Advanced Crystallization Technology for Stable 3-Chloromethyl-3-Cephem Antibiotic Intermediates
Introduction to Patent CN1849324A Technology
The pharmaceutical industry continuously seeks robust manufacturing routes for beta-lactam antibiotics, particularly cephalosporins, where intermediate stability dictates overall process efficiency. Patent CN1849324A introduces a groundbreaking industrially advantageous process for producing 3-chloromethyl-3-cephem derivative crystals, addressing long-standing challenges associated with the instability of traditional oily intermediates. This technology outlines a sophisticated three-step synthetic sequence that culminates in the direct isolation of the target compound as a stable crystalline solid, bypassing the problematic oily state that has historically plagued manufacturers. By integrating precise pH control and a specialized solvent system comprising alcohols and ethers, this method ensures high purity and yield while enabling the recycling of valuable byproducts. For global supply chains, this represents a significant leap forward in reliability, offering a reliable pharmaceutical intermediates supplier the ability to deliver consistent quality without the degradation risks associated with liquid intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-chloromethyl-3-cephem derivatives has been fraught with difficulties stemming from the physical state of the final product. As illustrated in prior art methodologies, such as those referenced in Patent Documentation 7, the reaction typically yields the target compound as an unstable oily substance rather than a solid. This oily form possesses a highly reactive intramolecular chlorine atom, making it extremely susceptible to decomposition even under mild storage conditions. At room temperature, these oily intermediates tend to emit hydrochloric acid, which catalyzes further self-decomposition, leading to a drastic reduction in quality and yield over time. Furthermore, conventional methods often rely on dimethylformamide as a solvent and weak bases like ammonia, which can introduce water into the system, exacerbating hydrolysis and complicating purification. The necessity to convert this unstable oil into a stable form often requires additional, complex crystallization steps from refrigerated alcohol solutions, which are not only operationally burdensome but also result in significant material loss.
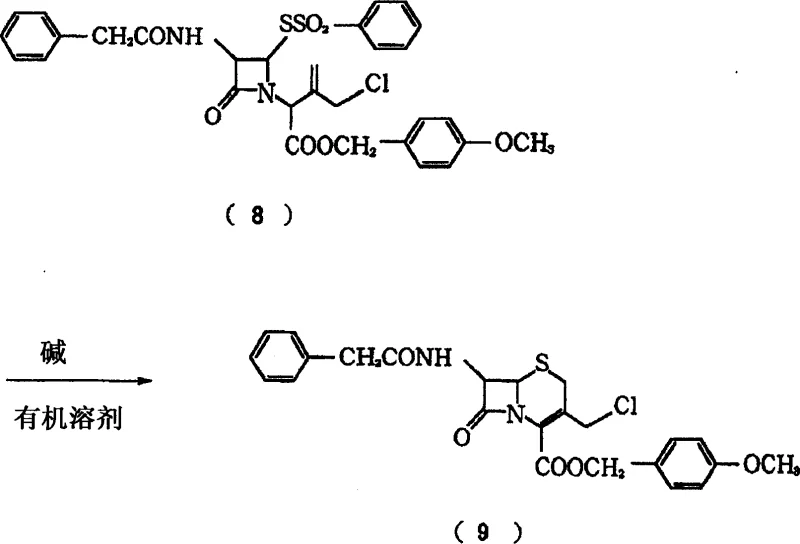
The Novel Approach
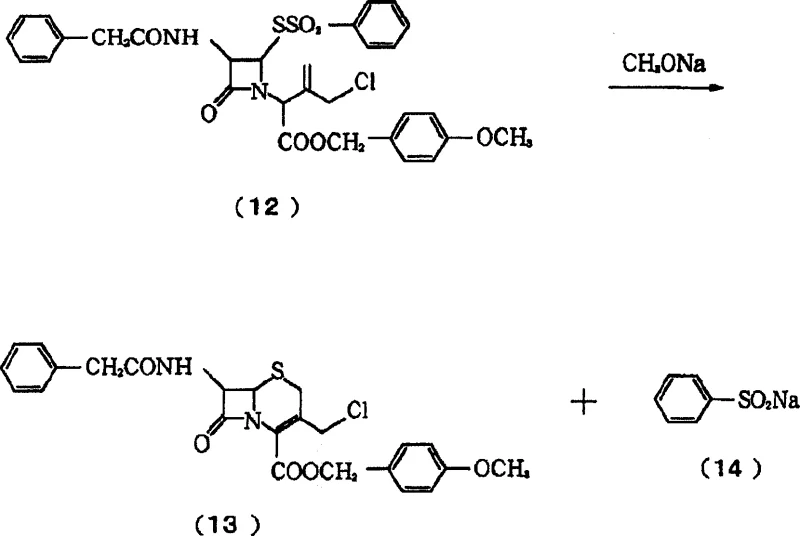
In stark contrast to these legacy methods, the novel approach detailed in CN1849324A achieves the direct formation of stable crystals through a meticulously controlled cyclization step. The core innovation lies in the third operational step, where a chlorinated azetidinone derivative is reacted with an alkoxide in a specific solvent mixture containing both alcohol and ether. Crucially, the reaction is maintained at a pH of 8 or lower, preventing the alkaline decomposition that typically destroys the product in conventional basic conditions. This precise control allows the 3-chloromethyl-3-cephem derivative to precipitate immediately as high-purity crystals, eliminating the need for post-reaction stabilization of an oily residue. Additionally, the process incorporates a closed-loop recycling system where the metal sulfinate byproduct generated during cyclization is recovered and converted back into the sulfonyl halide starting material. This not only enhances the economic viability of cost reduction in API manufacturing but also aligns with modern green chemistry principles by minimizing waste generation and maximizing atom economy throughout the production lifecycle.
