours, leading to substantial overall cost savings in the production of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of the ionic liquid catalyst system contributes to greater supply chain resilience by reducing the risk of production stoppages due to equipment failure or catalyst depletion. Since the catalyst is non-volatile and thermally stable, there is no risk of evaporative loss during storage or handling, ensuring consistent inventory levels. The ease of catalyst recovery means that production cycles can be shortened, as there is no lengthy downtime associated with sourcing and loading fresh batches of expensive catalysts. This reliability is crucial for meeting the tight delivery schedules demanded by global pharmaceutical clients who require just-in-time delivery of critical vitamin precursors.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to commercial production is facilitated by the benign nature of the reagents involved. The absence of toxic gas evolution (such as sulfur dioxide seen in some prior art) simplifies the engineering requirements for off-gas treatment systems, making it easier to obtain environmental permits in strictly regulated jurisdictions. The water-soluble nature of the catalyst ensures that any trace residues in the final product can be easily washed away, guaranteeing compliance with stringent heavy metal limits imposed by pharmacopeias. This environmental compatibility not only future-proofs the manufacturing site against tightening regulations but also enhances the brand reputation of the supplier as a leader in sustainable chemistry.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,3,5-trimethylhydroquinone diester using this patented ionic liquid technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the method for potential partners and technical stakeholders.
Q: What are the primary advantages of using acidic ionic liquids over traditional mineral acids for DA-TMHQ synthesis?
A: Unlike traditional mineral acids like sulfuric acid which generate significant corrosive waste and require neutralization, acidic ionic liquids are non-volatile, thermally stable, and can be easily separated from the organic product via water extraction, allowing for efficient recycling and reuse.
Q: Can the ionic liquid catalyst be recovered and reused in this process?
A: Yes, the patent explicitly demonstrates that the acidic ionic liquid catalyst remains in the aqueous phase after extraction and can be recovered by distilling the water, maintaining its catalytic activity for subsequent batches without significant loss.
Q: What represents the optimal molar ratio of oxyisophorone to acid anhydride in this novel method?
A: The process allows for a wide range of molar ratios, typically between 1:2 and 1:20, with a preferred range of 1:2 to 1:10, providing flexibility to optimize conversion rates based on specific production constraints.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5-Trimethylhydroquinone Diester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting next-generation synthetic methodologies to maintain competitiveness in the global vitamin market. Our technical team has extensively analyzed the potential of acidic ionic liquid catalysis for the production of DA-TMHQ and is fully prepared to translate this laboratory-scale innovation into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot studies to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of 2,3,5-trimethylhydroquinone diester meets the exacting standards required for pharmaceutical and nutraceutical applications.
We invite forward-thinking partners to collaborate with us on optimizing this green synthesis route for your specific supply chain needs. By leveraging our expertise in process chemistry and scale-up engineering, we can help you achieve significant efficiencies in your vitamin E precursor sourcing. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your current volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how this advanced catalytic technology can drive value for your organization.
