Advanced Manufacturing of High-Purity Biperiden Intermediates via Stereocontrolled Grignard Synthesis
Advanced Manufacturing of High-Purity Biperiden Intermediates via Stereocontrolled Grignard Synthesis
The pharmaceutical industry continuously seeks robust manufacturing pathways for neurological agents, particularly for Parkinson's disease treatments where stereochemical purity is paramount. Patent CN1525964A introduces a groundbreaking methodology for the preparation of Biperiden, addressing long-standing challenges in stereoselectivity and yield that have plagued conventional synthesis routes. This technology leverages a sophisticated understanding of Diels-Alder cycloaddition dynamics and Grignard reaction equilibria to produce the active aminoalcohol intermediate with exceptional precision. By strictly controlling the exo/endo isomeric ratio of the bicyclic precursors, manufacturers can bypass the inefficient separation steps typical of older processes. For R&D directors and procurement specialists, this represents a significant opportunity to secure a reliable biperiden intermediate supplier capable of delivering material that meets stringent pharmacological specifications without the burden of excessive waste or complex downstream processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
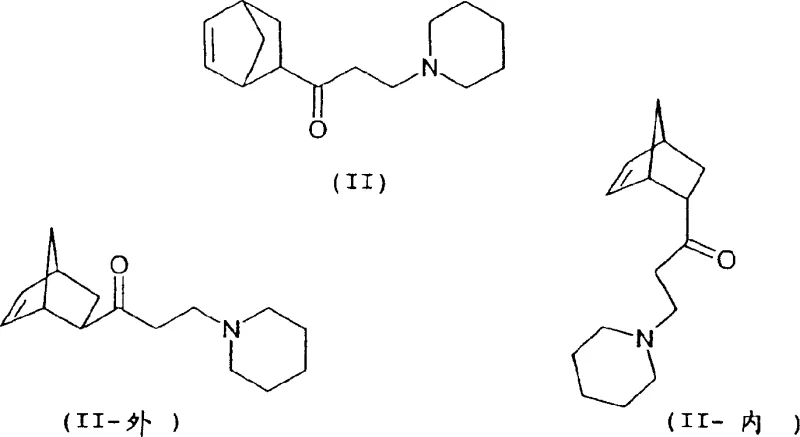
Historically, the synthesis of Biperiden relied on reacting mixtures of 1-(bicyclo[2.2.1]hept-5-en-2-yl)-3-piperidino-1-propanone with phenylmagnesium halides, as described in earlier patents like DE 1 005 067. A critical flaw in these legacy methods was the lack of control over the stereochemistry of the starting ketone precursors. The Diels-Alder reaction between cyclopentadiene and methyl vinyl ketone naturally favors the endo-isomer, typically resulting in a 1:4 exo/endo ratio. Since only the exo-isomer effectively converts into the desired Biperiden (Ia), traditional processes were inherently inefficient, generating large quantities of unwanted diastereomers (Ib, Ic, Id). Furthermore, prior art failed to disclose methods for isolating pure isomers, forcing manufacturers to deal with complex mixtures that required energy-intensive and low-yielding purification sequences. This lack of stereocontrol not only depressed overall yields but also introduced significant variability in the impurity profile, posing risks for regulatory compliance in API manufacturing.
The Novel Approach
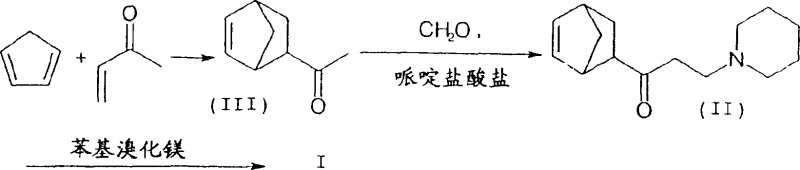
The innovative process detailed in CN1525964A fundamentally alters the production landscape by enforcing strict stereochemical control at the earliest stages of synthesis. Instead of accepting the thermodynamic equilibrium of the Diels-Alder adduct, this method employs a base-catalyzed isomerization strategy to enrich the exo-content of the ethanone precursor (III) to at least 96%. This enriched precursor is then subjected to a Mannich reaction to generate the propanone intermediate (II) with a controlled exo/endo ratio of at least 2.5:1. By ensuring the feedstock is predominantly the reactive exo-isomer, the subsequent Grignard addition proceeds with much higher fidelity towards the target molecule. This proactive approach to isomer management eliminates the need for difficult separations of inactive endo-byproducts later in the stream, thereby streamlining the entire workflow and significantly enhancing the economic viability of producing high-purity biperiden intermediate for commercial scale-up.
Mechanistic Insights into Stereocontrolled Grignard Addition
The core of this technological advancement lies in the optimization of the Grignard reaction conditions to favor the formation of the desired aminoalcohol (I). The patent specifies the use of phenylmagnesium compounds, with a particular preference for phenylmagnesium alkoxides or diphenylmagnesium generated in situ. A key mechanistic feature is the manipulation of the Schlenk equilibrium by adding dioxane to phenylmagnesium halides. This addition precipitates magnesium halide-dioxane complexes, shifting the equilibrium towards the formation of the more reactive diphenylmagnesium species. This shift is crucial for driving the nucleophilic attack on the sterically hindered bicyclic ketone. The reaction is conducted in anhydrous ethers such as THF or diethyl ether at controlled temperatures ranging from -20°C to 70°C. The precise modulation of temperature and the specific molar ratios of the Grignard reagent to the ketone (preferably 1:1 to 1.3:1) ensure that the addition occurs selectively at the carbonyl carbon without inducing unwanted side reactions or epimerization at the chiral centers of the bicyclic ring system.
