Revolutionizing Cefprozil Production: A Cost-Efficient 7-APCA Synthesis Strategy from 7-ACA
The pharmaceutical industry constantly seeks more efficient pathways for producing beta-lactam antibiotics, particularly second-generation cephalosporins like Cefprozil. A pivotal development in this domain is disclosed in patent CN101058584A, which outlines a novel method for preparing 7-amino-3-propenylcephalosporanic acid (7-APCA), the critical parent nucleus for Cefprozil. Unlike traditional methods that rely on expensive and less accessible starting materials, this innovation leverages 7-aminocephalosporanic acid (7-ACA) as the foundational feedstock. This strategic shift addresses long-standing economic and logistical bottlenecks in antibiotic manufacturing. The structural integrity of the target molecule, 7-APCA, is maintained through a sophisticated sequence of protection and functionalization steps, ensuring high purity suitable for downstream API synthesis.
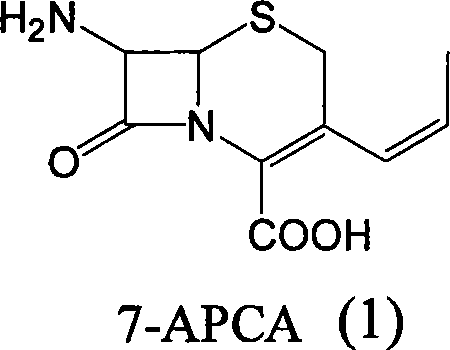
For R&D directors and process chemists, understanding the departure from legacy methodologies is crucial for evaluating process viability. The conventional industrial standard for synthesizing 7-APCA has largely depended on the GCLE (7-ACA chloromethyl ester) route. While effective, the GCLE pathway is fraught with inefficiencies, including multi-step sequences that suffer from cumulative yield losses, often hovering around 60%. Furthermore, the reliance on GCLE introduces significant supply chain vulnerability due to its specialized nature and higher market price point. The operational complexity of the GCLE route, involving hazardous chloromethylation reagents and difficult purification stages, adds to the environmental burden and production costs. These limitations necessitate a robust alternative that can deliver the same molecular architecture with greater economic and operational efficiency.
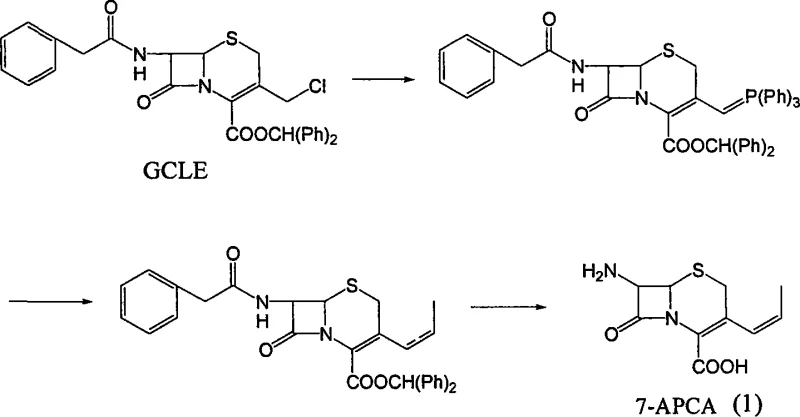
The novel approach presented in the patent fundamentally reimagines the synthetic entry point by utilizing 7-ACA, a commodity chemical with abundant global supply. This new route streamlines the construction of the 3-propenyl side chain directly onto the cephalosporin nucleus. By employing a tandem sequence of silylation, iodination, and Wittig olefination, the process bypasses the need for pre-functionalized esters like GCLE. The result is a significantly shortened synthetic timeline where intermediates are generated in situ without the need for energy-intensive isolation and drying steps. This "one-pot" philosophy not only accelerates throughput but also minimizes solvent consumption and waste generation, aligning with modern green chemistry principles while drastically simplifying the operational workflow for plant managers.
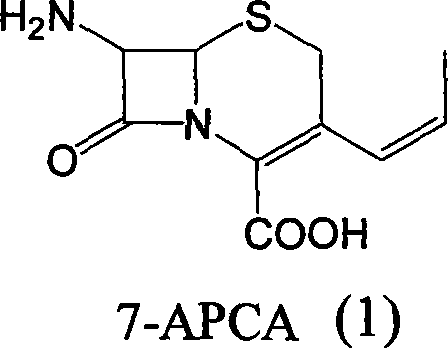
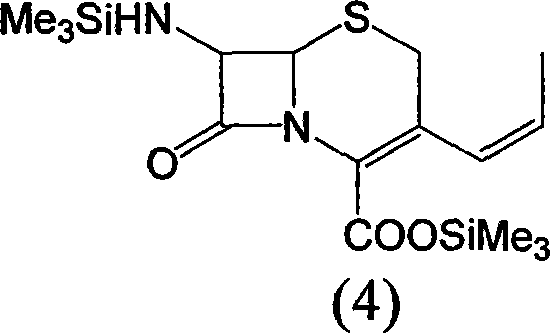
Mechanistically, the process relies on precise control of the cephalosporin nucleus reactivity through temporary protection. The initial phase involves the silylation of the 7-amino group and the carboxylic acid functionality of 7-ACA using agents such as hexamethyldisilazane (HMDS) or trimethylchlorosilane. This dual protection is essential to prevent side reactions during the subsequent nucleophilic substitutions. Following protection, an iodine substitution occurs at the C-3 position, typically utilizing iodotrimethylsilane, which serves both as an iodinating agent and a silylating source. This generates a reactive 3-iodomethyl intermediate which is immediately converted into a phosphonium salt upon treatment with triphenylphosphine. This in situ generation of the Wittig reagent precursor eliminates the stability issues associated with isolating reactive halides.
