Advanced Synthetic Route for 8-Chloro-1,7-Naphthyridine-3-Formaldehyde Manufacturing
Introduction to Next-Generation Immunomodulator Intermediates
The pharmaceutical landscape is continuously evolving, driven by the demand for novel immunomodulators and kinase inhibitors that offer improved therapeutic profiles. A pivotal building block in this domain is 8-chloro-1,7-naphthyridine-3-formaldehyde, a versatile intermediate essential for constructing the core scaffolds of next-generation drugs targeting PDE4 and p38 kinase pathways. Recent intellectual property developments, specifically patent CN110950865A, have unveiled a transformative synthetic methodology that addresses long-standing bottlenecks in the production of this critical heterocycle. This innovation shifts the paradigm from hazardous, multi-step sequences to a streamlined, two-step protocol that prioritizes both economic efficiency and environmental sustainability. For R&D leaders and procurement strategists, understanding this technological leap is crucial for securing a reliable supply chain for high-value active pharmaceutical ingredients.
The significance of this patent lies not merely in the chemical transformation itself but in its profound implications for industrial scalability. Traditional approaches to synthesizing 1,7-naphthyridine derivatives have often been plagued by low atom economy and the necessity for exotic reagents. By contrast, the disclosed method leverages widely available starting materials and benign reaction conditions to achieve high yields with exceptional purity. This report delves deep into the mechanistic underpinnings and commercial advantages of this route, providing a comprehensive analysis for stakeholders aiming to optimize their manufacturing strategies for complex nitrogen-containing heterocycles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 8-chloro-1,7-naphthyridine-3-carbaldehyde has been a formidable challenge, characterized by convoluted reaction pathways that hinder efficient commercial production. As illustrated in prior art such as WO201354291 and US20170174679, conventional strategies typically commence with 5-bromo-3-methylpyridine-2-carboxylic acid ethyl ester, necessitating a tedious sequence of urethane exchange, imidization, cyclization, and subsequent chlorination. These multi-step processes inherently accumulate impurities at each stage, requiring rigorous and costly purification protocols that erode overall yield. Furthermore, alternative routes reported in patents like US2014213581 rely heavily on palladium-catalyzed coupling reactions involving toxic reagents such as zinc cyanide.
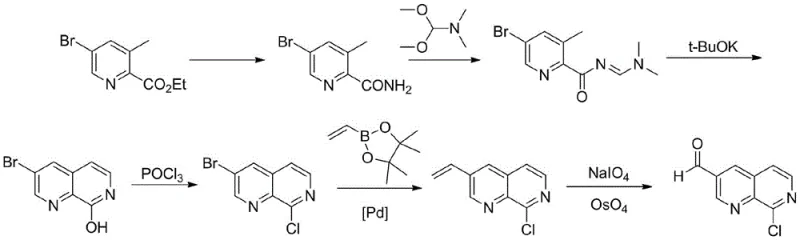
The reliance on transition metal catalysis introduces significant supply chain vulnerabilities, including the fluctuating costs of precious metals and the stringent regulatory requirements for residual metal removal in final drug substances. Additionally, many of these legacy processes involve high-pressure reactions and the use of pyrophoric or hypertoxic substances, posing severe safety risks in a manufacturing environment. The cumulative effect of these factors is a production process that is not only capital-intensive but also environmentally burdensome, generating substantial hazardous waste that complicates disposal and compliance. For procurement managers, these inefficiencies translate into higher unit costs and extended lead times, making the sourcing of high-purity intermediates a persistent logistical challenge.
The Novel Approach
In stark contrast to the cumbersome legacy methods, the innovative strategy disclosed in CN110950865A offers a remarkably concise and robust alternative. This novel approach bypasses the need for complex ring construction or hazardous cyanation steps by utilizing a direct functional group interconversion strategy. The process begins with the reduction of a readily accessible 8-chloro-1,7-naphthyridine-3-carboxylic acid ester, transforming it directly into the corresponding alcohol intermediate. This is followed by a highly selective oxidation step to generate the target aldehyde. By collapsing the synthesis into just two high-yielding transformations, the new method drastically reduces the number of unit operations, thereby minimizing material loss and processing time.
