Advanced Diastereoselective Synthesis of Fosinopril Intermediates for Commercial Scale-up
The pharmaceutical industry continuously seeks robust synthetic routes for antihypertensive agents, particularly for complex molecules like Fosinopril sodium. Patent CN1026791C discloses a pivotal advancement in the diastereoselective preparation of phosphinate esters, which serve as critical intermediates in the synthesis of Angiotensin Converting Enzyme (ACE) inhibitors. This technology addresses the longstanding challenge of controlling stereochemistry during the alkylation of phosphinic acid esters. By shifting from conventional tertiary amines to specialized bases such as 4-methylmorpholine, diazabicyclooctane, or cinchonidine, the process achieves a marked improvement in the ratio of desired diastereomers. For global procurement teams and R&D directors, this represents a significant opportunity to enhance the efficiency of high-purity pharmaceutical intermediates manufacturing. The ability to steer stereochemical outcomes at the early alkylation stage reduces the load on subsequent resolution steps, directly impacting the overall cost of goods and supply chain reliability for this essential cardiovascular medication.
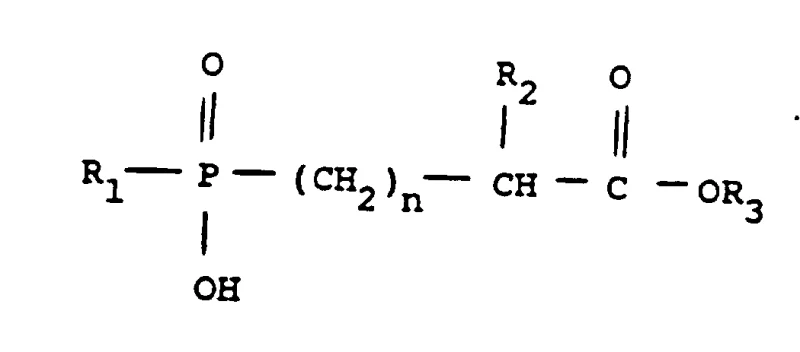
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phosphonate intermediates for Fosinopril relied heavily on standard organic bases like triethylamine to facilitate the alkylation reaction between phosphinic acid esters and halo esters. While functional, this conventional approach suffers from suboptimal diastereoselectivity, typically yielding a mixture where the ratio of the desired diastereomeric pair to the undesired pair is only about 1.2. This low selectivity creates a bottleneck in the downstream processing, as a significant portion of the reaction mass consists of unwanted isomers that must be separated and often discarded or recycled. The presence of these impurities complicates the crystallization process, requiring multiple recrystallization cycles to meet the stringent purity specifications required for API intermediate production. Furthermore, the reliance on less selective conditions can lead to variable batch-to-batch consistency, posing risks for commercial scale-up of complex pharmaceutical intermediates where reproducibility is paramount for regulatory compliance.
The Novel Approach
The innovative method described in the patent introduces a strategic modification by employing specific nitrogenous bases such as 4-methylmorpholine, quinuclidine, or cinchonidine during the alkylation step. This change in the reaction environment fundamentally alters the transition state energetics, favoring the formation of the desired stereochemical configuration. Consequently, the diastereomeric ratio improves from approximately 1.2 to roughly 1.5. Although this numerical increase may appear modest, in the context of industrial kilogram-to-ton scale manufacturing, it translates to a substantial reduction in the volume of waste material and a higher throughput of the valuable intermediate. This novel approach not only streamlines the synthesis but also enhances the economic viability of producing Fosinopril Sodium precursors. By optimizing the initial bond-forming step, manufacturers can achieve cost reduction in pharmaceutical intermediates manufacturing through improved material efficiency and reduced solvent consumption associated with extensive purification protocols.
Mechanistic Insights into Diastereoselective Alkylation
The core of this technological breakthrough lies in the nuanced interaction between the phosphinate anion and the halo ester electrophile in the presence of the selected base. When 4-methylmorpholine or similar cyclic amines are used, their specific steric and electronic properties likely stabilize the transition state leading to the desired (S*,R*) configuration more effectively than linear amines like triethylamine. The reaction proceeds in organic solvents such as toluene at elevated temperatures, typically around 95°C, which provides the necessary activation energy while the base scavenges the generated acid. This controlled environment minimizes side reactions and epimerization, ensuring that the stereochemical integrity established during the alkylation is preserved. Understanding this mechanism is crucial for R&D teams aiming to replicate or further optimize the process, as it highlights the importance of base selection beyond simple acid scavenging functionality.