Following the Grignard addition, the isolation of Biperiden (Ia) from the resulting mixture of four diastereomeric pairs (Ia-Id) is achieved through a highly selective crystallization protocol. The crude mixture, which typically contains at least 50% of the desired Ia enantiomer pair when starting with exo-rich precursors, is treated with hydrogen chloride in a mixture of water and a polar organic solvent like isopropanol. The formation of the hydrochloride salt is performed at elevated temperatures (50-70°C), which aids in solubilizing impurities while allowing the target salt to crystallize upon cooling. Subsequent liberation of the free base is carried out in a biphasic system of water and a dialkyl ether, such as diisopropyl ether, at temperatures above 25°C. This specific thermal condition facilitates the separation of phases and the removal of water-soluble impurities, ultimately yielding Biperiden with a purity of at least 99.0%. This rigorous purification logic ensures that the final product meets the strict quality standards required for neurological therapeutics.
How to Synthesize Biperiden Efficiently
The synthesis of this complex aminoalcohol requires precise adherence to the stereochemical enrichment and Grignard protocols outlined in the patent data. The process begins with the preparation of the exo-enriched ketone, followed by the Mannich condensation and the critical Grignard addition step. Operators must maintain strict anhydrous conditions during the organometallic phase and carefully control the pH and temperature during the salt formation and liberation steps to maximize recovery. The detailed standardized synthesis steps see the guide below for specific operational parameters regarding solvent volumes, reaction times, and temperature ramps that are essential for reproducing the high yields and purity described in the intellectual property.
- Prepare exo-rich 1-(bicyclo[2.2.1]hept-5-en-2-yl)-ethanone (III) via Diels-Alder reaction and isomerization.
- Conduct Mannich reaction with piperidine and formaldehyde to obtain ketone (II) with exo/endo ratio > 2.5: 1.
- React ketone (II) with phenylmagnesium compound followed by hydrochloride salt formation and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented methodology offers substantial strategic benefits beyond mere technical superiority. By resolving the stereochemical bottleneck early in the synthesis, the process drastically reduces the volume of waste solvents and reagents associated with separating inactive isomers. This reduction in material intensity translates directly into cost reduction in pharmaceutical intermediates manufacturing, as fewer distillation columns and chromatography steps are required to achieve the final specification. Furthermore, the reliance on commodity chemicals like cyclopentadiene, methyl vinyl ketone, and piperidine ensures that the raw material supply chain remains robust and less susceptible to the volatility often seen with exotic catalysts or specialized reagents. The ability to source high-purity biperiden intermediate from a supplier utilizing this route mitigates the risk of batch failures due to isomeric impurities, ensuring a consistent flow of material for downstream API production.
- Cost Reduction in Manufacturing: The elimination of extensive purification steps required to separate endo-isomers significantly lowers operational expenditures. By starting with an exo-enriched precursor, the process avoids the costly loss of material inherent in resolving racemic or mixed isomeric streams. The use of in situ generated Grignard reagents further optimizes reagent costs by maximizing atom economy and reducing the need for expensive pre-formed organometallics. Additionally, the simplified workup procedure involving direct crystallization from the reaction mixture minimizes solvent consumption and energy usage associated with evaporation and drying, leading to a leaner and more cost-effective production model that enhances overall margin potential for the final drug product.
- Enhanced Supply Chain Reliability: The synthetic route relies on widely available bulk chemicals, reducing dependency on single-source specialty suppliers. The robustness of the Diels-Alder and Mannich reactions ensures high throughput capabilities, allowing manufacturers to respond quickly to fluctuations in market demand for Parkinson's disease medications. The process stability, characterized by well-defined temperature and pressure parameters, minimizes the risk of unplanned downtime or batch deviations. This reliability is critical for maintaining continuous supply lines for essential neurological therapies, providing procurement teams with the confidence to establish long-term contracts without fear of supply interruptions caused by complex or finicky chemical transformations.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as distillation, crystallization, and liquid-liquid extraction that are easily transferred from pilot plant to commercial scale. The reduction in solvent usage and the avoidance of heavy metal catalysts align with modern green chemistry principles, simplifying waste treatment and environmental compliance. The ability to recycle solvents like diisopropyl ether and isopropanol further enhances the sustainability profile of the manufacturing site. This environmental stewardship not only reduces disposal costs but also positions the supply chain favorably against increasingly stringent global regulations regarding pharmaceutical manufacturing emissions and effluent quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: Why is the exo/endo ratio of the precursor critical for Biperiden synthesis?
A: Only the exo-isomer of the precursor effectively converts into the pharmacologically active Biperiden (Ia). Conventional methods produce mixtures with low exo content, leading to poor yields and difficult purification.
Q: What is the advantage of using phenylmagnesium alkoxide in this process?
A: Using phenylmagnesium alkoxide, particularly generated in situ with dioxane, shifts the Schlenk equilibrium favorably, enhancing reactivity and selectivity compared to standard phenylmagnesium halides.
Q: How does the new purification method improve product quality?
A: The process utilizes a specific sequence of hydrochloride salt formation in aqueous alcohol followed by base liberation in dialkyl ether at elevated temperatures, allowing for the crystallization of Biperiden with purity exceeding 99.0%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biperiden Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity in the production of neurological agents like Biperiden. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate balance of exo/endo isomers is maintained throughout the manufacturing lifecycle. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise quantification of diastereomeric impurities that define the efficacy of the final API. We are committed to translating the innovations found in patents like CN1525964A into reliable, GMP-compliant supply chains that support our partners' global regulatory filings and commercial launch timelines.
We invite you to engage with our technical procurement team to discuss how our advanced synthesis capabilities can optimize your Biperiden supply strategy. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our process efficiencies can lower your total cost of ownership. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project's unique volume and quality requirements, ensuring a seamless transition from development to commercial success.