Mechanistic Insights into Alkoxide-Mediated Cyclization and Crystallization
The chemical mechanism driving this transformation is a delicate balance of nucleophilic attack and solubility management. In the critical third step, the alkoxide acts as a base to deprotonate the beta-lactam nitrogen, facilitating the ring expansion from the four-membered azetidinone to the six-membered dihydrothiazine ring characteristic of cephems. However, unlike traditional methods that use excess strong base leading to degradation, this process utilizes a controlled addition of alkoxide in a non-aqueous environment. The solvent system is engineered such that the alcohol component dissolves the alkoxide reagent effectively, while the ether component helps modulate the solubility of the growing crystal lattice. By keeping the pH below 8, the system avoids the formation of highly alkaline conditions that would otherwise trigger the elimination of the chloromethyl group or the opening of the sensitive beta-lactam ring. This mechanistic precision ensures that the reaction pathway favors the desired cyclization over competing decomposition pathways, resulting in a product with exceptional chemical integrity and minimal impurity profiles suitable for stringent pharmaceutical applications.
Furthermore, the impurity control mechanism is intrinsically linked to the crystallization dynamics of the solvent system. The specific ratio of alcohol to ether is calibrated to ensure that while the starting chlorinated azetidinone remains soluble, the product 3-chloromethyl-3-cephem derivative reaches its saturation point and precipitates out selectively. This selective precipitation acts as a purification step in itself, excluding soluble byproducts and unreacted starting materials from the crystal lattice. The patent specifies that if the solvent ratio deviates significantly, either the yield drops due to product solubility or the purity suffers due to co-precipitation of impurities. Moreover, the absence of water in the reaction medium is paramount, as water would dissolve the alkoxide, creating localized high-pH zones that degrade the product. This anhydrous, pH-controlled environment ensures that the impurity profile remains exceptionally clean, reducing the burden on downstream purification processes and enhancing the overall robustness of high-purity cephem intermediates production.
How to Synthesize 3-Chloromethyl-3-Cephem Derivative Efficiently
The synthesis protocol described in the patent offers a streamlined pathway for producing this critical antibiotic intermediate with industrial scalability. The process begins with the sulfonylation of a thiazoline azetidinone derivative, followed by chlorination to activate the side chain for cyclization. The final and most critical stage involves the reaction of the chlorinated precursor with an alkoxide, such as sodium methylate, in a mixed solvent of methanol and tetrahydrofuran. Detailed operational parameters, including temperature ranges of -20°C to 5°C and precise pH monitoring, are essential to replicate the high yields reported in the examples. The following guide outlines the standardized synthesis steps derived from the patent data, providing a clear roadmap for technical teams aiming to implement this superior crystallization technology.
