Advanced Stereoselective Synthesis of Fosinopril Intermediates for Commercial Scale-up
Introduction to Advanced Phosphonate Ester Synthesis
The pharmaceutical industry continuously seeks robust methodologies for producing high-purity antihypertensive agents, and patent CN1053431A presents a significant breakthrough in the preparation of Fosinopril sodium intermediates. This technology addresses the critical challenge of stereoselectivity during the alkylation of phosphonic acid esters, a pivotal step in constructing the complex molecular architecture required for angiotensin-converting enzyme (ACE) inhibitors. By leveraging specific organic bases such as 4-methylmorpholine, diazabicyclooctane, or quinuclidine, the process achieves a marked improvement in the cis-selectivity of the reaction, directly impacting the yield of the therapeutically active diastereomers. This innovation is particularly relevant for manufacturers aiming to optimize their production lines for high-purity pharmaceutical intermediates while maintaining stringent quality standards required for global regulatory compliance.
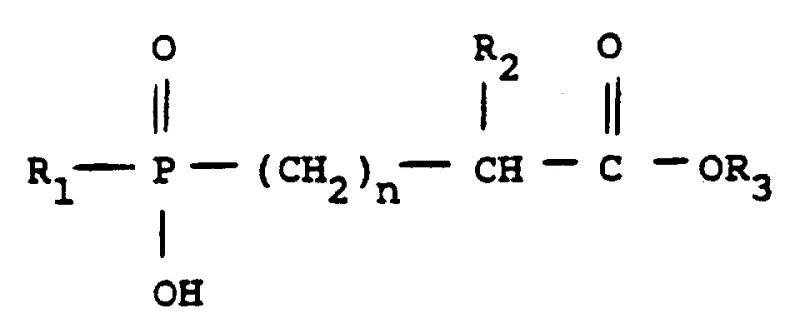
The core of this invention lies in the strategic manipulation of reaction conditions to favor the formation of specific stereochemical configurations that are otherwise difficult to isolate efficiently. Traditional methods often struggle with the generation of multiple isomeric byproducts, necessitating complex and costly purification sequences that erode profit margins and extend lead times. In contrast, the methodology described in CN1053431A utilizes a refined alkylation protocol that inherently biases the reaction pathway toward the desired structural motifs. This approach not only simplifies the downstream processing requirements but also ensures a more consistent supply of key building blocks for the final drug substance, making it an invaluable asset for any reliable pharmaceutical intermediate supplier looking to enhance their portfolio of cardiovascular medications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of phosphono-alkanoyl substituted prolines like Fosinopril has been plagued by issues related to poor diastereoselectivity and the formation of intractable mixtures of isomers. Conventional alkylation strategies often rely on generic organic bases that fail to exert sufficient stereochemical control during the carbon-phosphorus bond formation or subsequent chain elongation steps. This lack of specificity results in a crude reaction mixture containing nearly equal amounts of desired and undesired diastereomers, typically yielding a ratio close to 1.2:1. Such a distribution imposes a heavy burden on the purification infrastructure, requiring extensive chromatographic separations or multiple recrystallization cycles that drastically reduce overall material throughput. Furthermore, the reliance on less selective catalysts can lead to higher levels of trace impurities that are challenging to remove to parts-per-million levels, posing risks for patient safety and regulatory approval.
The Novel Approach
The novel approach detailed in the patent data introduces a paradigm shift by employing sterically and electronically tuned organic bases to drive the reaction equilibrium toward the target isomers. By substituting traditional amines with agents like 4-methylmorpholine or cinchonidine, the process achieves a superior isomer ratio of approximately 1.5:1 in favor of the desired diastereomeric pair (III A/B). This seemingly incremental numerical improvement translates into substantial operational advantages, as it reduces the load on resolution steps and minimizes the loss of valuable chiral material. The reaction is conducted in robust solvents like toluene at elevated temperatures around 95°C, ensuring rapid kinetics and complete conversion of the starting phosphonate esters. This method represents a significant leap forward in cost reduction in pharmaceutical intermediate manufacturing, as it streamlines the workflow and enhances the economic viability of producing complex ACE inhibitor precursors at a commercial scale.
