Advanced Synthesis of 8-Chloro-1,7-Naphthyridine-3-Formaldehyde for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic intermediates, particularly those serving as core scaffolds for kinase inhibitors and immunomodulators. Patent CN110964011B, published in June 2022, introduces a groundbreaking synthetic methodology for 8-chloro-1,7-naphthyridine-3-formaldehyde, a critical building block in the development of novel therapeutic agents targeting PDE4 and p38 kinase pathways. This patent addresses the longstanding challenges associated with the construction of the 1,7-naphthyridine ring system, proposing a six-step sequence that begins with the readily available commodity chemical 2-methoxy-3-aminopyridine. By shifting away from precious metal catalysis and hazardous reagents, this innovation offers a compelling value proposition for reliable pharmaceutical intermediate supplier networks aiming to optimize their manufacturing portfolios. The technical depth of this disclosure provides a clear roadmap for overcoming the purity and cost barriers that have historically plagued the production of this specific naphthyridine derivative.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 8-chloro-1,7-naphthyridine-3-carbaldehyde has been fraught with significant technical and economic hurdles, primarily due to the reliance on inefficient multi-step sequences and dangerous reagents. As illustrated in prior art such as WO201354291 and US20170174679, traditional routes often commence with 5-bromo-3-methylpyridine-2-carboxylic acid ethyl ester, necessitating a cumbersome series of urethane exchange, imidization, and oxidation reactions that drive up operational costs. Furthermore, alternative strategies reported in patents like US2014213581 utilize 2-cyano-3-bromo-5-chloropyridine as a starting material, which critically depends on palladium-catalyzed coupling reactions.
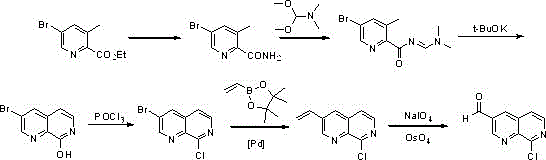
These conventional approaches are not only capital intensive due to the requirement for expensive transition metal catalysts but also pose severe safety risks. The involvement of pressurized reactions and hypertoxic substances such as zinc cyanide creates substantial environmental and occupational health liabilities, rendering these methods unsuitable for modern, green chemical manufacturing standards. The cumulative effect of these drawbacks results in a fragile supply chain where yield losses and purification difficulties significantly impact the final cost of goods, thereby limiting the accessibility of this key intermediate for drug development programs.
The Novel Approach
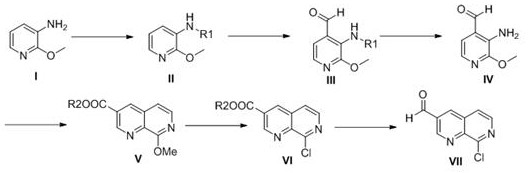
In stark contrast to the convoluted pathways of the past, the methodology disclosed in CN110964011B presents a streamlined and economically viable alternative that fundamentally reimagines the construction of the naphthyridine core. This novel approach leverages 2-methoxy-3-aminopyridine as a strategic starting point, employing a protective group strategy to facilitate selective functionalization before executing a Lewis acid-catalyzed cyclization. By avoiding the need for palladium coupling and toxic cyanide sources, this route drastically simplifies the process flow and enhances the overall safety profile of the manufacturing operation. The integration of a direct reduction step to convert the ester intermediate into the target aldehyde further exemplifies the efficiency of this design, minimizing unit operations and solvent consumption.
The strategic advantage of this new synthesis lies in its modularity and reliance on commodity chemicals, which ensures a stable and predictable supply of raw materials regardless of market fluctuations in precious metals. For procurement teams, this translates to a significant reduction in cost reduction in pharmaceutical intermediates manufacturing, as the elimination of specialized catalysts removes a major variable from the budget. Moreover, the operational simplicity of the reaction conditions, which do not require extreme pressures or cryogenic temperatures beyond standard laboratory capabilities, makes this process exceptionally well-suited for commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and delivery performance.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The cornerstone of this innovative synthetic route is the cyclization step, where the protected amino-aldehyde intermediate reacts with an acrylate compound under the influence of a Lewis acid to form the fused naphthyridine ring system. The patent specifies the use of tetrafluoroborate salts, such as lithium tetrafluoroborate or sodium tetrafluoroborate, which act as potent electrophilic activators to facilitate the intramolecular condensation. This mechanistic pathway is superior to traditional thermal cyclizations because it proceeds under milder conditions, typically between 50°C and 80°C, thereby suppressing the formation of polymeric byproducts and thermal degradation impurities that often compromise the purity of the final API. The choice of solvent, preferably acetonitrile or ethyl acetate, is critical in stabilizing the transition state and ensuring high conversion rates.
