Advanced Catalytic Synthesis of 2,3,5-Trimethylhydroquinone Diester for Industrial Vitamin E Production
The pharmaceutical and fine chemical industries continuously seek robust methodologies for producing high-value intermediates, particularly those serving as precursors for essential nutrients like Vitamin E. Patent CN102180793A introduces a groundbreaking synthetic pathway for 2,3,5-trimethylhydroquinone diester, a critical compound in the vitamin synthesis value chain. This innovation addresses long-standing inefficiencies in traditional manufacturing by utilizing alpha-isophorone as the primary starting material, activated through a unique enol esterification strategy. Unlike conventional routes that struggle with the reactivity of ketone structures, this method employs a heteropolyacid-catalyzed aerobic oxidation system that significantly enhances molecular activity. By modifying the molecular structure of alpha-isophorone prior to oxidation, the process achieves superior yields and operational simplicity. This technical breakthrough represents a paradigm shift for reliable pharmaceutical intermediate suppliers aiming to optimize their production portfolios with greener, more cost-effective chemistry.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,5-trimethylhydroquinone derivatives has relied heavily on the oxidation of beta-isophorone, a process fraught with significant technical and economic challenges. Prior art, such as US4046813 and US4898985, describes the use of expensive transition metal catalysts like lead, vanadium, or porphyrin complexes, which not only inflate production costs but also introduce severe environmental burdens due to heavy metal contamination. Furthermore, the preparation of beta-isophorone itself is problematic; it often requires the isomerization of alpha-isophorone using corrosive basic metal compounds that damage conversion units and generate substantial by-products. These traditional oxidative methods frequently suffer from low space-time yields, the formation of difficult-to-separate superpolymers, and safety hazards associated with low flash-point solvent mixtures. Consequently, achieving high purity and consistent yield on an industrial scale has remained a persistent bottleneck for manufacturers relying on these legacy technologies.
The Novel Approach
The methodology disclosed in the patent data circumvents these obstacles by fundamentally altering the reaction trajectory, starting directly with the more stable and accessible alpha-isophorone. Instead of attempting direct oxidation, the process first protects the carbonyl group of alpha-isophorone through acylation with carboxylic acid anhydride, forming an enol isomer ester. This structural modification dramatically increases the oxidation activity of the molecule, allowing for efficient conversion using air as the oxidant in the presence of a heteropolyacid catalyst. 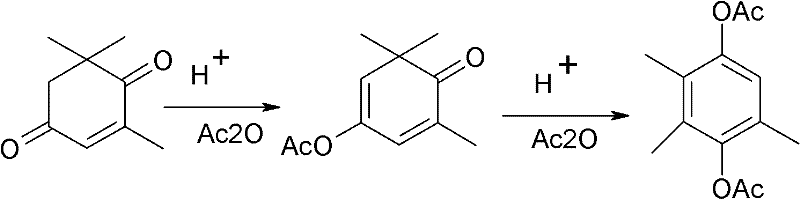 This novel sequence eliminates the need for hazardous strong alkaline environments and expensive noble metal catalysts. The subsequent steps involve a streamlined workup where solvents are recovered and recycled, leading to a final acylation that yields the target diester with high efficiency. This approach not only simplifies the operational workflow but also aligns with modern green chemistry principles by reducing waste and energy consumption.
This novel sequence eliminates the need for hazardous strong alkaline environments and expensive noble metal catalysts. The subsequent steps involve a streamlined workup where solvents are recovered and recycled, leading to a final acylation that yields the target diester with high efficiency. This approach not only simplifies the operational workflow but also aligns with modern green chemistry principles by reducing waste and energy consumption.
Mechanistic Insights into Heteropolyacid-Catalyzed Aerobic Oxidation
The core of this synthetic innovation lies in the sophisticated interplay between the enol ester intermediate and the heteropolyacid catalytic system. In the initial step, the carbonyl oxygen of alpha-isophorone is acylated, which serves a dual purpose: it protects the ketone functionality from unwanted side reactions and electronically activates the adjacent carbon-carbon double bond for oxidation. When subjected to aerobic conditions in a dimethyl sulfoxide (DMSO) solvent, the heteropolyacid catalyst, often in conjunction with an alkali promoter like potassium tert-butoxide, facilitates the insertion of oxygen into the molecular framework. This mechanism avoids the radical pathways typical of free-radical autoxidation that lead to polymerization, instead guiding the reaction towards the formation of the ketoisophorone monoester with high selectivity. The use of heteropolyacids provides a robust acidic environment that stabilizes reaction intermediates without the corrosive aggression of mineral acids like sulfuric acid.
Following the oxidation phase, the process leverages the inherent reactivity of the monoester to undergo a final rearrangement and acylation. The removal of the solvent and the introduction of fresh acylating agents under acidic catalysis drive the aromatization of the ring structure, completing the transformation into 2,3,5-trimethylhydroquinone diester. This mechanistic pathway effectively bypasses the formation of stubborn by-products such as 3,5,5-trimethyl-cyclohex-2-en-4-hydroxyl-1-ketone, which plague direct oxidation methods. By controlling the electronic environment of the substrate through temporary esterification, the reaction achieves a level of impurity control that is difficult to replicate with standard catalytic systems. This precision ensures that the final product meets the stringent purity specifications required for downstream pharmaceutical applications, minimizing the need for extensive purification steps.