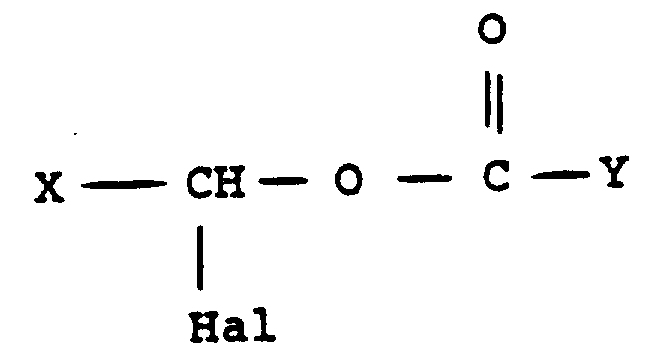
Mechanistic Insights into Stereoselective Alkylation
The mechanistic underpinning of this improved synthesis involves a nuanced interaction between the phosphonate enolate and the haloester electrophile, mediated by the specific choice of non-nucleophilic organic bases. When reagents such as 4-methylmorpholine are introduced into the reaction matrix, they facilitate the deprotonation of the phosphonate species without inducing unwanted side reactions like elimination or hydrolysis of the sensitive ester functionalities. The resulting nucleophile attacks the haloester with greater geometric fidelity, likely due to transition state stabilization effects provided by the base or solvent cage interactions. This precise control over the reaction trajectory is essential for generating the correct stereochemistry at the alpha-carbon relative to the phosphorus atom, which is a defining feature of the biologically active Fosinopril molecule. Understanding these mechanistic details allows process chemists to fine-tune parameters such as temperature and stoichiometry to maximize the yield of the target diastereomers while suppressing the formation of the inactive III C/D isomeric pair.
Impurity control is further reinforced through a sophisticated resolution strategy that exploits the differential solubility of diastereomeric salts. Following the initial alkylation and hydrogenation steps to remove protecting groups, the crude acid mixture is treated with chiral resolving agents like L-cinchonidine. This step is critical because it physically separates the desired [R-(R*,S*)] isomer from its mirror image counterparts based on their distinct crystallization behaviors in solvents like ethyl acetate. The patent specifies that treating the racemic mixture with L-cinchonidine yields a salt that can be purified to high optical purity through recrystallization, effectively filtering out the unwanted stereochemical noise. This rigorous purification protocol ensures that the final coupled product meets the stringent purity specifications demanded by pharmacopeial standards, thereby safeguarding the therapeutic efficacy and safety profile of the final antihypertensive medication.
How to Synthesize Fosinopril Intermediates Efficiently
Executing this synthesis requires careful attention to the sequential addition of reagents and the maintenance of specific thermal profiles to ensure optimal reaction outcomes. The process begins with the alkylation of the protected phosphonate ester in toluene, followed by catalytic hydrogenation to unveil the free acid functionality necessary for subsequent coupling. Detailed standard operating procedures for each unit operation, including precise molar ratios and workup techniques, are essential for replicating the high yields reported in the patent literature. For a comprehensive breakdown of the exact experimental conditions and safety protocols required for implementation, please refer to the standardized guide below.
- React phosphonate esters with haloesters in the presence of specific organic bases like 4-methylmorpholine to enhance cis-selectivity.
- Perform hydrogenation to remove protecting groups, followed by fractional crystallization to isolate the desired diastereomeric pair.
