Advanced Asymmetric Hydrogenation for Scalable Production of Chiral Lactone Intermediates
Advanced Asymmetric Hydrogenation for Scalable Production of Chiral Lactone Intermediates
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral building blocks with high stereochemical fidelity, particularly for complex therapeutic agents such as matrix metalloproteinase inhibitors. Patent CN1160350C introduces a groundbreaking process for the preparation of compounds of Formula I, which serve as critical intermediates in the synthesis of pharmacologically active substances. This technology leverages advanced asymmetric hydrogenation techniques using optically active metal diphosphine complexes to achieve superior enantioselectivity. By utilizing specific rhodium-based catalysts, manufacturers can bypass traditional resolution steps that often halve theoretical yields, thereby significantly enhancing overall process efficiency. The ability to precisely control the absolute stereochemistry at multiple centers makes this route invaluable for developing next-generation therapeutics where biological activity is strictly dependent on chirality. Furthermore, the versatility of the substituents R1 and R2 allows for the generation of a diverse library of analogs, facilitating rapid structure-activity relationship studies during drug discovery phases.
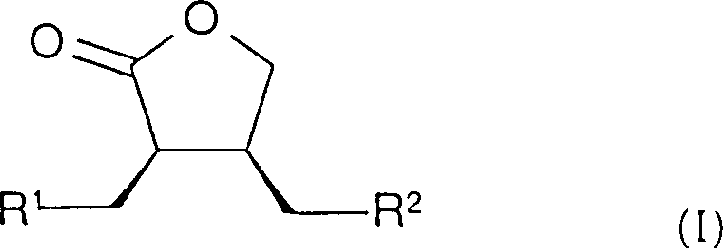
For global procurement teams, understanding the structural integrity of the final product is paramount. The compound of Formula I represents a highly functionalized gamma-lactone scaffold, which is a privileged structure in medicinal chemistry due to its metabolic stability and ability to mimic peptide bonds. The presence of the N-heterocyclic moiety at the R2 position further enhances the molecule's potential for specific receptor binding, making it a versatile precursor for various drug classes including anti-inflammatory and anti-cancer agents. Access to such high-value intermediates through a reliable pharmaceutical intermediate supplier ensures that downstream synthesis remains uninterrupted. The structural complexity depicted in Formula I underscores the necessity for a sophisticated catalytic approach, as conventional chemical methods struggle to install the requisite stereocenters with the necessary precision. Consequently, adopting the methodology outlined in CN1160350C provides a strategic advantage in securing a stable supply of high-purity chiral lactones for commercial drug production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral lactones with adjacent stereocenters has relied heavily on stoichiometric chiral auxiliaries or enzymatic resolutions, both of which present significant logistical and economic challenges for large-scale manufacturing. Stoichiometric methods generate substantial amounts of waste, as the auxiliary must be attached and subsequently removed, adding multiple synthetic steps and reducing the overall atom economy of the process. Enzymatic resolutions, while green, are often limited by substrate specificity and can suffer from low throughput when dealing with bulky or non-natural substrates. Furthermore, traditional chemical hydrogenation using heterogeneous catalysts like Raney Nickel or Palladium on Carbon typically yields racemic mixtures, necessitating expensive and yield-limiting chiral chromatography or crystallization steps to isolate the desired enantiomer. These inefficiencies result in prolonged lead times and inflated costs, creating bottlenecks in the supply chain for high-purity pharmaceutical intermediates. The inability to consistently produce single enantiomers with high optical purity also poses regulatory risks, as impurities with incorrect stereochemistry can exhibit toxicological profiles that jeopardize drug approval.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a homogeneous catalytic system that directly installs chirality during the bond-forming event, effectively eliminating the need for downstream resolution. The core of this innovation lies in the asymmetric hydrogenation of the unsaturated lactone precursor, specifically compounds of Formula II, using chiral rhodium-diphosphine complexes. This method allows for the direct conversion of achiral or racemic starting materials into the desired chiral product with exceptional enantiomeric excess. The process is highly adaptable, accommodating a wide range of N-heterocyclic substituents as defined by R2, including pyrrolidinyl, piperidino, and imidazolidinyl groups. 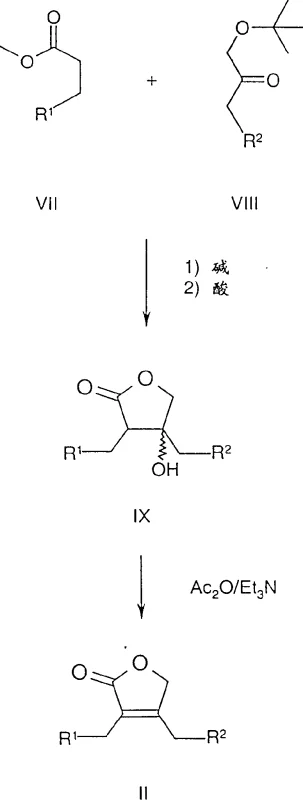 As illustrated in Reaction Scheme A, the precursor itself is synthesized efficiently through a condensation reaction followed by cyclization, ensuring that the entire pathway from simple esters to the final chiral lactone is streamlined. This integrated approach not only reduces the number of unit operations but also minimizes solvent usage and waste generation, aligning with modern principles of green chemistry. For procurement managers, this translates to cost reduction in API manufacturing by simplifying the bill of materials and reducing the dependency on scarce chiral pool starting materials.
