Advanced Stereoselective Synthesis of Biperiden for Commercial API Production
Introduction to Advanced Biperiden Manufacturing Technology
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients (APIs), particularly for neurological agents like biperiden, which is a critical anticholinergic medication used in the treatment of Parkinson's disease. The technical landscape for synthesizing this complex molecule has evolved significantly, moving away from crude mixtures towards highly stereoselective processes. Patent CN1247543C represents a pivotal advancement in this domain, addressing the long-standing challenge of isomeric contamination during the synthesis of the key amino alcohol intermediate. This patent discloses a refined methodology that leverages precise pH-controlled extraction techniques to dramatically enhance the exo/endo ratio of the ketone precursor, thereby ensuring a superior quality profile for the final API. For R&D directors and procurement specialists, understanding this technology is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory specifications without compromising on yield or cost-efficiency.
The core innovation lies in the manipulation of the Mannich reaction workup, a step traditionally prone to generating difficult-to-separate diastereomeric mixtures. By implementing a multi-stage extraction protocol that exploits the subtle physicochemical differences between the exo and endo isomers, manufacturers can achieve an exo/endo ratio of at least 4.5:1, with optimized conditions reaching ratios as high as 22:1. This level of stereochemical control is not merely an academic exercise; it translates directly into reduced downstream processing burdens and higher overall process mass intensity (PMI). As we delve deeper into the mechanistic nuances and commercial implications of this process, it becomes clear that adopting such advanced synthetic routes is vital for maintaining competitiveness in the global market for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of biperiden relied on methodologies described in older patents such as DE 1 005 067 and US 2,789,110, which utilized a straightforward Mannich condensation followed by a Grignard addition. However, these conventional approaches suffered from a fundamental flaw: they failed to adequately control the stereochemistry of the intermediate 1-(bicyclo[2.2.1]hept-5-en-2-yl)-3-piperidino-1-propanone (II). Even when starting with high-purity exo-1-(bicyclo[2.2.1]hept-5-en-2-yl)ethanone (III), traditional distillation and workup methods typically yielded a maximum exo/endo ratio of only 4.0:1. This limitation is critical because the endo-isomer does not convert efficiently into the desired pharmacological agent; instead, it generates inactive diastereomers (Ib, Ic, Id) that act as persistent impurities. Removing these structurally similar impurities requires extensive and costly purification steps, such as repeated recrystallizations or preparative chromatography, which drastically reduce the overall yield and increase the cost reduction in API manufacturing. Furthermore, the presence of these isomers complicates the regulatory filing process, as impurity profiles must be rigorously characterized and controlled to meet ICH guidelines.
The Novel Approach
The novel approach detailed in the patent data introduces a sophisticated workup strategy that fundamentally alters the impurity profile of the intermediate. Instead of relying solely on distillation, which often fails to separate the exo and endo isomers effectively due to their similar boiling points, the new method employs a sequential liquid-liquid extraction protocol governed by precise pH adjustments. Initially, the reaction mixture is extracted at a pH not exceeding 7 to remove non-basic organic impurities and unreacted starting materials. Subsequently, the aqueous raffinate is carefully basified to a pH range of 7.5 to 8.5, preferably between 8.1 and 8.3. At this specific alkalinity, the desired exo-isomer exhibits a significantly higher partition coefficient into the organic phase compared to the endo-isomer. This selective extraction allows for the isolation of the ketone intermediate with an exo/endo ratio exceeding 6:1, and under optimal conditions using piperidinium mesylate, ratios up to 22:1 are achievable. 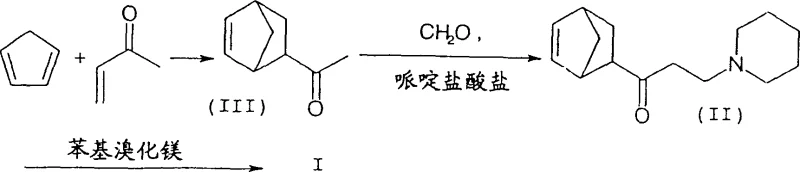 This breakthrough ensures that the subsequent Grignard reaction proceeds with a substrate that is already enriched in the correct stereochemistry, thereby maximizing the formation of the target biperiden (Ia) and minimizing the generation of wasteful byproducts.
This breakthrough ensures that the subsequent Grignard reaction proceeds with a substrate that is already enriched in the correct stereochemistry, thereby maximizing the formation of the target biperiden (Ia) and minimizing the generation of wasteful byproducts.
