Scalable Synthesis of 7-Amino-3-Propenylcephalosporanic Acid Intermediates for Oral Cephalosporins
Scalable Synthesis of 7-Amino-3-Propenylcephalosporanic Acid Intermediates for Oral Cephalosporins
The pharmaceutical industry continuously demands higher purity and more efficient synthetic routes for beta-lactam antibiotics, particularly orally active cephalosporins. Patent CN1015714B, originally filed as a continuation of US application 564,604 and related to US Patent 4,520,022, introduces a groundbreaking methodology for preparing 7-beta-amino-3-[(Z)-1-propen-1-yl]-3-cephem-4-carboxylic acid and its esters. This technology addresses a critical bottleneck in antibiotic manufacturing: the stereochemical control of the 3-position side chain. By leveraging a modified Wittig reaction enhanced with lithium halides, this process achieves a superior Z/E isomer ratio, directly impacting the biological efficacy of the final drug substance. For R&D directors and procurement specialists, understanding this proprietary pathway offers a strategic advantage in securing reliable pharmaceutical intermediates supplier partnerships that prioritize both quality and process robustness.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-substituted cephems has been plagued by poor stereocontrol and complex purification requirements. Prior art, such as United Kingdom Patent Specification 1,342,241 and US Patent 3,769,277, disclosed compounds like Structure VI but failed to provide an efficient description for preparing the specific 7-beta-amino-3-((Z)-1-propenyl) configuration required for high-potency oral antibiotics. 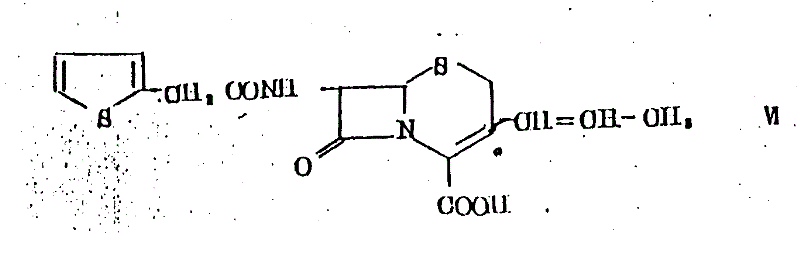 Conventional Wittig reactions often yield a mixture of cis (Z) and trans (E) olefins, necessitating difficult and costly chromatographic separations that drastically reduce overall yield. Furthermore, earlier methods described in US Patent 4,409,214 utilized 7-benzylidene amino precursors but lacked the specific catalytic conditions to enforce Z-selectivity, resulting in products with suboptimal antibacterial profiles. The reliance on standard conditions without stereochemical enhancers meant that manufacturers faced significant batch-to-batch variability and increased waste generation.
Conventional Wittig reactions often yield a mixture of cis (Z) and trans (E) olefins, necessitating difficult and costly chromatographic separations that drastically reduce overall yield. Furthermore, earlier methods described in US Patent 4,409,214 utilized 7-benzylidene amino precursors but lacked the specific catalytic conditions to enforce Z-selectivity, resulting in products with suboptimal antibacterial profiles. The reliance on standard conditions without stereochemical enhancers meant that manufacturers faced significant batch-to-batch variability and increased waste generation.