As illustrated in Reaction Scheme A, the precursor itself is synthesized efficiently through a condensation reaction followed by cyclization, ensuring that the entire pathway from simple esters to the final chiral lactone is streamlined. This integrated approach not only reduces the number of unit operations but also minimizes solvent usage and waste generation, aligning with modern principles of green chemistry. For procurement managers, this translates to cost reduction in API manufacturing by simplifying the bill of materials and reducing the dependency on scarce chiral pool starting materials.
Mechanistic Insights into Rhodium-Catalyzed Asymmetric Hydrogenation
The success of this synthetic route hinges on the precise interaction between the rhodium metal center and the chiral diphosphine ligand, which creates a sterically defined environment for substrate binding. The catalyst system, generally represented as [Rh(Y)(Ln)]+A-, employs optically active atropisomeric diphosphine ligands such as MeOBIPHEP, BINAP, or DuPHOS to induce asymmetry. During the catalytic cycle, the unsaturated substrate (Formula II) coordinates to the rhodium center, where the bulky substituents on the ligand direct the approach of molecular hydrogen to one specific face of the double bond. This facial selectivity is critical for establishing the (3R, 4R) or (3S, 4S) configuration with high fidelity.  The electronic properties of the ligand, modulated by substituents like methoxy or trifluoromethyl groups, further tune the reactivity of the metal center, allowing for optimization of reaction rates and selectivity. For R&D directors, understanding this mechanism is vital for troubleshooting potential issues related to catalyst deactivation or incomplete conversion. The patent highlights that specific ligand-metal combinations, such as Rh-(R)-3,5-iPr-MeOBIPHEP, are particularly effective, suggesting that fine-tuning the steric bulk around the phosphorus atoms is key to maximizing performance. This level of mechanistic control ensures that the process is robust against minor variations in feedstock quality, a crucial factor for maintaining consistent product quality in a commercial setting.
The electronic properties of the ligand, modulated by substituents like methoxy or trifluoromethyl groups, further tune the reactivity of the metal center, allowing for optimization of reaction rates and selectivity. For R&D directors, understanding this mechanism is vital for troubleshooting potential issues related to catalyst deactivation or incomplete conversion. The patent highlights that specific ligand-metal combinations, such as Rh-(R)-3,5-iPr-MeOBIPHEP, are particularly effective, suggesting that fine-tuning the steric bulk around the phosphorus atoms is key to maximizing performance. This level of mechanistic control ensures that the process is robust against minor variations in feedstock quality, a crucial factor for maintaining consistent product quality in a commercial setting.
Impurity control is another critical aspect addressed by the mechanistic design of this process. The high selectivity of the rhodium catalyst minimizes the formation of diastereomeric byproducts, such as the trans-isomer (Formula Ia), which can be difficult to separate due to similar physical properties. By optimizing reaction parameters such as temperature (60-100°C) and hydrogen pressure (40-100 bar), the formation of these unwanted isomers is suppressed, leading to a cleaner crude product profile. Additionally, the use of specific counterions like hexafluoroantimonate (SbF6) or tetrafluoroborate (BF4) enhances catalyst stability and solubility in organic solvents like ethyl acetate, preventing the precipitation of inactive metal species that could contaminate the final product. This rigorous control over the reaction environment ensures that the resulting chiral lactones meet stringent purity specifications required for pharmaceutical applications. The ability to predict and manage the impurity profile through catalyst selection empowers quality assurance teams to establish tighter control strategies, ultimately reducing the risk of batch failures and ensuring a reliable supply of high-purity chiral lactones for downstream processing.
How to Synthesize Chiral Lactones Efficiently
The synthesis of these valuable intermediates follows a logical progression from readily available starting materials, leveraging well-established organic transformations adapted for high stereocontrol. The process begins with the preparation of the unsaturated lactone precursor, which serves as the substrate for the key asymmetric hydrogenation step. Detailed operational parameters, including specific molar ratios, solvent choices, and workup procedures, are critical for reproducing the high yields and enantioselectivity reported in the patent literature. Manufacturers must pay close attention to the exclusion of oxygen during the hydrogenation phase to prevent catalyst oxidation, which can lead to reduced activity and increased metal contamination.
- Prepare the unsaturated lactone precursor (Formula II) by reacting an ester enolate with a protected ketone, followed by acidification and cyclization.
- Conduct asymmetric hydrogenation using a chiral rhodium-diphosphine complex (e.g., Rh-MeOBIPHEP) under high pressure (40-100 bar) in ethyl acetate.
