Revolutionizing Vitamin A Acetate Production: A High-Yield C13+C7 Grignard Strategy
Introduction to Patent CN112961088B and Technological Breakthroughs
The global demand for high-purity nutritional chemicals continues to surge, driving the need for more efficient and environmentally sustainable manufacturing processes. Patent CN112961088B, published in early 2023, introduces a paradigm-shifting preparation method for Vitamin A acetate that addresses critical bottlenecks in current industrial synthesis. This innovation centers on a novel C13+C7 coupling strategy that utilizes a continuous in-situ Grignard reaction, fundamentally altering the economic and technical landscape of retinoid production. By replacing hazardous reagents and complex multi-step sequences with a streamlined approach, this technology offers a compelling value proposition for manufacturers seeking to optimize their supply chains. The patent details a specific pathway that not only enhances reaction yields but also drastically improves the stereochemical purity of the final product, ensuring superior bioavailability for end-users in the pharmaceutical and feed additive sectors.
At the heart of this technological advancement is the strategic avoidance of triphenylphosphine, a costly reagent associated with significant waste generation in traditional C15+C5 routes. Instead, the process leverages readily available starting materials like 1-bromo-2-chloroethane and specific C5 aldehydes to construct the necessary carbon backbone. The implementation of a modified solid acid catalyst for the final dehydration step represents a major leap forward in process safety and product stability. This catalyst system mitigates the risk of product deterioration often caused by strong liquid acids, thereby preserving the integrity of the sensitive conjugated double bond system inherent to Vitamin A. For industry stakeholders, this patent signifies a move towards greener chemistry without compromising on the rigorous quality standards required for regulatory compliance in food and drug applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Vitamin A acetate has been dominated by two primary technical routes, both of which suffer from inherent structural inefficiencies that impact cost and scalability. The C14+C6 route, while established, is plagued by an excessive number of reaction steps involving over 50 different raw materials, leading to high fixed capital investment and complex process control challenges. Furthermore, this method relies on intermittent series reactions and the production of double Grignard reagents, which pose significant safety risks during industrial scale-up due to the potential for thermal runaway. Another critical drawback is the use of hydrogen bromide for catalytic dehydration; since Vitamin A is highly sensitive to pH levels, this often results in product deterioration and the formation of unwanted by-products, necessitating expensive purification downstream.
Alternatively, the C15+C5 route presents a different set of economic and environmental hurdles, primarily centered around the use of triphenylphosphine. This reagent is not only expensive but generates a substantial volume of triphenylphosphine oxide solid waste, which is notoriously difficult and costly to dispose of in an environmentally compliant manner. Moreover, the Wittig-type reactions employed in this pathway frequently result in a product mixture containing high levels of cis-isomers, which possess lower biological activity and reduce the overall utilization value of the batch. These legacy methods create a bottleneck for manufacturers aiming to reduce their carbon footprint and operational expenditures, highlighting the urgent need for a more streamlined and selective synthetic strategy that can deliver high-purity outputs with minimal waste.
The Novel Approach
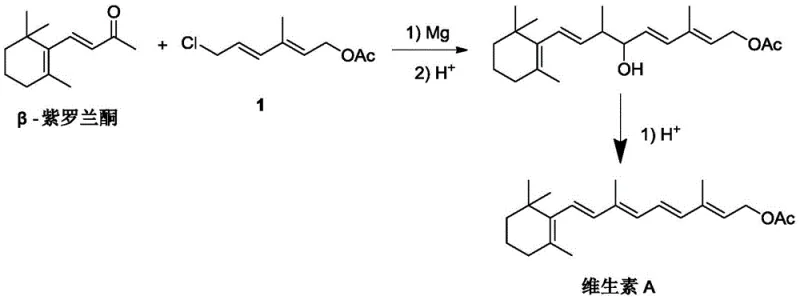
The novel C13+C7 synthesis route described in the patent data offers a robust solution to these longstanding industry pain points by fundamentally redesigning the molecular assembly process. By utilizing a continuous in-situ Grignard reaction between 1-bromo-2-chloroethane and a five-carbon aldehyde, the process efficiently constructs the C7 side chain intermediate, 3-methyl-6-chloro-2,4-hexadienol acetate, with high selectivity. This intermediate is then coupled with β-ionone in a second Grignard step, bypassing the need for phosphine-based olefination entirely. The elimination of triphenylphosphine not only removes a major cost driver but also eradicates the generation of phosphine oxide waste, aligning the manufacturing process with modern green chemistry principles and simplifying waste management protocols for facility operators.