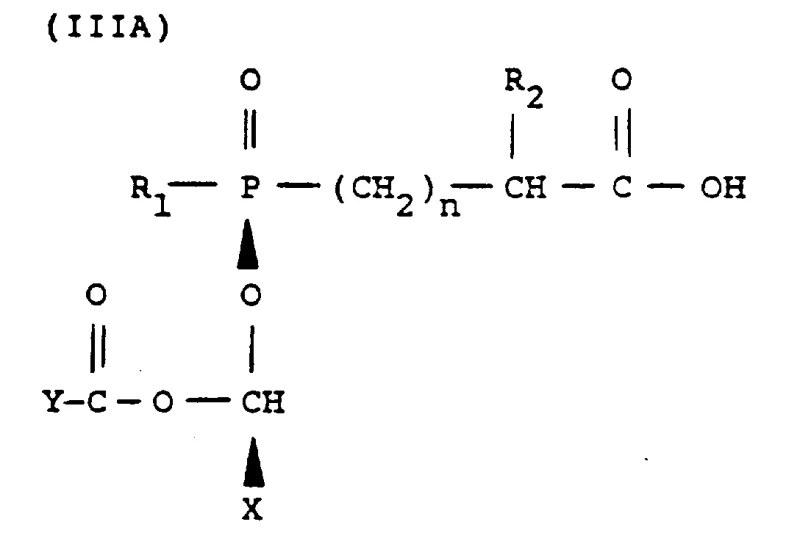
Following the alkylation, the process involves a hydrogenolysis step to remove the protecting group, typically a benzyl ester, yielding a mixture of diastereomeric acids. The improved selectivity from the previous step means this mixture is already enriched with the target isomers (IIIA and IIIB). To achieve the final optical purity required for coupling with 4-substituted L-proline, a resolution step is employed using a chiral resolving agent like L-cinchonidine. The enhanced diastereomeric ratio entering this stage facilitates a more efficient crystallization of the cinchonidine salt. The desired salt crystallizes preferentially, leaving the undesired isomers in the mother liquor. This step is critical for ensuring that the final high-purity pharmaceutical intermediates meet the rigorous quality standards necessary for human therapeutic use, minimizing the risk of chiral impurities in the final drug product.
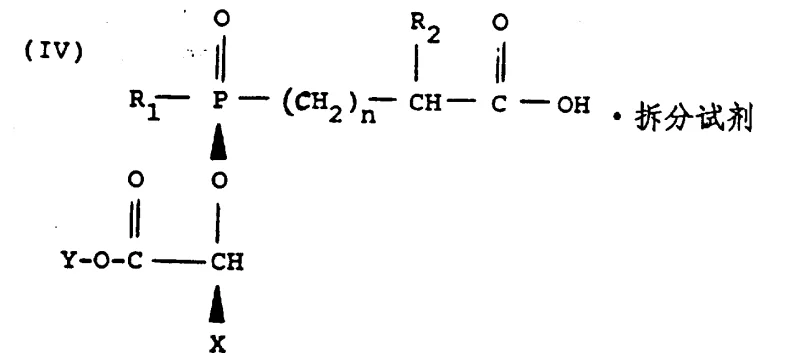
How to Synthesize Fosinopril Intermediate Efficiently
The synthesis of the key Fosinopril intermediate involves a sequence of alkylation, deprotection, and resolution steps that must be carefully controlled to maximize yield and purity. The process begins with the reaction of a phosphinate ester with a halo ester in the presence of the optimized base system, followed by catalytic hydrogenation to reveal the free acid functionality. The resulting mixture is then subjected to chiral resolution to isolate the specific stereoisomer required for the final coupling reaction. Detailed operational parameters, including precise temperature ramps, stoichiometric ratios, and crystallization kinetics, are essential for successful execution. For a comprehensive guide on the standardized operating procedures and safety protocols associated with this synthesis, please refer to the technical documentation below.
- React phosphinate ester Formula I with halo ester Formula II in toluene using 4-methylmorpholine at 95°C for 18-96 hours.
- Hydrogenate the resulting diester mixture over Pd/C to remove the protecting group, yielding a mixture of diastereomers.