Mechanistic Insights into Stereoselective Mannich Reaction and Extraction
To fully appreciate the technical depth of this process, one must analyze the stereochemical dynamics of the bicyclic system involved. The core structure, derived from the Diels-Alder cycloaddition of cyclopentadiene and methyl vinyl ketone, exists as exo and endo isomers. While the exo-isomer is thermodynamically favored in certain contexts, the kinetic product of the cycloaddition often contains a significant proportion of the endo-form. The innovation here is not just in the formation of the bond but in the separation of these spatial arrangements. The Mannich reaction introduces a piperidine moiety, creating a chiral center adjacent to the bicyclic system. The interaction between the bulky bicyclic ring and the piperidine nitrogen creates distinct electronic environments for the exo and endo diastereomers. 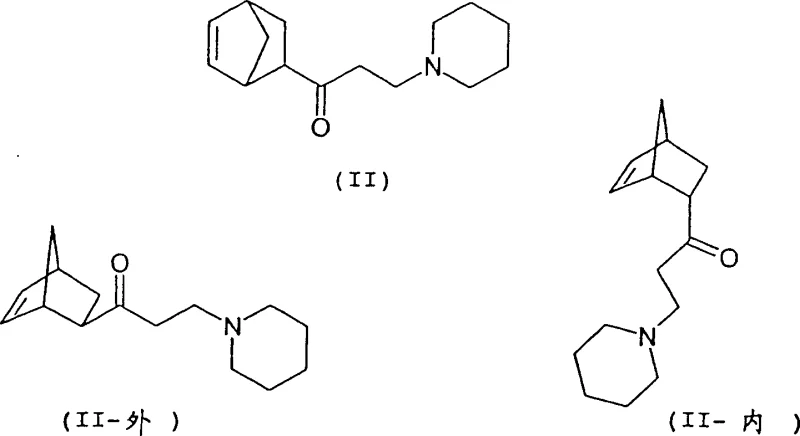 During the extraction phase, the protonation state of the piperidine nitrogen is manipulated. At lower pH levels, both isomers remain protonated and water-soluble. However, as the pH is raised to the narrow window of 8.0-8.5, the exo-isomer deprotonates and becomes lipophilic enough to migrate into the organic solvent (such as diisopropyl ether) more readily than the endo-isomer. This differential solubility is the key mechanism driving the purification, allowing for the effective removal of the endo-contaminant before it can interfere with the final carbon-carbon bond-forming step.
During the extraction phase, the protonation state of the piperidine nitrogen is manipulated. At lower pH levels, both isomers remain protonated and water-soluble. However, as the pH is raised to the narrow window of 8.0-8.5, the exo-isomer deprotonates and becomes lipophilic enough to migrate into the organic solvent (such as diisopropyl ether) more readily than the endo-isomer. This differential solubility is the key mechanism driving the purification, allowing for the effective removal of the endo-contaminant before it can interfere with the final carbon-carbon bond-forming step.
Furthermore, the choice of the Grignard reagent plays a pivotal role in the final diastereoselectivity. The patent highlights the superiority of diphenylmagnesium or phenylmagnesium alkoxides over standard phenylmagnesium halides. The mechanism involves the coordination of the magnesium species with the carbonyl oxygen and potentially the piperidine nitrogen, creating a rigid transition state that favors the attack of the phenyl group from a specific face of the molecule. This coordination minimizes the formation of unwanted diastereomers (Ib-d) and ensures that the ratio of biperiden (Ia) to other isomers is at least 1.5:1, often reaching 2.4:1 in the crude product. This high initial selectivity reduces the burden on the final crystallization step, where the hydrochloride salt is formed to achieve the required >99.0% purity for pharmacological application. Understanding these mechanistic details is crucial for any reliable pharmaceutical intermediate supplier aiming to replicate or scale this process effectively.
How to Synthesize Biperiden Efficiently
The synthesis of biperiden via this optimized route involves a sequence of highly controlled chemical transformations designed to maximize stereochemical integrity. The process begins with the preparation of the exo-enriched ethanone precursor, followed by the critical pH-controlled Mannich reaction, and concludes with a selective Grignard addition and salt crystallization. Each step requires precise monitoring of temperature, pH, and stoichiometry to ensure the exclusion of endo-isomers and other structural impurities. The detailed standardized synthesis steps are outlined below to guide technical teams in implementing this robust methodology.
- Perform Mannich reaction using exo-1-(bicyclo[2.2.1]hept-5-en-2-yl)ethanone, formaldehyde source, and piperidine acid addition salt in an alcohol solvent.
- Extract the reaction mixture at pH ≤ 7 to remove non-basic organic impurities, then adjust pH to ≥ 7.5 to selectively extract the exo-isomer of the ketone intermediate.
