Revolutionizing Vitamin A Derivative Production: Advanced Dehydration Technology for Commercial Scale-Up
Revolutionizing Vitamin A Derivative Production: Advanced Dehydration Technology for Commercial Scale-Up
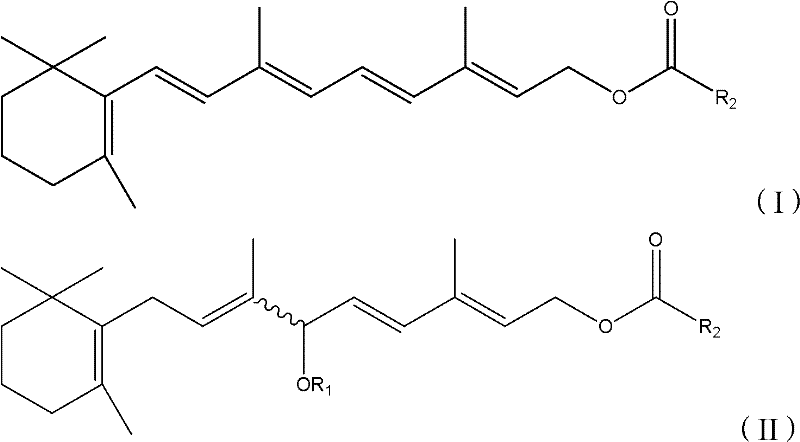
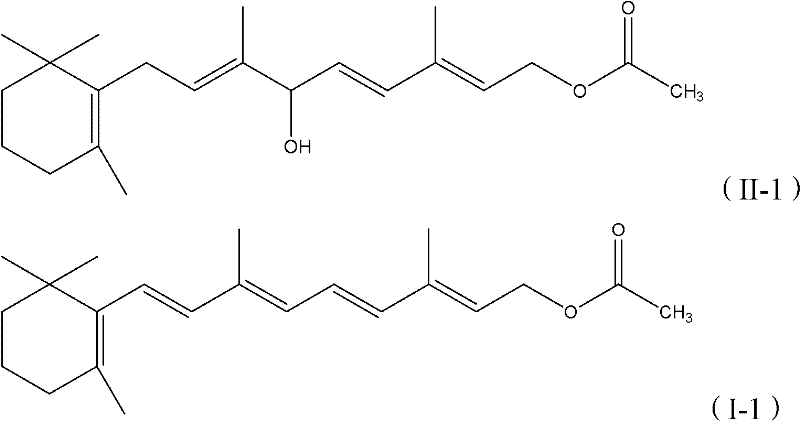
The global demand for high-purity Vitamin A derivatives, particularly Retinol Acetate and Retinol Palmitate, continues to surge across the pharmaceutical and animal nutrition sectors. Patent CN102603588A introduces a transformative methodology for constructing the critical conjugated double bond system found in these molecules. Unlike legacy processes that rely on multi-step halogenation sequences, this innovation employs a direct acid-catalyzed dehydration strategy. By converting hydroxyl or alkoxy-substituted Vitamin A intermediates directly into the desired conjugated polyene structure, this technology offers a robust pathway for manufacturers seeking to optimize their production lines. The ability to utilize a wide range of catalysts, from simple protonic acids to sophisticated Lewis acids, provides unparalleled flexibility for process chemists aiming to balance cost, yield, and stereochemical purity in the synthesis of these vital nutrients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Vitamin A acetate has been dominated by routes that necessitate the construction of the polyene chain through halogenation and elimination reactions. Traditional methodologies often involve the bromination of the intermediate followed by a dehydrobromination step to establish the conjugated system. While these methods, such as the Roche C14+C6 route, are well-established, they suffer from inherent inefficiencies. The requirement for handling hazardous brominating agents introduces significant safety risks and environmental burdens related to waste disposal. Furthermore, the multi-step nature of these traditional pathways inevitably leads to cumulative yield losses at each stage. The formation of unwanted stereoisomers during the elimination phase can also complicate downstream purification, requiring extensive chromatographic separation or recrystallization efforts that drive up operational expenditures and extend production lead times for bulk active ingredients.