Furthermore, this new approach incorporates a sophisticated dehydration step utilizing a modified solid acid catalyst, specifically SO4 2-/ZrO2 supported on polyethylene glycol diamine modified carboxymethyl-β-cyclodextrin. This specialized catalyst provides the necessary acidic environment for dehydration while buffering the reaction to prevent the strong acid-induced degradation of the Vitamin A molecule. The result is a process that achieves an overall yield of over 95% based on β-ionone, with an all-trans isomer content exceeding 85%. This represents a yield improvement of 10-15% compared to existing processes, translating directly into higher throughput and reduced raw material consumption per unit of finished product, making it an exceptionally attractive option for large-scale commercial production.
Mechanistic Insights into Continuous In-situ Grignard and Solid Acid Catalysis
The core mechanistic advantage of this synthesis lies in the precise control of the Grignard reaction conditions and the subsequent dehydration kinetics. In the first stage, 1-bromo-2-chloroethane reacts with magnesium and 4-acetoxy-2-methyl-2-butenal in a solvent system such as tetrahydrofuran at temperatures between 20-80°C. The in-situ generation of the Grignard reagent allows for immediate reaction with the aldehyde, minimizing side reactions and maximizing the formation of the desired chloro-dienol intermediate. Following hydrolysis with dilute acid, the intermediate undergoes dehydration catalyzed by strong acids like sulfuric acid or p-toluenesulfonic acid to yield the stable C7 building block. This step-wise construction ensures that the reactive functional groups are protected and activated at the optimal moments, reducing the formation of oligomeric impurities that often complicate purification in batch processes.
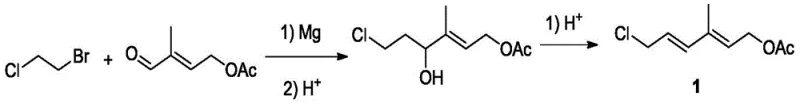
In the second critical stage, the C7 intermediate is converted into its corresponding Grignard reagent and coupled with β-ionone to form the full Vitamin A skeleton. The novelty here is the final dehydration step, which employs the modified cyclodextrin-supported solid acid catalyst. Unlike traditional liquid acids that create a homogeneous, highly aggressive acidic environment, this heterogeneous catalyst offers a controlled surface acidity. The polyethylene glycol diamine modification on the cyclodextrin support plays a crucial role in moderating the proton activity, preventing the isomerization of the sensitive all-trans polyene chain into less active cis-forms. This mechanistic nuance is vital for maintaining the high all-trans content (>85%) reported in the patent, ensuring that the final Vitamin A acetate meets the stringent quality specifications required for high-end nutritional and pharmaceutical applications.
How to Synthesize Vitamin A Acetate Efficiently
Implementing this novel C13+C7 synthesis route requires careful attention to reaction parameters, particularly regarding the preparation of the Grignard reagents and the handling of the solid acid catalyst. The process is designed to be scalable, moving from laboratory benchtop conditions to industrial reactors with minimal loss of efficiency. Operators must ensure strict moisture control during the Grignard formation stages, utilizing solvents like tetrahydrofuran that have been thoroughly dried. The dropwise addition of reactants is critical to manage the exothermic nature of the Grignard reaction, with preferred addition times ranging from 1 to 5 hours depending on the scale. Detailed standard operating procedures for temperature control, specifically maintaining the dehydration step between 100-120°C, are essential to maximize yield while preventing thermal degradation of the product.
- React 1-bromo-2-chloroethane with magnesium and C5 aldehyde (4-acetoxy-2-methyl-2-butenal) to form 3-methyl-6-chloro-2,4-hexadienol acetate via Grignard, hydrolysis, and dehydration.