The elegance of this route lies in its use of commodity chemicals rather than specialized reagents. The elimination of palladium catalysts and toxic cyanides not only lowers the raw material bill but also simplifies the downstream processing, as there is no need for expensive metal scavenging resins or complex aqueous workups to remove heavy metals. This streamlined workflow significantly enhances the overall throughput of the manufacturing line, allowing for faster batch turnover. For supply chain heads, this translates to a more resilient sourcing model where the risk of production delays due to reagent shortages or safety incidents is substantially mitigated. The result is a cost reduction in API manufacturing that is both immediate and sustainable, aligning perfectly with the industry's push towards greener chemistry.
Mechanistic Insights into Lewis Acid-Catalyzed Reduction and TEMPO Oxidation
The core of this technological breakthrough rests on two distinct yet complementary chemical transformations: a Lewis acid-promoted reduction and a TEMPO-mediated oxidation. In the first step, the 8-chloro-1,7-naphthyridine-3-carboxylic acid ester is subjected to reduction using sodium borohydride in the presence of a Lewis acid catalyst such as calcium chloride or zinc chloride. The role of the Lewis acid is critical; it coordinates with the carbonyl oxygen of the ester, increasing the electrophilicity of the carbonyl carbon and facilitating the nucleophilic attack by the borohydride species. This activation allows the reduction to proceed under mild thermal conditions, typically between 0°C and 50°C, avoiding the harsh conditions often required for reducing sterically hindered or electron-deficient heterocyclic esters.
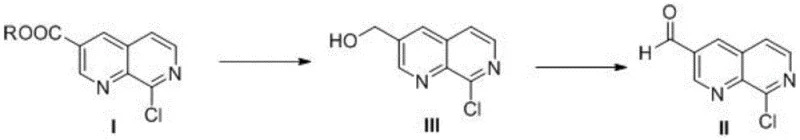
Following the isolation of the 2-chloro-1,7-naphthyridine-3-methanol intermediate, the second step employs a TEMPO (2,2,6,6-tetramethylpiperidine-1-oxyl) catalytic system for oxidation. This green chemistry approach utilizes sodium hypochlorite as the terminal oxidant, which is both inexpensive and environmentally benign compared to chromium-based oxidants like Jones reagent. The reaction is carefully buffered to maintain a pH between 7 and 12, ideally around 9-10, using acid-binding agents like sodium bicarbonate. This pH control is vital for preventing the over-oxidation of the aldehyde to the corresponding carboxylic acid, ensuring high selectivity for the desired product. The mechanistic precision of this oxidation step ensures that the sensitive chloro-substituent on the naphthyridine ring remains intact, preserving the integrity of the molecule for subsequent derivatization.
From an impurity control perspective, this mechanism offers distinct advantages. The mildness of the Lewis acid reduction minimizes side reactions such as dehalogenation, which is a common issue when reducing halogenated heterocycles with stronger reducing agents like lithium aluminum hydride. Similarly, the TEMPO oxidation is highly chemoselective, targeting only the primary alcohol without affecting other potential oxidation-sensitive sites on the scaffold. This inherent selectivity results in a crude product profile that is significantly cleaner than those obtained from traditional routes, reducing the burden on purification teams. For R&D directors focused on quality by design, this mechanistic robustness provides a solid foundation for developing a stable and reproducible commercial process that consistently meets stringent purity specifications.
How to Synthesize 8-Chloro-1,7-Naphthyridine-3-Formaldehyde Efficiently
Implementing this synthetic route in a pilot or production setting requires careful attention to reaction parameters, particularly temperature control and stoichiometry during the reduction phase. The process begins by dissolving the ester starting material in a polar aprotic solvent such as tetrahydrofuran or methyl tert-butyl ether, followed by the addition of the Lewis acid catalyst. The reducing agent, typically sodium borohydride, is added in batches to manage the exotherm, ensuring the temperature remains within the optimal 0-10°C range initially before warming to 40-50°C for completion. The subsequent oxidation step demands precise pH monitoring to maintain the activity of the TEMPO catalyst while preventing degradation of the product. Detailed standardized operating procedures for these steps are outlined below to ensure reproducibility and safety.
- Dissolve 8-chloro-1,7-naphthyridine-3-carboxylic acid ester in a solvent like THF and reduce it using sodium borohydride under Lewis acid catalysis (e.g., CaCl2) to obtain the alcohol intermediate.