The Novel Approach
The technology disclosed in patent CN102603588A represents a paradigm shift by bypassing the halogenation step entirely. This novel approach utilizes a direct dehydration mechanism where the hydroxyl group on the Vitamin A intermediate is eliminated under acidic conditions to form the carbon-carbon double bond directly. This simplification reduces the number of unit operations required, thereby minimizing material handling and potential points of failure in the manufacturing process. The method demonstrates remarkable versatility, accommodating various catalysts including sulfuric acid, phosphorus oxychloride, and metal halides like titanium tetrachloride. By operating under relatively mild temperatures ranging from -60°C to 50°C, the process ensures the stability of the sensitive polyene chain while achieving high conversion rates. This streamlined workflow not only enhances the overall throughput but also aligns with modern green chemistry principles by reducing the reliance on toxic halogens and minimizing solvent consumption.
Mechanistic Insights into Acid-Catalyzed Dehydration
The core of this synthetic breakthrough lies in the precise activation of the allylic alcohol moiety within the Vitamin A intermediate. Upon exposure to a protonic or Lewis acid catalyst, the hydroxyl group is protonated or coordinated, transforming it into a superior leaving group. This activation facilitates the departure of a water molecule, generating a resonance-stabilized allylic carbocation intermediate. The subsequent elimination of a proton from the adjacent carbon atom restores the double bond, extending the conjugation of the polyene system. The choice of catalyst plays a pivotal role in directing the stereochemical outcome of this elimination. Strong Lewis acids, such as boron trifluoride etherate or titanium tetrachloride, can coordinate with the oxygen atom to lower the energy barrier for dehydration while potentially influencing the E/Z geometry of the newly formed double bond. This mechanistic control is crucial for ensuring that the final product possesses the all-trans configuration required for optimal biological activity in pharmaceutical applications.
Impurity control is another critical aspect managed by the specific reaction conditions outlined in the patent. The use of inert atmosphere protection prevents oxidative degradation of the sensitive conjugated system, which is prone to polymerization or oxidation upon exposure to air. Furthermore, the selection of solvent systems, such as N,N-dimethylformamide or diethyl ether, influences the solubility of the intermediates and the stability of the carbocation species. By carefully tuning the molar ratio of the catalyst to the substrate, typically between 0.1 to 5 equivalents, manufacturers can suppress side reactions such as rearrangement or polymerization. The quenching step, often involving a base like triethylamine or pyridine, is equally important to neutralize residual acidity immediately after the reaction reaches completion, thereby preventing acid-catalyzed degradation of the freshly formed Vitamin A derivative during workup and isolation.
How to Synthesize Vitamin A Acetate Efficiently
The implementation of this dehydration protocol requires careful attention to reaction parameters to maximize yield and purity. The process begins with the dissolution of the hydroxyl-substituted intermediate in a dry, aprotic solvent under a nitrogen or argon blanket to exclude moisture and oxygen. Following this, the chosen catalyst is introduced slowly to manage the exotherm, and the mixture is stirred at a controlled temperature for a duration typically ranging from 3 to 30 hours. Monitoring the reaction progress via HPLC is recommended to determine the optimal endpoint before quenching. The detailed standardized synthesis steps, including specific workup procedures and purification techniques like silica gel chromatography or recrystallization, are provided in the guide below to ensure reproducible results for process development teams.
- Dissolve the Vitamin A intermediate (Formula II) in an appropriate organic solvent such as DMF or diethyl ether under inert gas protection.
- Add the selected catalyst, such as sulfuric acid, phosphorus oxychloride, or titanium tetrachloride, and maintain the reaction temperature between -60°C and 50°C.
