Industrial Scale Production of Stable 3-Chloromethyl-3-Cephem Derivative Crystals via Alkoxide Cyclization
The pharmaceutical industry continuously seeks robust methodologies for synthesizing key antibiotic intermediates that balance high purity with operational efficiency. Patent CN1580058A introduces a groundbreaking approach for the manufacture of 3-chloromethyl-3-cephem derivative crystals, a pivotal precursor in the production of various cephalosporin antibiotics. Unlike traditional methods that often yield unstable oily residues requiring complex downstream processing, this invention enables the direct formation of stable crystalline products through a continuous, uninterrupted reaction sequence. The core innovation lies in the precise manipulation of solvent systems and pH levels during the cyclization of chlorinated azetidinone derivatives, ensuring that the final product precipitates directly from the reaction mixture with exceptional purity. This technological leap addresses long-standing challenges regarding the stability and handling of cephem intermediates, offering a reliable pathway for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
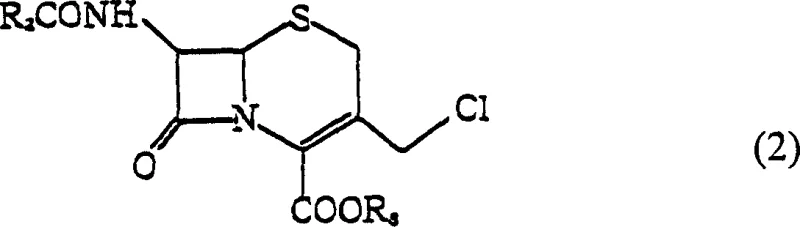
The limitations of conventional synthesis routes for 3-chloromethyl-3-cephem derivatives have historically hindered industrial efficiency and product quality. Prior art methods, such as those described in Japanese Patent Laid-Open No. 58-74689, typically relied on reacting azetidinone derivatives in solvents like N,N-dimethylformamide using weak bases like ammonia. These processes invariably produced the target compound as an oily substance rather than a solid crystal. The oily state presents severe logistical and chemical drawbacks; specifically, the molecule contains a highly reactive chlorine atom that renders it unstable at room temperature. Over time, these oily intermediates tend to release hydrochloric acid, promoting self-decomposition and leading to significant quality degradation before they can even be utilized in subsequent synthesis steps. Furthermore, converting these unstable oils into a usable solid form traditionally required additional, labor-intensive crystallization steps involving cooling and anti-solvent addition, which increased production time and reduced overall throughput.
In stark contrast, the novel approach detailed in the patent data utilizes a sophisticated alkoxide-mediated cyclization strategy that bypasses the formation of unstable oils entirely. By employing a specific combination of alcohol and ether solvents—preferably a mixture of methanol or ethanol with dioxane—the reaction environment is tuned to favor immediate crystallization. The process involves reacting a chlorinated azetidinone derivative with an alkoxide, such as sodium methoxide or sodium ethoxide, under strictly controlled anhydrous conditions. Crucially, the reaction is maintained at a pH of 8 or lower, preferably between 6 and 8, which prevents the alkaline decomposition of the sensitive beta-lactam ring. This method not only simplifies the workflow by eliminating the need for a separate crystallization unit operation but also ensures that the resulting crystals possess excellent long-term stability, making them far superior for storage and transport compared to their oily counterparts.
Mechanistic Insights into Alkoxide-Mediated Cyclization and Crystallization
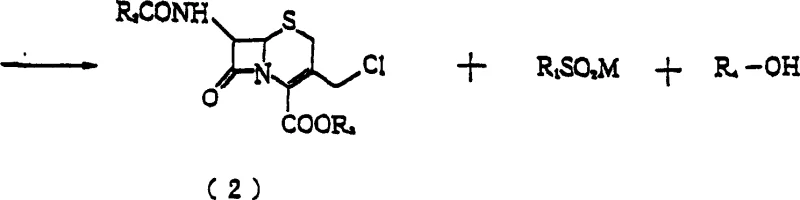
The chemical transformation at the heart of this process involves the intramolecular cyclization of a chlorinated azetidinone derivative to form the cephem nucleus. Mechanistically, the alkoxide acts as a base to deprotonate the azetidinone ring, facilitating the nucleophilic attack that closes the six-membered dihydrothiazine ring characteristic of cephalosporins. However, the success of this reaction hinges on the delicate balance of basicity; while the alkoxide is necessary to drive the cyclization, excess alkalinity (pH > 8) triggers the hydrolysis or degradation of the newly formed cephem structure. The patent data highlights that the reaction proceeds rapidly, necessitating real-time monitoring of the pH to ensure it remains within the narrow window of 6 to 8. This is achieved by the simultaneous or sequential dropwise addition of the alkoxide solution and the substrate solution, allowing the base to be consumed immediately by the reaction rather than accumulating in the bulk medium.
Beyond the reaction kinetics, the crystallization mechanism is driven by the differential solubility properties of the components in the alcohol-ether solvent system. The chosen solvent mixture, typically containing 30% to 95% alcohol by weight, is engineered to dissolve the starting chlorinated azetidinone and the sulfonic acid metal salt by-products, while keeping the 3-chloromethyl-3-cephem product insoluble. As the reaction progresses, the product concentration exceeds its solubility limit in this specific medium, leading to nucleation and crystal growth. The presence of dioxane is particularly critical; if the ether content is too low, impurities tend to co-precipitate with the product, forming agglomerates that lower purity. Conversely, if the ether content is too high, the product may remain dissolved, reducing yield. This precise solubility tuning allows for the direct isolation of high-purity crystals, effectively purifying the compound during the synthesis step itself.
