Advanced Manufacturing of 4(5)-Amino-5(4)-Carbamoylimidazole Derivatives for Oncology Applications
Advanced Manufacturing of 4(5)-Amino-5(4)-Carbamoylimidazole Derivatives for Oncology Applications
The pharmaceutical industry continuously seeks robust synthetic routes for critical heterocyclic intermediates, particularly those serving as the backbone for potent anticancer agents. Patent CN1150169C introduces a transformative methodology for the preparation of 4(5)-amino-5(4)-carbamoylimidazoles, a class of compounds essential for synthesizing life-saving medications such as dacarbazine and temozolomide. This innovation addresses long-standing inefficiencies in existing manufacturing protocols by leveraging a direct cyclization and hydrolysis strategy starting from readily accessible amidine precursors. The core breakthrough lies in the ability to convert N-(2-amino-1,2-dicyanovinyl)amidine derivatives directly into the target imidazole scaffold under mild alkaline conditions, bypassing the hazardous and low-yielding steps characteristic of legacy technologies. For procurement leaders and R&D directors alike, this represents a pivotal shift towards more sustainable and economically viable supply chains for high-value pharmaceutical intermediates.

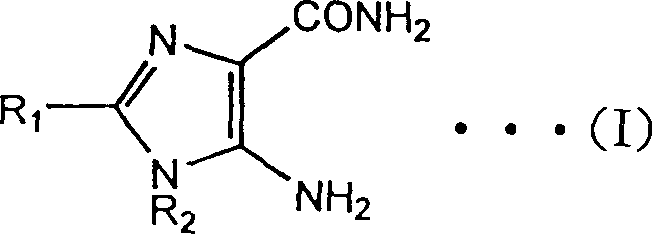
Understanding the structural versatility of these compounds is crucial for downstream application development. The general formula encompasses a wide range of substituents at the nitrogen and carbon positions, allowing for the tailored synthesis of diverse analogues required for various therapeutic pipelines. The ability to manipulate R1 and R2 groups independently provides medicinal chemists with the flexibility to optimize pharmacokinetic profiles without compromising the integrity of the core imidazole ring. This adaptability makes the technology described in CN1150169C not just a single-product solution, but a platform technology capable of supporting a broad spectrum of fine chemical intermediates needed in modern oncology and hepatoprotective drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1H-4(5)-aminoimidazole-5(4)-carboxamide (AICA) and its derivatives has been plagued by significant operational and economic drawbacks. Traditional routes often relied on the contact reduction of 4-nitroimidazole-5-carboxamide or the decomposition of purine nuclei, both of which involve expensive starting materials and complex purification sequences. Perhaps the most notorious bottleneck was the reliance on the Hofmann rearrangement reaction to convert cyano-imidazoles into amino-imidazoles. This classical transformation typically suffers from poor atom economy, requires the handling of hazardous halogenating reagents, and frequently results in low overall yields due to side reactions and difficult isolation steps. Furthermore, earlier attempts to synthesize the necessary amidine intermediates from diaminomaleonitrile (DAMN) yielded dismal results, with some literature reporting conversion rates as low as 2%, rendering such methods entirely impractical for commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent data utilizes a streamlined cyclization-hydrolysis sequence that fundamentally simplifies the manufacturing landscape. By employing N-(2-amino-1,2-dicyanovinyl)amidine (Formula II) as the key precursor, the process enables a one-pot transformation into the target imidazole carboxamide (Formula I) using simple aqueous alkaline solutions. This eliminates the need for toxic organic solvents and aggressive reagents, replacing them with benign bases like sodium hydroxide or potassium hydroxide. The reaction proceeds efficiently at reflux temperatures, driving the cyclization and subsequent nitrile hydrolysis to completion with remarkable selectivity. This methodological shift not only drastically improves the theoretical yield but also simplifies the downstream processing, as the product can often be isolated directly via pH adjustment and crystallization, thereby offering substantial cost reduction in API manufacturing.
Mechanistic Insights into Alkaline Cyclization and Hydrolysis
The chemical elegance of this process lies in the dual functionality of the alkaline medium, which acts simultaneously as a catalyst for ring closure and a reagent for nitrile hydration. When the Formula II intermediate is introduced into the aqueous base, the electron-deficient nitrile groups become susceptible to nucleophilic attack by hydroxide ions. This initiates an intramolecular cyclization where the exocyclic amino group attacks the adjacent nitrile carbon, forming the five-membered imidazole ring. Following ring closure, the remaining nitrile group undergoes hydrolysis to form the primary amide functionality, completing the construction of the 4(5)-amino-5(4)-carbamoylimidazole skeleton. The reaction kinetics are highly favorable under reflux conditions, ensuring that the equilibrium shifts decisively towards the product side, minimizing the formation of partially hydrolyzed byproducts or open-chain impurities that often plague alternative synthetic routes.