Furthermore, the precursor formylation step employs a highly controlled low-temperature protocol, utilizing strong bases like n-butyllithium at temperatures ranging from -100°C to -10°C to generate the reactive lithiated species. This precision in temperature control is essential for directing the regioselectivity of the formylation, ensuring that the aldehyde group is installed exclusively at the desired position on the pyridine ring. Following this, the acidic deprotection step cleanly removes the protecting group to reveal the free amine necessary for the subsequent cyclization. This careful orchestration of protection, functionalization, and deprotection creates a robust impurity control mechanism, resulting in a final product with a superior impurity profile that meets the stringent purity specifications required for clinical trial materials.
How to Synthesize 8-Chloro-1,7-Naphthyridine-3-Formaldehyde Efficiently
The execution of this synthesis requires strict adherence to the specified reaction parameters to maximize yield and minimize side reactions, particularly during the sensitive formylation and cyclization stages. Operators must ensure an inert atmosphere is maintained throughout the process to prevent the quenching of reactive organolithium intermediates, while the sequential addition of reagents must be carefully timed to control exotherms. The detailed procedural nuances, including specific workup protocols and purification techniques, are critical for translating this laboratory-scale success into a reliable manufacturing process. For a comprehensive breakdown of the standardized operating procedures, please refer to the technical guide below.
- Protect the amino group of 2-methoxy-3-aminopyridine using a carbonate reagent to form Compound II.
- Perform formylation under alkaline conditions at low temperature to obtain Compound III, followed by acidic deprotection to yield Compound IV.
- Execute Lewis acid-catalyzed cyclization with an acrylate, followed by chlorination and final reduction to obtain the target aldehyde.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers transformative benefits for supply chain resilience and cost management, addressing the primary pain points faced by procurement managers in the fine chemical sector. By eliminating the dependency on volatile and expensive palladium catalysts, manufacturers can achieve substantial cost savings that are not merely theoretical but are rooted in the fundamental chemistry of the process. The removal of toxic reagents like zinc cyanide also simplifies waste treatment protocols, reducing the environmental compliance burden and associated disposal fees, which contributes to a lower overall cost of production. This economic efficiency allows suppliers to offer more competitive pricing structures without compromising on the quality or reliability of the material supplied.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with inexpensive Lewis acids and commodity starting materials fundamentally alters the cost structure of the synthesis. This shift eliminates the need for costly metal scavenging steps and reduces the raw material expenditure significantly, allowing for a more lean and efficient production model that can withstand market pressures. Additionally, the higher yields reported in the patent examples indicate less material waste, further driving down the unit cost and enhancing the profitability of the manufacturing campaign.
- Enhanced Supply Chain Reliability: Sourcing 2-methoxy-3-aminopyridine and standard acrylates is far less risky than securing specialized halogenated pyridines or maintaining inventories of air-sensitive catalysts. This reliance on widely available bulk chemicals ensures that production schedules are not disrupted by raw material shortages, providing a stable and continuous supply stream for downstream drug manufacturers. The robustness of the process also means that lead times can be consistently met, fostering stronger partnerships between chemical suppliers and pharmaceutical clients.
- Scalability and Environmental Compliance: The absence of high-pressure reactions and hypertoxic substances makes this process inherently safer and easier to scale from pilot plant to full commercial production. Facilities can implement this route without requiring specialized high-pressure reactors or extensive safety upgrades for handling cyanides, accelerating the time to market for new drug candidates. Furthermore, the greener profile of the synthesis aligns with increasingly strict global environmental regulations, future-proofing the supply chain against potential regulatory crackdowns on hazardous chemical usage.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway, drawing directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement officers assessing the long-term viability of this supply source. The answers reflect the specific advantages of the Lewis acid cyclization and the strategic selection of starting materials that define this innovation.
Q: What are the key advantages of this new synthesis route over prior art?
A: The new route avoids expensive palladium catalysts and toxic reagents like zinc cyanide, utilizing commercially available starting materials for a safer and more cost-effective process.
Q: What specific conditions are required for the cyclization step?
A: The cyclization utilizes a Lewis acid such as lithium tetrafluoroborate in solvents like acetonitrile at temperatures between 50-80°C to ensure high yield and purity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process uses standard equipment without high-pressure requirements and avoids hazardous reagents, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 8-Chloro-1,7-Naphthyridine-3-Formaldehyde Supplier
As the demand for sophisticated heterocyclic intermediates continues to grow, partnering with an experienced CDMO like NINGBO INNO PHARMCHEM ensures access to cutting-edge synthetic technologies and unparalleled manufacturing expertise. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to guarantee that every batch meets stringent purity specifications. Our commitment to process optimization allows us to adopt innovative routes like the one described in CN110964011B, delivering high-quality intermediates that accelerate your drug development timelines while maintaining the highest standards of safety and compliance.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this greener, more efficient route. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