- Prepare Grignard reagent from the C7 intermediate and react with β-ionone, followed by hydrolysis and dehydration using a modified cyclodextrin-supported solid acid catalyst to yield Vitamin A Acetate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis method offers transformative benefits that extend far beyond simple yield improvements. The most immediate impact is seen in the drastic simplification of the raw material portfolio; by eliminating the need for triphenylphosphine and reducing the total number of distinct precursors required compared to the C14+C6 route, companies can significantly streamline their sourcing strategies. This reduction in material complexity leads to enhanced supply chain reliability, as the risk of disruption from a single niche supplier is minimized. Furthermore, the removal of hazardous reagents like hydrogen bromide gas reduces the regulatory burden and safety infrastructure costs associated with storage and handling, allowing for more flexible site selection and lower insurance premiums.
- Cost Reduction in Manufacturing: The economic argument for switching to this C13+C7 route is compelling, driven primarily by the elimination of expensive reagents and the reduction of waste disposal costs. By avoiding the use of triphenylphosphine, manufacturers remove a significant variable cost component, while the absence of triphenylphosphine oxide waste eliminates the need for costly solid waste treatment processes. Additionally, the increase in overall yield to over 95% means that less raw material is consumed per kilogram of final product, directly improving the gross margin profile. The use of a recoverable solid acid catalyst further contributes to cost efficiency by reducing the consumption of disposable liquid acids and minimizing the neutralization salts generated during workup.
- Enhanced Supply Chain Reliability: From a logistics perspective, this process enhances stability by relying on commodity chemicals like 1-bromo-2-chloroethane and β-ionone, which are widely available from multiple global suppliers. This diversification of the supply base reduces the risk of price volatility and allocation issues that often plague specialty reagents. The simplified process flow, with fewer intermediate isolation steps, also shortens the overall production cycle time, enabling faster response to market demand fluctuations. This agility is crucial for maintaining service levels in the fast-moving consumer goods and pharmaceutical sectors, where inventory turnover rates are high and downtime is costly.
- Scalability and Environmental Compliance: The design of this synthesis route is inherently scalable, utilizing continuous in-situ reactions that are easier to control in large vessels than the intermittent batch processes of older methods. The environmental profile is markedly improved, with a significant reduction in E-factor (mass of waste per mass of product) due to the absence of phosphine waste and reduced solvent usage. This alignment with green chemistry principles facilitates easier permitting and compliance with increasingly stringent environmental regulations, future-proofing the manufacturing asset against regulatory tightening. The ability to produce high-purity product with less environmental impact also strengthens the brand reputation of the manufacturer among eco-conscious downstream customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Vitamin A acetate synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in patent CN112961088B, offering a clear picture of the operational realities and benefits. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer and for commercial teams assessing the competitive positioning of the resulting product in the global market.
Q: How does the C13+C7 route improve upon traditional C15+C5 synthesis methods?
A: The C13+C7 route eliminates the use of expensive triphenylphosphine and avoids the generation of difficult-to-handle triphenylphosphine oxide solid waste. Furthermore, it significantly reduces the formation of cis-isomers, achieving an all-trans content greater than 85%, compared to lower values in conventional phosphine-based routes.
Q: What is the role of the modified solid acid catalyst in this process?
A: The modified solid acid catalyst, specifically SO4 2-/ZrO2 supported on polyethylene glycol diamine modified carboxymethyl-β-cyclodextrin, provides a controlled acidic environment for dehydration. This prevents the strong acidity that typically causes Vitamin A degradation and isomerization, thereby protecting the sensitive polyene chain.
Q: What yields can be expected from this novel synthesis method?
A: According to patent CN112961088B, the overall yield of Vitamin A Acetate based on β-ionone is increased to over 95%. This represents a significant improvement of 10-15% over existing industrial processes, offering substantial economic advantages for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vitamin A Acetate Supplier
As the global landscape of fine chemical manufacturing evolves, partnering with a technologically adept CDMO is essential for leveraging innovations like the C13+C7 synthesis route. NINGBO INNO PHARMCHEM stands at the forefront of this transition, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped to handle the specific requirements of Grignard chemistry and solid acid catalysis, ensuring that the theoretical benefits of this patent are fully realized in commercial batches. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Vitamin A acetate meets the highest international standards for potency and isomer distribution.
We invite industry leaders to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the specific economic benefits for your operation. We encourage you to request specific COA data and route feasibility assessments to validate the performance metrics against your current benchmarks. Together, we can drive down costs and enhance the sustainability of your Vitamin A supply, securing a competitive edge in the global marketplace.