How to Synthesize 3-Chloromethyl-3-Cephem Derivative Efficiently
Implementing this synthesis route requires careful attention to solvent preparation and temperature control to maximize yield and crystal quality. The process begins with the preparation of two distinct feed solutions: one containing the chlorinated azetidinone derivative dissolved in a dioxane-alcohol mixture to manage viscosity, and another containing the alkoxide base dissolved in anhydrous alcohol. These solutions are then introduced into a cooled reaction vessel containing a bulk solvent mixture of alcohol and dioxane. The reaction temperature must be rigorously maintained below 5°C, ideally between -2°C and 2°C, to suppress side reactions and control the rate of crystallization. By adhering to these parameters, manufacturers can achieve a seamless transition from liquid reagents to solid product, streamlining the entire manufacturing workflow. For a detailed breakdown of the specific operational steps and stoichiometric ratios, please refer to the standardized guide below.
- Prepare Solution A by dissolving the chlorinated azetidinone derivative in a mixed solvent of dioxane and anhydrous alcohol (methanol or ethanol) to reduce viscosity.
- Prepare Solution B by dissolving an alkoxide (such as sodium methoxide or sodium ethoxide) in anhydrous alcohol to serve as the cyclization agent.
- Cool a reaction vessel containing an alcohol-dioxane mixture (Solution C) to between -2°C and 2°C, then add a seed portion of Solution A to establish acidic conditions.
- Simultaneously dropwise add the remaining Solution A and Solution B into Solution C while maintaining the reaction pH between 6 and 8 to induce direct crystallization.
- Upon completion, neutralize the slurry with acetic acid, age at low temperature, filter the crystals, and wash with cold alcohol to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this direct crystallization technology offers substantial strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the manufacturing process, which translates directly into cost reduction in pharmaceutical intermediate manufacturing. By eliminating the need for a separate, multi-step crystallization procedure that was previously required to stabilize oily intermediates, facilities can reduce energy consumption, solvent usage, and labor hours. The ability to produce the final crystalline form directly in the reactor removes a major bottleneck in production scheduling, allowing for faster batch turnover and improved asset utilization. Furthermore, the enhanced stability of the crystalline product mitigates the risk of inventory loss due to decomposition, ensuring that valuable materials retain their specification compliance throughout the logistics network.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the removal of complex post-reaction purification steps significantly lower the operational expenditure associated with producing these intermediates. Traditional methods often required expensive Lewis acids or electrolytic processes followed by tedious workups, whereas this alkoxide-based route utilizes readily available, cost-effective reagents. The streamlined one-pot nature of the reaction reduces the total volume of waste solvents generated, thereby lowering disposal costs and environmental compliance burdens. Additionally, the high purity achieved directly from the reactor minimizes the need for resource-intensive recrystallization, further driving down the cost per kilogram of the final active pharmaceutical ingredient precursor.
- Enhanced Supply Chain Reliability: The superior stability of the crystalline 3-chloromethyl-3-cephem derivative ensures a more robust supply chain capable of withstanding longer lead times without quality degradation. Unlike oily intermediates that require strict cold-chain logistics and have short shelf lives, these crystals can be stored under mild conditions, reducing the complexity and cost of warehousing. This stability allows manufacturers to build strategic stockpiles to buffer against raw material fluctuations or unexpected demand surges, providing a critical safety net for global antibiotic production schedules. The consistency of the crystalline form also simplifies quality control testing, accelerating the release of batches for downstream processing.
- Scalability and Environmental Compliance: The reaction conditions described in the patent are highly amenable to commercial scale-up, utilizing standard stainless steel reactors and common organic solvents like methanol, ethanol, and dioxane. The process operates at near-ambient pressures and low temperatures that are easily achievable with standard industrial chilling systems, removing the need for specialized high-pressure or cryogenic equipment. From an environmental perspective, the anhydrous nature of the reaction prevents the formation of aqueous waste streams contaminated with hydrolyzed beta-lactams, simplifying wastewater treatment. The ability to recover and reuse by-product sulfonic acid salts further enhances the green chemistry profile of the process, aligning with modern sustainability goals in fine chemical synthesis.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this crystallization technology. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on process robustness and product performance. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines. For more specific technical data or custom feasibility studies, our team is available to provide detailed support.
Q: Why is the crystalline form of 3-chloromethyl-3-cephem derivatives preferred over the oily form?
A: The crystalline form offers superior long-term stability under mild conditions compared to the oily form, which is prone to self-decomposition and the release of hydrochloric acid at room temperature. This stability significantly reduces storage losses and ensures consistent quality for downstream antibiotic synthesis.
Q: What is the critical role of pH control in this cyclization reaction?
A: Maintaining the reaction pH below 8, ideally between 6 and 8, is essential because the 3-chloromethyl-3-cephem product is extremely unstable in alkaline conditions. Exceeding pH 8 leads to rapid decomposition of the beta-lactam ring, drastically reducing yield and purity.
Q: How does the solvent system contribute to product purity?
A: The specific combination of alcohol and ether (preferably dioxane) creates a solvent environment where the starting material and by-products remain soluble, while the desired product precipitates as crystals. This differential solubility allows for the direct isolation of high-purity crystals without needing complex post-reaction purification steps.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloromethyl-3-Cephem Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of life-saving antibiotics. Our technical team has extensively analyzed the potential of the CN1580058A process and possesses the expertise to implement this advanced crystallization technology at scale. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 3-chloromethyl-3-cephem derivative meets the exacting standards required for GMP-compliant antibiotic synthesis.
We invite pharmaceutical partners to collaborate with us to leverage these process improvements for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance both the economic and operational performance of your antibiotic manufacturing operations.