- Resolve the racemic mixture using L-cinchonidine and couple the purified acid with 4-substituted L-proline to form the final active compound.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this patented methodology offers compelling benefits for procurement managers and supply chain directors focused on efficiency and reliability. The primary advantage lies in the enhanced process efficiency derived from the improved diastereomeric ratio, which directly correlates to reduced raw material consumption and lower waste generation per kilogram of finished product. By minimizing the formation of unwanted isomers early in the synthetic sequence, manufacturers can avoid the costly and time-consuming processes associated with separating difficult-to-distinguish chemical species later in the pipeline. This streamlined approach leads to substantial cost savings in terms of both material inputs and processing time, allowing companies to offer more competitive pricing for their pharmaceutical intermediates without compromising on quality or compliance standards.
- Cost Reduction in Manufacturing: The elimination of inefficient separation steps and the reduction in solvent usage associated with multiple recrystallizations contribute to a leaner manufacturing footprint. By optimizing the selectivity of the alkylation step, the process reduces the volume of mother liquors that require treatment or disposal, thereby lowering environmental compliance costs. Furthermore, the use of commercially available and relatively inexpensive bases like 4-methylmorpholine ensures that the cost of goods sold remains stable and predictable, shielding the supply chain from volatility associated with exotic or scarce catalysts.
- Enhanced Supply Chain Reliability: The reliance on standard organic solvents such as toluene and ethyl acetate, along with common reagents like palladium on carbon for hydrogenation, ensures that the supply chain is robust and resilient to disruptions. These materials are widely sourced globally, reducing the risk of bottlenecks that can occur when depending on specialized or single-source chemicals. Additionally, the scalability of the reaction conditions, which operate effectively at moderate temperatures and pressures, facilitates easy technology transfer from laboratory to pilot plant and finally to full-scale commercial production, ensuring consistent availability of critical intermediates.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by maximizing atom economy through higher selectivity and minimizing the generation of hazardous byproducts. The ability to recycle solvents and the reduced need for extensive chromatographic purification align with modern environmental regulations, making the facility more sustainable and easier to permit. This alignment with eco-friendly manufacturing practices not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a responsible partner in the global pharmaceutical value chain.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route, providing clarity on the specific advantages and operational parameters defined in the patent documentation. These insights are derived directly from the experimental data and claims presented in CN1053431A, offering a reliable reference for technical teams evaluating this technology for adoption. Understanding these nuances is crucial for making informed decisions about process integration and resource allocation.
Q: How does the use of 4-methylmorpholine improve the Fosinopril synthesis process?
A: According to patent CN1053431A, using bases like 4-methylmorpholine or quinuclidine significantly increases the ratio of the desired diastereomeric pair (III A/B) versus the undesired pair (III C/D) from approximately 1.2 to 1.5, thereby enhancing overall process efficiency.
Q: What are the critical reaction conditions for the alkylation step?
A: The alkylation reaction is optimally conducted in organic solvents such as toluene at temperatures ranging from 40°C to 138°C, with a preferred temperature of around 95°C, ensuring complete conversion over a period of 18 to 96 hours.
Q: How is the optical purity of the intermediate ensured?
A: Optical purity is achieved through a resolution step involving L-cinchonidine or other optically active amines, which forms a separable salt with the desired isomer, allowing for the removal of unwanted stereoisomers before the final coupling stage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Fosinopril Sodium Supplier
At NINGBO INNO PHARMCHEM, we leverage advanced synthetic methodologies like the one described in CN1053431A to deliver superior quality Fosinopril intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demanding volume requirements of multinational pharmaceutical clients. We operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify the stereochemical purity and impurity profile of every batch, guaranteeing that our products consistently meet the stringent purity specifications required for API synthesis. Our commitment to technical excellence allows us to navigate the complexities of chiral synthesis with precision and reliability.
We invite potential partners to engage with our technical procurement team to discuss how our optimized manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to our supply chain. We encourage you to contact us today to obtain specific COA data and comprehensive route feasibility assessments that demonstrate our capacity to be your trusted long-term partner for high-value cardiovascular intermediates.