- Isolate the final chiral lactone (Formula I) through crystallization, achieving high enantiomeric excess (ee) suitable for downstream API synthesis.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this advanced catalytic technology offers profound benefits for supply chain resilience and cost management, addressing several pain points associated with traditional chiral synthesis. The elimination of resolution steps not only improves yield but also drastically simplifies the manufacturing workflow, reducing the requirement for specialized chiral columns or expensive resolving agents. This streamlining effect leads to substantial cost savings by lowering raw material consumption and minimizing waste disposal costs, which are increasingly significant regulatory burdens in the chemical industry. Furthermore, the use of homogeneous catalysts allows for lower operating temperatures compared to some heterogeneous alternatives, reducing energy consumption and enhancing safety profiles in large-scale reactors. For supply chain heads, the reliance on commercially available ligands and standard high-pressure equipment ensures that the process can be easily transferred between manufacturing sites without significant capital investment. This flexibility is essential for mitigating risks associated with geopolitical instability or localized production disruptions, ensuring continuous availability of critical intermediates.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the dramatic improvement in atom economy achieved through direct asymmetric synthesis. By avoiding the 50% theoretical yield loss inherent in racemic resolution processes, manufacturers can effectively double the output from the same amount of starting material. Additionally, the high turnover numbers (TON) achievable with rhodium-diphosphine complexes mean that catalyst loading can be kept low, offsetting the cost of the precious metal and chiral ligands. The simplified purification process further reduces operational expenditures by shortening cycle times and decreasing solvent usage. These factors combine to create a highly competitive cost structure that allows for better margin management in the face of fluctuating raw material prices. Ultimately, this efficiency translates into a more sustainable business model that can withstand market volatility while delivering value to customers.
- Enhanced Supply Chain Reliability: The robustness of the catalytic system contributes significantly to supply chain stability by reducing the sensitivity to feedstock variations. Unlike enzymatic processes that may require strict control of pH and temperature, this chemical catalysis operates effectively over a broader range of conditions, making it less prone to batch-to-batch variability. The availability of multiple ligand options, such as MeOBIPHEP and BINAP, provides a backup strategy in case of supply shortages for specific reagents, ensuring that production schedules remain intact. Moreover, the scalability of the process from laboratory to industrial scale has been demonstrated, giving procurement managers confidence in the ability to ramp up production quickly in response to market demand. This reliability is crucial for maintaining long-term contracts with pharmaceutical clients who require guaranteed delivery of GMP-grade intermediates.
- Scalability and Environmental Compliance: From an environmental perspective, the process aligns well with modern sustainability goals by minimizing waste generation and utilizing greener solvents like ethyl acetate. The high selectivity of the reaction reduces the burden on wastewater treatment facilities, as fewer organic byproducts need to be removed. The ability to recycle the rhodium catalyst, although not explicitly detailed in every example, is a standard practice in homogeneous catalysis that further reduces the environmental footprint and metal consumption. Scaling this process to multi-ton quantities is feasible using standard stainless steel autoclaves, avoiding the need for exotic materials of construction. This ease of scale-up facilitates rapid commercialization of new drug candidates, allowing companies to bring life-saving therapies to market faster while adhering to strict environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral lactone synthesis technology. Understanding these aspects helps stakeholders make informed decisions about integrating this process into their existing manufacturing portfolios. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance.
Q: What is the primary advantage of using Rhodium-BIPHEP catalysts in this synthesis?
A: The use of atropisomeric diphosphine ligands like MeOBIPHEP or BINAP with rhodium allows for exceptional stereocontrol, achieving enantiomeric excess (ee) values exceeding 98% without the need for costly chiral resolution steps.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the process utilizes standard high-pressure hydrogenation equipment and commercially available solvents like ethyl acetate, making it highly amenable to commercial scale-up from kilogram to multi-ton quantities.
Q: What are the typical reaction conditions for the hydrogenation step?
A: Optimal conditions typically involve temperatures between 60-100°C and hydrogen pressures ranging from 40 to 100 bar, ensuring rapid conversion and high selectivity for the desired cis-isomer.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Lactones Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of pharmaceutical products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the demands of both clinical trial material and commercial supply. We are committed to maintaining stringent purity specifications and operate rigorous QC labs to guarantee that every batch of chiral lactones meets the highest industry standards. Our expertise in asymmetric catalysis allows us to optimize routes for maximum efficiency and cost-effectiveness, providing our partners with a distinct competitive advantage in the marketplace. By choosing us as your partner, you gain access to a wealth of technical knowledge and a flexible manufacturing infrastructure capable of handling complex chemistries with precision.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can support your project goals. We offer a Customized Cost-Saving Analysis to help you understand the economic benefits of switching to our optimized synthesis routes. Please reach out to request specific COA data and route feasibility assessments tailored to your target molecules. Our dedicated account managers are ready to provide timely responses and transparent communication, fostering a partnership built on trust and mutual success. Let us help you accelerate your drug development timeline with our reliable supply of premium chiral intermediates.
