Advanced Asymmetric Hydrogenation for High-Purity Chiral Lactone Intermediates
The pharmaceutical industry continuously seeks robust methodologies for constructing chiral scaffolds, particularly those serving as critical intermediates for bioactive molecules. Patent CN1495164A presents a significant technological breakthrough in the synthesis of chiral lactones, specifically targeting the production of compounds defined by Formula I. These structures are not merely academic curiosities but serve as pivotal precursors for matrix metalloproteinase (MMP) inhibitors, a class of drugs with profound implications for treating degenerative joint diseases and invasive tumors. The core innovation lies in the utilization of optically active metal diphosphine complexes, particularly rhodium-based catalysts, to effectuate asymmetric hydrogenation. This approach bypasses the inefficiencies of traditional racemic resolution, offering a direct route to high-purity enantiomers. By leveraging specific ligands such as MeOBIPHEP derivatives, the process achieves exceptional stereocontrol, ensuring that the resulting intermediates meet the stringent purity specifications required for modern drug development. 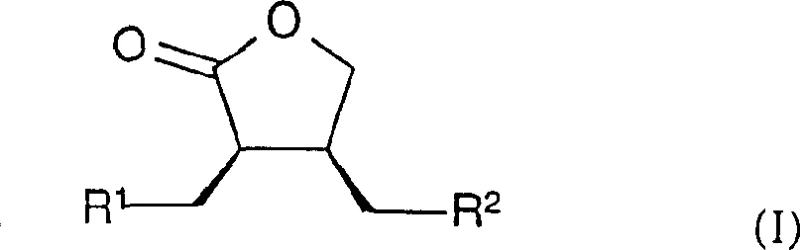
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral lactones relied heavily on the resolution of racemic mixtures, a process inherently flawed by its maximum theoretical yield of 50%. This limitation necessitates the processing of double the amount of raw materials to obtain the desired quantity of the active enantiomer, leading to substantial waste generation and inflated production costs. Furthermore, classical resolution techniques often involve multiple crystallization steps using chiral resolving agents, which introduces additional impurities and complicates the downstream purification process. The use of stoichiometric chiral auxiliaries also adds significant mass to the process, increasing the environmental burden through solvent usage and waste disposal. For supply chain managers, these inefficiencies translate into longer lead times and higher volatility in raw material pricing, making the reliable supply of high-purity pharmaceutical intermediates a persistent challenge. Additionally, older synthetic routes often employed harsh reaction conditions that could compromise the integrity of sensitive functional groups present in complex molecular architectures.
The Novel Approach
In stark contrast, the methodology outlined in CN1495164A employs a catalytic asymmetric hydrogenation strategy that fundamentally alters the economic and technical landscape of chiral lactone manufacturing. By utilizing a chiral rhodium catalyst, the process directly converts prochiral unsaturated precursors (Formula II) into the desired chiral lactones (Formula I) with high enantiomeric excess. This catalytic approach eliminates the 50% yield ceiling associated with resolution, effectively doubling the output from the same amount of starting material. The versatility of this method is further enhanced by its compatibility with a wide range of N-heterocyclic substituents, as illustrated by the diverse ring systems capable of being incorporated into the final structure. 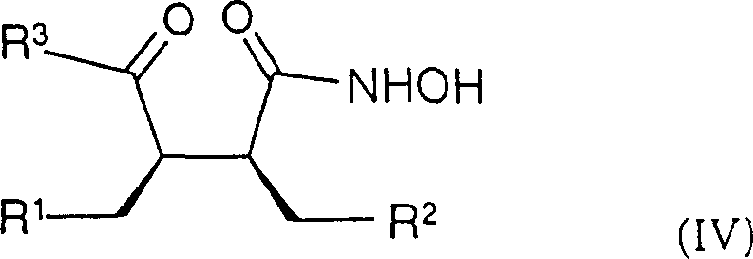
This capability is crucial for developing next-generation therapeutics where specific heterocyclic motifs are required for optimal binding affinity. The process operates under relatively mild conditions compared to alternative asymmetric syntheses, utilizing solvents like ethyl acetate which are favorable for green chemistry initiatives. For procurement teams, this translates to cost reduction in pharmaceutical intermediate manufacturing by minimizing raw material consumption and simplifying the overall process flow. The ability to tune the stereochemistry through catalyst selection provides a strategic advantage, allowing manufacturers to access specific enantiomers without redesigning the entire synthetic route.