Impurity control is another critical aspect where this mechanism excels, particularly concerning the isolation of the final product. The patent specifies that upon completion of the reaction, the aqueous solution can be cooled and the pH carefully adjusted to the isoelectric point of the specific compound, typically within the range of pH 9 to 13. At this specific pH level, the zwitterionic nature of the imidazole derivative minimizes its solubility in water, causing it to precipitate out of the solution as high-purity crystals. This pH-swing crystallization technique is highly effective at excluding soluble salts and organic impurities that remain in the mother liquor. Additionally, the use of activated carbon treatment prior to crystallization can further enhance the visual quality and chemical purity of the final solid, ensuring that the material meets the stringent specifications required for high-purity pharmaceutical intermediates intended for human consumption.
How to Synthesize 4(5)-Amino-5(4)-Carbamoylimidazole Efficiently
Implementing this synthesis route in a production environment requires careful attention to stoichiometry and thermal management to maximize efficiency. The process begins with the preparation of the Formula II intermediate, which can itself be synthesized efficiently from diaminomaleonitrile and various nitriles or orthoformates, ensuring a reliable supply of the starting material. Once the intermediate is secured, the cyclization step is straightforward but demands precise control over the base concentration and reaction time to prevent over-hydrolysis or degradation. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for technical teams aiming to replicate these results.
- Prepare an aqueous alkaline solution using sodium hydroxide or potassium hydroxide, ensuring a concentration suitable for dissolving the Formula II amidine intermediate.
- Add the Formula II compound to the alkaline solution and heat the mixture to reflux temperature, maintaining the reaction for 1 to 48 hours to ensure complete cyclization and nitrile hydrolysis.
- Cool the reaction mixture to room temperature, adjust the pH to the isoelectric point (typically pH 11-12) to precipitate the pure product, and filter the resulting crystals.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain managers and procurement officers, the transition to this novel synthetic route offers compelling strategic benefits that extend far beyond simple chemical yield improvements. The elimination of complex multi-step sequences and hazardous reagents translates directly into a more resilient and cost-effective manufacturing operation. By relying on commodity chemicals like sodium hydroxide and water as the primary reaction media, the process significantly reduces the dependency on specialized, high-cost catalysts or solvents that are subject to volatile market pricing. This stability in raw material sourcing ensures consistent production costs and mitigates the risk of supply disruptions, which is critical for maintaining the continuity of API intermediate supplies to downstream drug manufacturers.
- Cost Reduction in Manufacturing: The streamlined nature of this process inherently lowers the cost of goods sold by reducing the number of unit operations required. Traditional methods involving Hofmann rearrangement necessitate expensive halogenating agents and rigorous safety protocols for handling toxic gases, all of which add significant overhead to the production budget. In contrast, the alkaline cyclization method utilizes inexpensive bases and avoids the generation of hazardous waste streams that require costly disposal. Furthermore, the high selectivity of the reaction minimizes the loss of valuable starting materials to side products, ensuring that a greater proportion of the input mass is converted into saleable product, thereby driving substantial economic efficiency.
- Enhanced Supply Chain Reliability: A major strength of this technology is its reliance on diaminomaleonitrile (DAMN) as a foundational building block, a chemical that is produced on a large industrial scale and is readily available from multiple global suppliers. This abundance of raw materials prevents bottlenecks that often occur when synthesis depends on niche or custom-synthesized precursors. Additionally, the robustness of the aqueous reaction system means that production is less sensitive to minor fluctuations in environmental conditions or reagent quality, leading to more predictable batch cycles and reliable delivery schedules for clients seeking a reliable pharmaceutical intermediate supplier.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process aligns perfectly with modern green chemistry principles. The use of water as the primary solvent eliminates the need for large volumes of volatile organic compounds (VOCs), reducing the facility's carbon footprint and simplifying regulatory compliance. The absence of heavy metal catalysts or persistent organic pollutants in the waste stream facilitates easier wastewater treatment and disposal. This environmental compatibility not only reduces operational risks but also enhances the marketability of the final product to eco-conscious pharmaceutical partners who prioritize sustainable sourcing in their specialty chemical supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, offering clarity on the practical aspects of adopting this method for large-scale production.
Q: What are the primary advantages of this cyclization method over traditional Hofmann rearrangement?
A: This method eliminates the need for hazardous halogenating agents and complex multi-step sequences associated with Hofmann rearrangement, resulting in significantly higher yields and simplified purification processes suitable for industrial scale-up.
Q: How is high purity achieved in the final 4(5)-aminoimidazole-5(4)-carboxamide product?
A: High purity is achieved through precise pH control during the crystallization phase, specifically adjusting the alkaline hydrolysate to the isoelectric point (pH 9-13), which allows for the selective precipitation of the target compound while leaving impurities in the mother liquor.
Q: Is this process scalable for commercial production of anticancer drug intermediates?
A: Yes, the process utilizes readily available industrial raw materials like diaminomaleonitrile (DAMN) and common bases like sodium hydroxide in aqueous media, making it highly adaptable for large-scale manufacturing without requiring exotic catalysts or extreme pressure conditions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4(5)-Amino-5(4)-Carbamoylimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By leveraging advanced synthetic strategies like the alkaline cyclization process described in CN1150169C, we can offer our partners a competitive edge through superior product quality and consistent supply reliability.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need to evaluate the feasibility of scaling this specific imidazole derivative, our experts are ready to assist. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