The Novel Approach
The patented innovation introduces a decisive improvement by incorporating lithium halides into the Wittig reaction medium. Unlike traditional methods that rely solely on solvent effects or temperature control, this approach actively utilizes lithium bromide (LiBr) or lithium chloride to modulate the reaction transition state. As illustrated in the reaction scheme below, the process converts a phosphoranyl cephem intermediate (Compound III) into the desired propenyl derivative with remarkable precision. 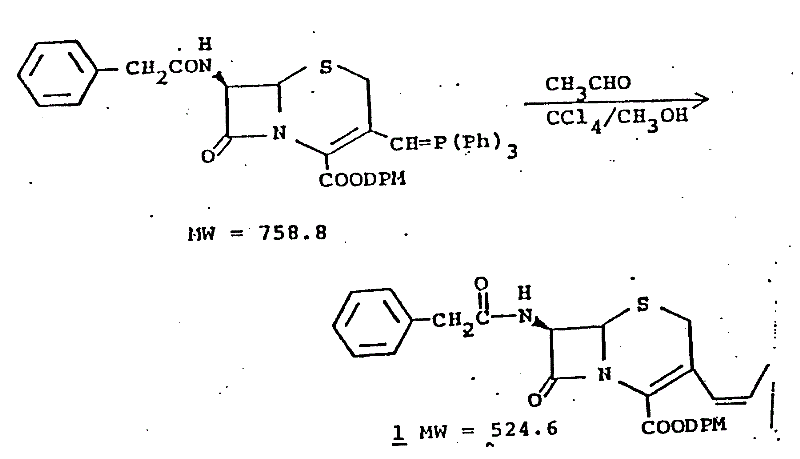 The use of methylene dichloride containing a solubility promoter like dimethyl formamide (DMF), combined with a stoichiometric excess of LiBr (5 to 15 equivalents), creates an environment that favors the formation of the Z-isomer. This method not only simplifies the downstream processing by reducing the burden of isomer separation but also ensures that the critical 3-propenyl group maintains the bioactive cis-configuration essential for Gram-negative coverage.
The use of methylene dichloride containing a solubility promoter like dimethyl formamide (DMF), combined with a stoichiometric excess of LiBr (5 to 15 equivalents), creates an environment that favors the formation of the Z-isomer. This method not only simplifies the downstream processing by reducing the burden of isomer separation but also ensures that the critical 3-propenyl group maintains the bioactive cis-configuration essential for Gram-negative coverage.
Mechanistic Insights into Lithium-Mediated Wittig Olefination
The core of this technological advancement lies in the interaction between the lithium cation and the Wittig ylide intermediate. In standard Wittig reactions involving semi-stabilized ylides, the E/Z selectivity is often modest. However, the addition of lithium salts alters the aggregation state of the ylide and stabilizes specific oxaphosphetane intermediates that lead to the Z-olefin. The patent data indicates that conducting the reaction at temperatures ranging from -10°C to +25°C, preferably 0°C to 25°C, allows for kinetic control over the olefination. The presence of LiBr likely coordinates with the oxygen of the aldehyde (acetaldehyde) and the phosphorus center, locking the conformation in a way that minimizes steric clash during the ring closure to the oxaphosphetane. This mechanistic nuance is vital for R&D teams aiming to replicate the process, as deviations in salt concentration or temperature could revert the selectivity to thermodynamic equilibrium, increasing the unwanted E-isomer.
Impurity control is another pillar of this mechanism. The process employs Girard Reagent T to selectively extract the amino-cephem product from the reaction mixture, effectively separating it from triphenylphosphine oxide and other non-ketonic byproducts. Following extraction, the use of trifluoroacetic acid (TFA) or hydrochloric acid for deprotection is carefully managed to prevent beta-lactam ring opening. The patent specifies that treating the protected intermediate with TFA yields the free acid with a Z/E ratio of 9/1, demonstrating the robustness of the initial stereochemical setup. This high level of purity is maintained through crystallization steps, where seed crystals are used to enforce the desired polymorph and isomer distribution, ensuring that the final high-purity cephalosporin intermediates meet stringent pharmacopeial standards without requiring preparative HPLC.
How to Synthesize 7-Amino-3-Propenylcephalosporanic Acid Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production. It begins with the preparation of the phosphoranyl precursor (Compound III) from 7-amino-3-chloromethyl cephem, followed by the critical lithium-mediated Wittig step with acetaldehyde. The subsequent workup involves extraction, Girard treatment, and acidolytic deprotection. While the general flow is straightforward, precise control over stoichiometry and temperature is paramount to achieving the reported 71% yield and 9:1 Z/E ratio. For detailed operational parameters, safety data, and exact reagent grades required for GMP compliance, please refer to the standardized synthesis guide below.
- Preparation of the phosphoranyl cephem intermediate (Compound III) via reaction of 7-benzylidene amino-3-chloromethyl cephem with triphenylphosphine.
- Execution of the Wittig reaction using acetaldehyde and lithium bromide (LiBr) in a methylene dichloride/DMF solvent system at -10°C to 25°C to favor Z-isomer formation.