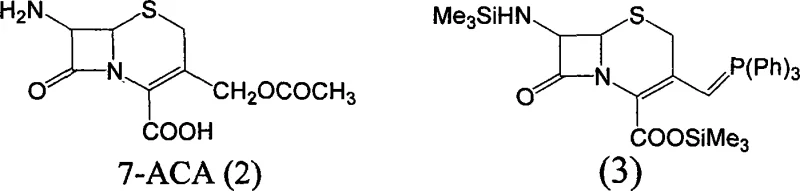
The cornerstone of this synthesis is the Wittig reaction that installs the propenyl moiety. In the presence of an alkali metal salt, such as lithium chloride or sodium iodide, the phosphonium salt reacts with acetaldehyde. The base facilitates the formation of the ylide, which then undergoes olefination with the aldehyde to extend the carbon chain at the C-3 position. The reaction conditions are meticulously controlled, typically between 0°C and 50°C, to favor the formation of the desired alkene geometry while preserving the sensitive beta-lactam ring. Finally, the silyl protecting groups are cleaved using small molecules containing reactive hydrogen, such as methanol or water, often under mild acidic or thermal conditions. This deprotection step releases the free amine and carboxylic acid, yielding the final 7-APCA product which can be crystallized to high purity specifications.
How to Synthesize 7-APCA Efficiently
The synthesis of 7-APCA via this patented route offers a compelling alternative for manufacturers seeking to optimize their Cefprozil supply chain. The procedure integrates protection, functionalization, and deprotection into a cohesive flow that minimizes handling. For detailed operational parameters, including specific solvent ratios, temperature ramps, and stoichiometric equivalents required to replicate this high-yield process, please refer to the standardized synthesis guide below. This guide encapsulates the critical process parameters (CPPs) necessary to achieve the reported purity and yield benchmarks consistently.
- Protect 7-ACA via silylation and perform iodine substitution to form the phosphonium salt intermediate.
- Conduct a Wittig reaction with acetaldehyde in the presence of an alkali metal salt to introduce the propenyl group.
- Remove silyl protecting groups through hydrolysis or alcoholysis to crystallize the final 7-APCA product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, the shift from GCLE to 7-ACA as the starting material represents a transformative opportunity for cost optimization and risk mitigation. The traditional reliance on GCLE exposes manufacturers to price volatility and supply shortages, as GCLE is a value-added derivative with a limited number of global producers. In contrast, 7-ACA is a fermentation-derived bulk commodity produced at massive scales worldwide, ensuring a stable and competitive pricing environment. By adopting this 7-ACA-based route, companies can decouple their production costs from the premium pricing of specialized intermediates, leading to substantial reductions in the overall cost of goods sold (COGS) for Cefprozil. This strategic sourcing advantage provides a buffer against market fluctuations and enhances long-term financial predictability.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the elimination of expensive starting materials and the reduction of unit operations. By avoiding the purchase of GCLE and utilizing cheaper 7-ACA, the raw material cost baseline is significantly lowered. Furthermore, the ability to perform reactions in tandem without isolating intermediates reduces labor costs, energy consumption for drying, and solvent recovery loads. The cumulative effect of these efficiencies results in a leaner manufacturing process that maximizes asset utilization and minimizes waste disposal costs, directly improving the bottom line for large-scale production facilities.
- Enhanced Supply Chain Reliability: Supply continuity is a critical metric for pharmaceutical supply chains, and this route offers superior resilience. Since 7-ACA is widely available from multiple suppliers across different geographic regions, the risk of single-source dependency is mitigated. This abundance ensures that production schedules can be maintained even during regional disruptions or logistics delays. Additionally, the simplified process flow reduces the lead time required to convert raw materials into the finished intermediate, allowing for more responsive inventory management and faster time-to-market for the final antibiotic formulation.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard chemical transformations that are well-understood in industrial settings. The reduction in the number of isolation steps decreases the physical footprint required for processing and lowers the potential for cross-contamination. From an environmental standpoint, the decreased solvent usage and waste generation align with increasingly stringent regulatory requirements for pharmaceutical manufacturing. This compliance readiness reduces the administrative burden of environmental reporting and permits, facilitating smoother audits and sustained operational licenses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this 7-ACA-based synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and benefits of adopting this technology for commercial production. Understanding these nuances is essential for making informed decisions about process transfer and vendor qualification.
Q: What is the primary advantage of using 7-ACA over GCLE for 7-APCA synthesis?
A: The primary advantage is significant cost reduction and improved supply stability. 7-ACA is a widely available, inexpensive bulk commodity compared to GCLE, which is a specialized, higher-cost intermediate often subject to supply constraints.
Q: Does this new route require isolation of intermediates?
A: No, the process is designed for efficiency. The intermediates (Compound 3 and Compound 4) do not require isolation; the reaction mixture proceeds directly to the next step, reducing operational time and solvent usage.
Q: What represents the key chemical transformation in this patent?
A: The core transformation is a Wittig reaction performed on a silyl-protected cephalosporin nucleus. This allows for the direct installation of the 3-propenyl side chain from the 3-iodomethyl precursor using acetaldehyde.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 7-APCA Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global antibiotic market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering high-purity 7-APCA that meets stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our infrastructure is designed to handle complex beta-lactam chemistry with the utmost safety and precision, guaranteeing a consistent supply of this critical building block for your Cefprozil requirements.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized 7-APCA production can enhance your operational efficiency and profitability.