- Oxidize the resulting 2-chloro-1,7-naphthyridine-3-methanol using a TEMPO catalyst system with sodium hypochlorite as the oxidant at controlled pH to yield the final aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain executives, the adoption of this novel synthetic route represents a strategic opportunity to optimize the cost structure and reliability of the supply base for immunomodulator intermediates. The primary driver of value creation here is the drastic simplification of the supply chain itself. By eliminating the dependency on precious metal catalysts like palladium and toxic reagents like zinc cyanide, manufacturers can source raw materials from a much broader and more stable vendor base. This diversification reduces the risk of supply disruptions caused by geopolitical issues or mining constraints associated with rare earth and precious metals. Furthermore, the use of commodity chemicals such as sodium borohydride and sodium hypochlorite ensures that raw material costs remain low and predictable, shielding the project from volatile market fluctuations.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming from both direct material savings and operational efficiencies. The elimination of expensive transition metal catalysts removes a significant line item from the bill of materials, while the avoidance of toxic cyanides negates the need for specialized containment equipment and hazardous waste disposal services. Additionally, the high selectivity of the TEMPO oxidation reduces the formation of by-products, which in turn lowers the consumption of solvents and energy required for purification. These factors combine to deliver substantial cost savings without compromising on the quality of the final intermediate, making it an economically superior choice for large-scale production.
- Enhanced Supply Chain Reliability: From a logistics perspective, the simplicity of this route enhances the agility of the supply chain. The starting materials are commercially available bulk chemicals, meaning that lead times for raw material procurement are significantly shorter compared to custom-synthesized precursors required by older methods. The robustness of the reaction conditions also means that the process is less susceptible to batch failures due to minor variations in reagent quality or environmental conditions. This reliability allows for more accurate production planning and inventory management, ensuring that downstream drug development timelines are met without delay. Reducing lead time for high-purity intermediates becomes a tangible reality with this streamlined approach.
- Scalability and Environmental Compliance: As the pharmaceutical industry faces increasing pressure to adopt greener manufacturing practices, this synthetic method offers a clear path to compliance. The absence of heavy metals and toxic cyanides simplifies the environmental permitting process and reduces the liability associated with waste treatment. The reaction operates at near-atmospheric pressure and moderate temperatures, which lowers the energy footprint of the manufacturing facility. These attributes make the process highly scalable, capable of transitioning smoothly from laboratory benchtop to multi-ton commercial production. For companies committed to sustainability goals, adopting this route demonstrates a proactive approach to environmental stewardship while maintaining commercial viability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthetic technology. These insights are derived directly from the patent data and practical considerations for scaling nitrogen-heterocycle synthesis. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this intermediate into their drug substance manufacturing workflows. The answers provided reflect the consensus on best practices for handling 1,7-naphthyridine derivatives and optimizing the specific reduction-oxidation sequence described.
Q: How does this new synthetic route improve safety compared to traditional methods?
A: Traditional routes often rely on palladium-catalyzed couplings and toxic reagents like zinc cyanide under high pressure. This novel method utilizes mild Lewis acid reduction and TEMPO oxidation, eliminating heavy metals and high-pressure hazards, significantly enhancing operational safety.
Q: What are the key cost drivers reduced in this manufacturing process?
A: The process eliminates expensive transition metal catalysts and complex multi-step sequences. By using commercially available esters and common oxidants like sodium hypochlorite, the raw material costs are drastically lowered, and the simplified workup reduces processing time and waste disposal expenses.
Q: Is this synthesis suitable for large-scale industrial production?
A: Yes, the reaction conditions are mild (0-50°C) and utilize standard solvents like THF and dichloromethane. The absence of sensitive reagents and the robustness of the TEMPO oxidation step make it highly amenable to commercial scale-up from kilogram to multi-ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Chloro-1,7-Naphthyridine-3-Formaldehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of novel immunomodulators depends on access to high-quality, reliably sourced intermediates. Our technical team has extensively analyzed the synthetic pathway disclosed in CN110950865A and possesses the expertise to execute this route with precision and efficiency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and speed. Our facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, delivering intermediates that meet the exacting standards required for clinical and commercial drug manufacturing.
We invite you to collaborate with us to leverage this advanced synthetic technology for your upcoming projects. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this streamlined route can improve your project economics. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to accelerate your development timeline and secure a competitive advantage in the rapidly evolving field of immunomodulator therapeutics.