- Purification and deprotection using Girard reagent T followed by acid treatment (TFA or HCl) to isolate the final 7-amino-3-propenylcephalosporanic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this lithium-enhanced Wittig protocol translates into tangible operational efficiencies. Traditional routes for 3-propenyl cephalosporins often suffer from low throughput due to the difficulty in separating cis and trans isomers, which acts as a bottleneck in the supply chain. By implementing a process that intrinsically favors the desired isomer, manufacturers can significantly streamline production schedules. This reduction in processing complexity directly correlates to cost reduction in antibiotic manufacturing, as it eliminates the need for expensive chiral columns or multiple recrystallization cycles that consume vast amounts of solvent and time. Furthermore, the use of readily available reagents like acetaldehyde and lithium bromide ensures that raw material sourcing remains stable and unaffected by geopolitical fluctuations common with exotic catalysts.
- Cost Reduction in Manufacturing: The elimination of complex isomer separation steps drastically lowers the cost of goods sold (COGS). By achieving a 9:1 Z/E ratio directly from the reaction, the process avoids the yield losses associated with discarding the trans-isomer fraction. Additionally, the ability to use diphenyl-methyl or tert-butyl protecting groups, which are easily removed under acidic conditions, reduces the consumption of harsh reagents and simplifies waste treatment protocols. This efficiency allows for a more competitive pricing structure for the final API, providing a strategic margin advantage in the generic antibiotic market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, operating effectively between 0°C and 25°C, reduces the dependency on specialized cryogenic equipment that can be prone to failure or maintenance downtime. The scalability of the process from 100 kgs to multi-tonne batches is supported by the use of common solvents like methylene dichloride and ethyl acetate, which are widely available in the global chemical supply chain. This accessibility ensures reducing lead time for high-purity pharmaceutical intermediates, allowing partners to respond quickly to market demand surges for oral cephalosporins without facing raw material shortages.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing solvent usage through efficient extraction and crystallization steps. The removal of triphenylphosphine oxide, a major solid waste product of Wittig reactions, is facilitated by the specific workup procedure described, making waste disposal more manageable and compliant with increasingly strict environmental regulations. The ability to scale this reaction without compromising the Z/E selectivity means that commercial scale-up of complex beta-lactam intermediates can be achieved with predictable quality outcomes, mitigating the risk of batch failures that often plague complex organic syntheses.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for process development decisions. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this technology into their existing manufacturing lines.
Q: Why is the Z-configuration critical for 3-propenyl cephalosporin intermediates?
A: The Z-configuration (cis-form) of the 3-propenyl group is essential because it determines the final cephalosporin product's favorable anti-Gram-negative bacterial performance. Isomers with the E-configuration often exhibit reduced biological activity.
Q: How does the addition of lithium halides improve the synthesis process?
A: Adding suitable lithium halides, such as lithium bromide (LiBr), during the Wittig reaction significantly improves the Z/E isomer ratio in the reaction product. The patent specifies that adding 5 to 15 stoichiometric equivalents of LiBr optimizes yield and stereoselectivity.
Q: What carboxyl-protecting groups are compatible with this synthesis route?
A: The process supports various common carboxyl-protecting groups including aralkyl (benzyl, diphenyl-methyl), alkyl (tert-butyl), and silyl groups. Diphenyl-methyl and tert-butyl are preferred as they are easily removed during acid treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-Amino-3-Propenylcephalosporanic Acid Supplier
The synthesis of 7-amino-3-propenylcephalosporanic acid represents a sophisticated intersection of stereochemistry and process engineering. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the delicate balance of the Wittig reaction is maintained even at industrial volumes. Our rigorous QC labs and stringent purity specifications guarantee that every batch of cephalosporin intermediate meets the exacting standards required for downstream API synthesis. We understand that consistency is key in the pharmaceutical supply chain, and our dedicated process chemists are ready to optimize this lithium-mediated route to suit your specific capacity needs.
We invite you to collaborate with us to leverage this advanced technology for your antibiotic portfolio. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your production volume. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain resilience and product quality.