How to Synthesize 2,3,5-Trimethylhydroquinone Diester Efficiently
Implementing this synthesis route requires careful attention to catalyst loading, temperature control, and solvent management to maximize yield and minimize waste. The process is designed to be telescoped where possible, with solvent recovery integrated between steps to enhance overall atom economy. Operators must ensure that the initial acylation proceeds to completion before introducing the oxidation catalyst system to prevent competing side reactions. The detailed standardized synthesis steps, including specific molar ratios and temperature profiles derived from the patent embodiments, are outlined below to guide process development teams in replicating this high-efficiency pathway.
- React alpha-isophorone with carboxylic acid anhydride using a catalyst to form the enol isomer esterification product.
- Oxidize the enol ester using air in DMSO solvent with a heteropolyacid catalyst and alkali promoter to obtain the ketoisophorone monoester.
- Perform a final acylation reaction on the monoester using carboxylic acid anhydride and a catalyst to yield 2,3,5-trimethylhydroquinone diester.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology offers tangible strategic benefits that extend beyond simple chemical yield. By shifting the raw material basis to alpha-isophorone, which is generally more abundant and easier to handle than its beta-isomer, companies can secure a more stable supply of starting materials. The elimination of expensive transition metal catalysts, such as porphyrin complexes or heavy metal salts, results in significantly reduced raw material costs and simplifies the regulatory compliance landscape regarding heavy metal residues in the final API. Furthermore, the ability to use air as the primary oxidant removes the logistical complexity and safety risks associated with handling pure oxygen or hazardous chemical oxidants, thereby enhancing overall plant safety and operational continuity.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the replacement of costly catalytic systems with reusable heteropolyacids and the implementation of solvent recycling loops. Traditional methods often incur high expenses due to the single-use nature of noble metal catalysts and the energy-intensive separation of polymeric by-products. In contrast, this novel route allows for the recovery of acetic acid and other solvents, which can be reused in subsequent batches, drastically lowering the variable cost per kilogram. Additionally, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a leaner manufacturing cost structure that improves margin potential for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Dependence on specialized, hard-to-source reagents creates vulnerability in the supply chain, particularly for complex intermediates used in vitamin production. This method utilizes commodity chemicals like acetic anhydride and air, which are readily available globally, reducing the risk of supply disruptions. The robustness of the heteropolyacid catalyst also means longer catalyst life and less frequent replenishment, ensuring consistent production schedules. By mitigating the risks associated with corrosive basic catalysts that damage equipment, manufacturers can also expect reduced maintenance downtime and longer asset life, further stabilizing the supply of critical vitamin E precursors to the market.
- Scalability and Environmental Compliance: As regulatory pressure mounts on chemical manufacturers to reduce their environmental footprint, this process offers a compliant pathway for commercial scale-up of complex pharmaceutical intermediates. The absence of heavy metal waste streams simplifies wastewater treatment and reduces the burden on effluent processing facilities. The process generates fewer hazardous by-products compared to traditional oxidation methods, aligning with increasingly strict environmental standards. This eco-friendly profile not only future-proofs the manufacturing site against tightening regulations but also appeals to end-customers who prioritize sustainable sourcing in their supply chains, adding a layer of brand value to the produced intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy processes in terms of yield, safety, and operational ease. Understanding these nuances is crucial for technical teams evaluating the feasibility of integrating this route into existing production lines.
Q: Why is alpha-isophorone preferred over beta-isophorone in this synthesis route?
A: Traditional methods relying on beta-isophorone oxidation often suffer from low yields, polymer formation, and difficult separation of isomers. This novel method activates the more stable alpha-isophorone directly through enol esterification, bypassing the need for difficult beta-isophorone purification and improving overall reaction activity.
Q: What are the advantages of using heteropolyacid catalysts in this process?
A: Heteropolyacids serve as efficient, environmentally friendly catalysts that replace expensive and toxic transition metal complexes like porphyrins or heavy metal salts. They facilitate high conversion rates under aerobic conditions while minimizing equipment corrosion and simplifying downstream waste treatment.
Q: How does this method impact the scalability of Vitamin E intermediate production?
A: The process utilizes air as the oxidant and allows for solvent recovery and recycling, significantly lowering operational costs. The mild reaction conditions and robust catalyst system make it highly suitable for large-scale commercial manufacturing without the safety hazards associated with strong alkaline oxidation environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5-Trimethylhydroquinone Diester Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that efficient synthesis pathways play in the global availability of essential vitamins and pharmaceutical ingredients. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the heteropolyacid-catalyzed oxidation of alpha-isophorone can be seamlessly transferred from the laboratory to full-scale manufacturing. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs that utilize advanced analytical techniques to verify the identity and quality of every batch. Our commitment to excellence ensures that clients receive intermediates that meet the highest industry standards for downstream drug synthesis.
We invite global partners to collaborate with us to leverage this advanced technology for their Vitamin E supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and regional logistics. We encourage potential clients to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both cost and quality. Let us be your trusted partner in delivering high-performance chemical solutions that drive your business forward.