- Quench the reaction with a base like triethylamine, followed by extraction, washing, and purification to isolate the high-purity Vitamin A derivative (Formula I).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this dehydration technology offers substantial strategic benefits beyond mere technical feasibility. The elimination of bromination reagents removes a significant cost driver associated with hazardous material handling, storage, and specialized waste treatment. This simplification of the raw material basket allows for more stable pricing and reduces dependency on volatile halogen markets. Additionally, the shortened synthetic route translates directly into reduced manufacturing cycle times, enabling faster response to market demand fluctuations. The robustness of the reaction conditions, which tolerate a variety of commercially available solvents and catalysts, ensures supply continuity even if specific reagent grades face temporary shortages. This flexibility is invaluable for maintaining uninterrupted production schedules in a competitive global marketplace.
- Cost Reduction in Manufacturing: The direct dehydration pathway significantly lowers production costs by removing entire processing stages associated with halogenation and subsequent elimination. By avoiding the use of expensive and hazardous brominating agents, facilities can reduce expenditure on safety infrastructure and environmental compliance measures. The high yields reported in the patent examples indicate efficient atom economy, meaning less raw material is wasted as byproducts. Furthermore, the ability to use common industrial solvents and catalysts reduces the overall bill of materials. These cumulative savings allow manufacturers to offer more competitive pricing for high-purity Vitamin A derivatives while maintaining healthy profit margins in a cost-sensitive industry.
- Enhanced Supply Chain Reliability: Relying on a shorter synthesis route inherently reduces the complexity of the supply chain. Fewer intermediate steps mean fewer opportunities for bottlenecks or quality failures that could disrupt the flow of goods. The catalysts employed, such as sulfuric acid or aluminum chloride, are commodity chemicals with stable global supply networks, mitigating the risk of raw material scarcity. This reliability ensures that production timelines remain predictable, allowing supply chain planners to commit to delivery schedules with greater confidence. The simplified process also facilitates easier technology transfer between manufacturing sites, providing redundancy and resilience against regional disruptions or logistical challenges.
- Scalability and Environmental Compliance: The mild operating temperatures and the absence of toxic halogenated byproducts make this process highly amenable to large-scale commercialization. Scaling up dehydration reactions is generally more straightforward than managing exothermic halogenations, reducing the engineering burden on plant operations. From an environmental perspective, the reduction in hazardous waste generation aligns with increasingly stringent global regulations on chemical manufacturing. Lower waste volumes decrease disposal costs and minimize the environmental footprint of the facility. This sustainability advantage is becoming a key differentiator for suppliers seeking to partner with multinational corporations that prioritize green chemistry and responsible sourcing in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented dehydration technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, offering a clear understanding of the process capabilities. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into existing production frameworks or new facility designs. The answers reflect the practical realities of scaling acid-catalyzed reactions for sensitive polyene compounds.
Q: What are the primary advantages of this dehydration method over traditional bromination routes?
A: This method eliminates the need for hazardous bromination and subsequent dehydrobromination steps, significantly shortening the synthetic route and improving overall yield while reducing waste generation.
Q: Which catalysts are most effective for controlling stereochemistry in this reaction?
A: Lewis acids such as titanium tetrachloride and boron trifluoride etherate, as well as heteropoly acids, offer excellent control over the formation of the conjugated double bond system, ensuring high purity of the E-isomers.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the reaction operates under mild temperature conditions (-60°C to 50°C) and utilizes commercially available solvents and catalysts, making it highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin A Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable manufacturing processes for essential nutrients like Vitamin A derivatives. Our team of expert chemists has extensively evaluated the dehydration methodology described in patent CN102603588A and confirmed its potential for delivering high-quality intermediates at scale. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent supply regardless of volume requirements. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Retinol Acetate or Palmitate meets the highest international standards for pharmaceutical and feed grade applications.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. By partnering with us, you gain access to deep process knowledge and the ability to secure specific COA data and route feasibility assessments that validate the commercial viability of this approach. Contact us today to discuss how we can support your long-term goals with reliable, cost-effective, and high-purity Vitamin A derivative solutions.