Mechanistic Insights into Rh-Catalyzed Asymmetric Hydrogenation
The heart of this technological advancement lies in the sophisticated interaction between the rhodium metal center and the chiral diphosphine ligand. The catalyst system, typically generated in situ from precursors like [Rh(COD)2]SbF6 and ligands such as (R)-3,5-iPr-MeOBIPHEP, creates a highly defined chiral environment around the metal. During the hydrogenation cycle, the unsaturated substrate coordinates to the rhodium center, and the bulky substituents on the phosphine ligands dictate the facial selectivity of hydrogen addition. This steric guidance ensures that hydrogen is delivered to only one face of the double bond, resulting in the formation of a single enantiomer with high fidelity. The patent data indicates that specific catalyst formulations can achieve enantiomeric excess (ee) values exceeding 98%, as demonstrated in Example 4c where conditions of 100 bar pressure and 80°C in ethyl acetate yielded superior results. Such high levels of stereocontrol are essential for R&D directors who must ensure that impurity profiles remain within strict regulatory limits throughout the drug development lifecycle.
Beyond the initial hydrogenation, the patent also discloses a mechanism for stereochemical adjustment through base-mediated epimerization. This process allows for the conversion of the cis-isomer (Formula I) into the trans-isomer (Formula Ia), providing a safety net for process optimization. 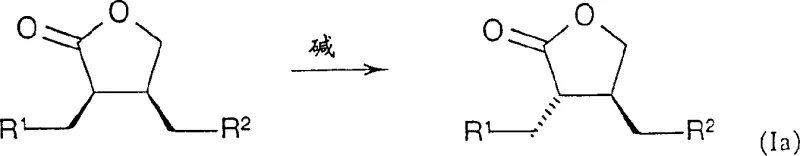
If the primary hydrogenation yields a mixture or the wrong diastereomer, treatment with a base such as DBU at elevated temperatures can equilibrate the stereocenters to the thermodynamically more stable configuration. This flexibility is invaluable for process chemists aiming to maximize yield and purity. Furthermore, the subsequent transformation of these lactones into hydroxamic acid derivatives (Formula IV) involves mild amidation and oxidation steps that preserve the established chirality. The use of TEMPO-mediated oxidation for converting hydroxyl groups to ketones exemplifies the gentle yet effective chemistry employed throughout the sequence, ensuring that the delicate chiral integrity of the molecule is maintained from start to finish.
How to Synthesize Chiral Lactones Efficiently
The synthesis of these high-value intermediates follows a logical progression designed for scalability and reproducibility. The process begins with the preparation of the unsaturated precursor, followed by the critical asymmetric hydrogenation step which sets the stereochemistry. Detailed operational parameters, including catalyst loading, pressure, and temperature, are optimized to balance reaction rate with enantioselectivity. The final isolation involves crystallization techniques that further enhance purity, removing trace catalyst residues and minor impurities. For a comprehensive understanding of the specific reagents and conditions required to replicate this high-efficiency pathway, please refer to the standardized synthesis guide below.
- Preparation of the unsaturated lactone precursor (Formula II) via condensation and cyclization reactions.
- Asymmetric hydrogenation using a chiral Rhodium-diphosphine catalyst (e.g., [Rh((R)-3,5-iPr-MeOBIPHEP)(COD)]SbF6) in ethyl acetate at elevated pressure.