- React thiazoline azetidinone derivative with sulfonyl halide in the presence of acid to obtain an azetidinone derivative intermediate.
- Chlorinate the azetidinone derivative in an organic solvent using a chlorinating agent to form the chlorinated azetidinone precursor.
- React the chlorinated precursor with alkoxide in an alcohol-ether solvent mixture at pH 8 or lower to precipitate the product as stable crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this crystallization technology translates into tangible strategic advantages beyond mere chemical yield. The primary benefit lies in the enhanced stability of the final product; because the intermediate is isolated as a crystal rather than an oil, it can be stored for extended periods without significant degradation. This stability drastically simplifies inventory management and reduces the risk of stock spoilage, ensuring a continuous supply of high-quality material for downstream antibiotic synthesis. Furthermore, the ability to recycle the sulfonic acid byproduct back into the process creates a circular economy within the manufacturing plant, significantly lowering the consumption of fresh raw materials and reducing the volume of chemical waste requiring disposal. These factors combined contribute to a more resilient and cost-efficient supply chain capable of meeting the rigorous demands of global pharmaceutical markets.
- Cost Reduction in Manufacturing: The integrated recycling loop for the sulfonyl halide reagent represents a major opportunity for cost optimization. By recovering the metal sulfinate byproduct from the mother liquor and converting it back into the active sulfonyl halide, the process minimizes the net consumption of this expensive reagent. This qualitative reduction in raw material usage directly lowers the variable cost per kilogram of the final intermediate. Additionally, the direct crystallization eliminates the need for complex and yield-loss-prone purification steps associated with oily intermediates, further streamlining the production workflow and reducing utility and labor costs associated with extended processing times.
- Enhanced Supply Chain Reliability: The physical stability of the crystalline product fundamentally improves supply chain reliability compared to unstable oily alternatives. Oily intermediates often require cold chain logistics or immediate usage to prevent decomposition, adding complexity and cost to transportation and storage. In contrast, the stable crystals produced by this method can be handled under standard warehouse conditions, reducing lead time for high-purity antibiotic precursors and allowing for larger batch sizes to be produced and stored safely. This flexibility enables manufacturers to buffer against demand fluctuations and ensures consistent availability for clients requiring just-in-time delivery of critical antibiotic building blocks.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the process is designed for large-scale commercial implementation. The use of common organic solvents like alcohols and ethers, which are easily recoverable and recyclable, aligns with modern environmental regulations regarding volatile organic compound emissions. The reduction in waste generation through byproduct recycling minimizes the environmental footprint of the manufacturing site. Moreover, the robustness of the crystallization step ensures that the process can be scaled from pilot batches to multi-ton production without the unpredictability often associated with handling unstable oily substances, facilitating the commercial scale-up of complex beta-lactams with confidence.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this crystallization process. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the specific advantages over prior art methods. Understanding these nuances is crucial for technical teams evaluating the feasibility of adopting this route for their own manufacturing portfolios.
Q: Why is the crystalline form of 3-chloromethyl-3-cephem derivative preferred over the oily form?
A: The oily form is chemically unstable at room temperature, prone to releasing hydrochloric acid and self-decomposition. The crystalline form offers superior long-term stability and purity, essential for downstream antibiotic synthesis.
Q: How does this process reduce manufacturing costs?
A: The process allows for the recovery and recycling of metal sulfinate byproducts back into sulfonyl halides, significantly reducing raw material consumption and waste disposal costs.
Q: What is the critical parameter for successful crystallization in the final step?
A: Maintaining the reaction pH at 8 or lower is critical. Higher pH levels cause the product to decompose, while the specific alcohol-ether solvent system ensures the product precipitates as high-purity crystals rather than remaining in solution.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Chloromethyl-3-Cephem Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of stable and high-purity intermediates in the synthesis of life-saving antibiotics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated crystallization techniques described in CN1849324A can be seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 3-chloromethyl-3-cephem derivative meets the highest international standards for pharmaceutical use. Our commitment to quality assurance means that clients can rely on us for consistent supply without the risks associated with unstable intermediate forms.
We invite potential partners to engage with our technical procurement team to discuss how this advanced manufacturing route can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching to this crystalline process. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your specific production requirements, ensuring a partnership built on transparency, technical excellence, and mutual growth in the competitive pharmaceutical landscape.