- React the purified exo-enriched ketone intermediate with a phenylmagnesium compound (e.g., diphenylmagnesium) followed by crystallization of the hydrochloride salt to isolate pure biperiden.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this advanced synthesis route offers tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the purification train. By achieving a high exo/endo ratio early in the synthesis (at the ketone stage), the need for expensive and time-consuming downstream purification technologies is significantly reduced. Traditional methods often require multiple recrystallizations or complex chromatographic separations to remove endo-derived impurities, which consume large volumes of solvents and result in substantial material loss. In contrast, this pH-selective extraction method achieves high purity through a scalable liquid-liquid separation, which is inherently easier to operate in large-scale reactors. This translates to substantial cost savings in terms of solvent consumption, waste disposal, and labor hours, directly impacting the bottom line of cost reduction in API manufacturing.
- Cost Reduction in Manufacturing: The elimination of inefficient distillation steps and the reduction in solvent usage for purification lead to a leaner manufacturing process. Since the extraction selectively removes impurities before the expensive Grignard step, valuable reagents are not wasted on converting inactive isomers. This efficiency gain means that the overall cost of goods sold (COGS) for the intermediate is lowered, providing a competitive pricing advantage in the global market. Additionally, the use of common solvents like isopropanol and diisopropyl ether ensures that raw material costs remain stable and predictable, avoiding reliance on exotic or volatile reagents.
- Enhanced Supply Chain Reliability: The robustness of the pH-controlled extraction process enhances supply chain continuity. Unlike sensitive catalytic reactions that might be prone to batch-to-batch variability due to catalyst deactivation, this chemical separation method is highly reproducible. The ability to consistently produce intermediate II with an exo/endo ratio of at least 4.5:1 ensures that downstream API synthesis runs smoothly without unexpected delays caused by out-of-specification raw materials. This reliability is critical for reducing lead time for high-purity anticholinergic agents, allowing pharmaceutical companies to maintain steady production schedules and meet market demand without interruption.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this process aligns well with modern green chemistry principles. The avoidance of heavy metal catalysts and the use of aqueous workups reduce the toxic load of the effluent. The scalability of liquid-liquid extraction is well-established in the chemical industry, meaning that scaling from pilot plant to commercial scale-up of complex pharmaceutical intermediates can be achieved with minimal engineering risk. The process generates less hazardous waste compared to traditional methods that rely heavily on distillation residues, facilitating easier compliance with increasingly stringent environmental regulations regarding waste discharge and solvent emissions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of biperiden using this stereoselective methodology. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical principles underlying the process. Understanding these details helps stakeholders make informed decisions regarding supplier qualification and process integration.
Q: Why is the exo/endo ratio critical in biperiden synthesis?
A: Only the exo-isomer of the ketone intermediate effectively converts to the pharmacologically active biperiden (Ia). The endo-isomer leads to inactive diastereomers (Ib-d), complicating purification and reducing overall yield.
Q: How does the patented pH-controlled extraction improve purity?
A: By carefully adjusting the aqueous phase pH to between 7.5 and 8.5 during extraction, the process selectively partitions the desired exo-isomer into the organic phase while leaving the endo-isomer behind, achieving ratios up to 22:1.
Q: What Grignard reagents are preferred for this synthesis?
A: Diphenylmagnesium or phenylmagnesium alkoxides are preferred over standard phenylmagnesium halides, as they demonstrate higher selectivity for the desired diastereomer Ia during the nucleophilic addition step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Biperiden Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stereochemical purity in the production of neurological APIs like biperiden. Our technical team has extensively analyzed the methodologies described in patent CN1247543C and possesses the expertise to implement these advanced pH-controlled extraction techniques at an industrial scale. We understand that achieving an exo/endo ratio of >20:1 requires not just theoretical knowledge but practical mastery of process parameters such as mixing efficiency, phase separation dynamics, and precise pH metering. Our facility is equipped with state-of-the-art reactor systems capable of handling the specific solvent systems and temperature profiles required for this synthesis, ensuring that every batch meets the rigorous quality standards expected by top-tier pharmaceutical companies.
We invite you to collaborate with us to leverage this technology for your supply chain. Our commitment to excellence extends beyond simple manufacturing; we offer a Customized Cost-Saving Analysis to demonstrate how switching to our optimized process can reduce your total procurement costs. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot batches and comprehensive route feasibility assessments. With our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, combined with our stringent purity specifications and rigorous QC labs, NINGBO INNO PHARMCHEM stands ready to be your long-term partner in delivering high-quality biperiden intermediates.