- Purification of the resulting chiral lactone (Formula I) through crystallization to achieve high enantiomeric excess.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented technology offers tangible benefits that extend far beyond the laboratory bench, directly impacting the bottom line and supply chain resilience. For procurement managers, the shift from resolution to asymmetric catalysis represents a fundamental improvement in material efficiency. By eliminating the discard of the unwanted enantiomer, the process significantly reduces the consumption of expensive starting materials and chiral auxiliaries. This reduction in material intensity leads to substantial cost savings in the overall manufacturing budget, allowing for more competitive pricing of the final active pharmaceutical ingredient. Moreover, the catalysts used, while sophisticated, can be employed at low loadings with high turnover numbers, further driving down the cost per kilogram of the product. The reliance on commercially available reagents and standard solvents ensures that the supply chain remains robust and less susceptible to disruptions caused by specialty chemical shortages.
- Cost Reduction in Manufacturing: The elimination of resolution steps removes the inherent 50% yield loss, effectively doubling the output from existing infrastructure without requiring additional capital investment in reactors. This dramatic increase in throughput directly lowers the unit cost of production. Additionally, the simplified workup procedures reduce solvent usage and energy consumption associated with multiple crystallizations and separations. The ability to recycle or recover catalyst components, although dependent on specific plant capabilities, offers further opportunities for expense mitigation. Consequently, the overall cost structure for producing these complex chiral building blocks becomes much more favorable compared to legacy methods.
- Enhanced Supply Chain Reliability: The process utilizes raw materials that are widely available in the global chemical market, reducing the risk of supply bottlenecks. Unlike processes dependent on rare natural products or highly specialized chiral pool starting materials, this synthetic route builds complexity from simpler, commodity-grade chemicals. This accessibility ensures a steady flow of materials even during periods of market volatility. Furthermore, the robustness of the hydrogenation step means that the process is less sensitive to minor variations in feed quality, leading to more consistent batch-to-batch performance and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: High-pressure hydrogenation is a well-established unit operation in the fine chemical industry, meaning that scaling from pilot plant to commercial production involves minimal technical risk. The process avoids the use of toxic heavy metals often associated with stoichiometric chiral reagents, aligning with increasingly stringent environmental regulations. The use of greener solvents like ethyl acetate and the reduction of waste streams through higher atom economy contribute to a smaller environmental footprint. This compliance not only mitigates regulatory risk but also enhances the sustainability profile of the supply chain, a key metric for modern pharmaceutical sourcing strategies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral lactone synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances is critical for evaluating the feasibility of integrating this route into your existing manufacturing portfolio. We encourage technical teams to review these points closely to assess alignment with their specific project requirements.
Q: What is the primary advantage of the Rh-catalyzed method over traditional resolution?
A: The asymmetric hydrogenation method described in CN1495164A avoids the 50% yield loss inherent in racemic resolution, significantly improving atom economy and reducing raw material costs while achieving high enantiomeric purity.
Q: Can the stereochemistry of the lactone be adjusted post-synthesis?
A: Yes, the patent describes a base-mediated epimerization process that allows for the conversion of cis-isomers to trans-isomers (Formula Ia), providing flexibility in accessing different stereoisomers required for specific biological activities.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Absolutely. The process utilizes standard industrial unit operations such as high-pressure hydrogenation and crystallization, and the catalyst loading can be optimized (S/C ratios up to 5000) to ensure economic viability on a multi-ton scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Lactones Supplier
The potential of this asymmetric hydrogenation technology to revolutionize the production of MMP inhibitor intermediates is immense, offering a pathway to higher purity and lower costs. At NINGBO INNO PHARMCHEM, we possess the technical expertise and infrastructure to translate such complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of chiral lactones meets the highest industry standards. Our commitment to quality assurance means that you can rely on us for critical intermediates that demand exacting stereochemical control.
We invite you to engage with our technical procurement team to discuss how we can tailor this synthesis route to your specific volume and cost targets. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this catalytic process. We are prepared to provide specific COA data and route feasibility assessments to support your internal validation processes. Partnering with us ensures not just a supplier, but a strategic ally dedicated to optimizing your supply chain for the future of pharmaceutical manufacturing.