- Resolve the desired diastereomeric pair using L-cinchonidine in ethyl acetate, followed by acid treatment to isolate the pure acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this diastereoselective synthesis route offers compelling strategic benefits beyond mere technical elegance. The primary advantage lies in the significant enhancement of process efficiency, which directly correlates to reduced manufacturing costs and improved supply security. By increasing the ratio of the desired isomer early in the synthesis, the process minimizes the generation of hard-to-separate impurities, thereby reducing the consumption of expensive solvents and resolving agents. This efficiency gain allows for a more streamlined production schedule, enabling suppliers to respond more rapidly to market demand fluctuations without compromising on quality. Furthermore, the use of readily available reagents like 4-methylmorpholine ensures that the supply chain remains resilient against raw material shortages, a critical factor for maintaining reliable pharmaceutical intermediates supplier status in a volatile global market.
- Cost Reduction in Manufacturing: The improved diastereoselectivity fundamentally alters the economics of the production line by reducing the amount of material that needs to be processed to obtain a unit of pure product. Since the reaction yields a higher proportion of the desired isomer, fewer batches are required to meet production targets, leading to substantial savings in utility costs, labor, and waste disposal. Additionally, the reduced burden on the resolution step means less resolving agent is consumed per kilogram of final product, further driving down the variable costs associated with cost reduction in pharmaceutical intermediates manufacturing. These cumulative efficiencies allow for a more competitive pricing structure without sacrificing margin, providing a distinct advantage in tender negotiations.
- Enhanced Supply Chain Reliability: A more robust synthetic route translates directly into greater supply chain stability. The simplified purification profile reduces the risk of batch failures due to inability to meet purity specifications, which is a common cause of supply disruption in fine chemical manufacturing. With a process that is inherently more forgiving and selective, manufacturers can maintain consistent inventory levels and shorter lead times. This reliability is crucial for downstream API manufacturers who depend on a steady flow of high-purity pharmaceutical intermediates to keep their own production lines running. By mitigating the risk of production bottlenecks, this technology supports a more agile and responsive supply network capable of adapting to sudden spikes in demand for antihypertensive medications.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common organic solvents like toluene and standard reaction conditions that are easily transferable from pilot plant to commercial scale. The reduction in waste generation, driven by higher selectivity, also aligns with increasingly stringent environmental regulations regarding solvent discharge and hazardous waste management. A cleaner process profile simplifies the permitting and compliance landscape, reducing the administrative burden and potential liabilities associated with large-scale chemical production. This environmental stewardship not only protects the company's reputation but also ensures long-term operational continuity in regions with strict ecological oversight, securing the commercial scale-up of complex pharmaceutical intermediates for the future.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this diastereoselective synthesis technology. These insights are derived directly from the patent specifications and practical experience in scaling similar phosphinate chemistries. Understanding these details helps stakeholders evaluate the feasibility and benefits of integrating this route into their existing supply chains. For further technical clarification or custom synthesis requests, our team is available to provide detailed route feasibility assessments.
Q: How does the new base catalyst improve the process compared to triethylamine?
A: The use of 4-methylmorpholine or cinchonidine increases the diastereomeric ratio of the desired isomer pair from approximately 1.2 to 1.5, significantly reducing the burden on downstream purification.
Q: What are the optimal reaction conditions for this alkylation?
A: The reaction is best conducted in toluene at approximately 95°C for 18 to 96 hours, ensuring complete conversion while maintaining selectivity.
Q: How is the final optical purity achieved?
A: Optical purity is secured through fractional crystallization of the cinchonidine salt, followed by acidification to release the free acid intermediate free of the undesired diastereomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fosinopril Sodium Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving medications. Our expertise in fine chemical synthesis allows us to master complex pathways like the diastereoselective alkylation described in CN1026791C, ensuring that we deliver products with exceptional purity and consistency. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities and rigorous QC labs to meet the most demanding specifications. Our commitment to process optimization means we can offer high-purity pharmaceutical intermediates that not only meet regulatory standards but also drive efficiency for our partners' downstream operations.
We invite you to collaborate with us to explore how this advanced synthesis route can benefit your specific project requirements. Our technical procurement team is ready to discuss your needs in detail and provide a Customized Cost-Saving Analysis tailored to your volume and quality expectations. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our technical prowess can become a strategic asset for your supply chain.
