Advanced Manufacturing of 4-Amino-5-Carbamoylimidazole Derivatives for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for critical heterocyclic building blocks, particularly those serving as precursors for potent anticancer agents. Patent CN1374951A introduces a transformative methodology for the preparation of 4(5)-amino-5(4)-carbamoylimidazoles, a class of compounds essential for synthesizing drugs like dacarbazine and temozolomide. This innovation addresses long-standing inefficiencies in existing manufacturing protocols by leveraging diaminomaleonitrile (DAMN) as a versatile and industrially accessible starting material. The core breakthrough lies in a streamlined cyclization and hydrolysis sequence conducted in an alkaline aqueous environment, which drastically simplifies the operational complexity compared to legacy methods. By optimizing reaction conditions and purification strategies, this process delivers high-purity intermediates suitable for stringent pharmaceutical applications. 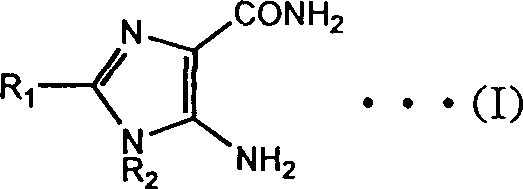 For procurement and supply chain leaders, this represents a significant opportunity to secure a more reliable pharmaceutical intermediate supplier capable of meeting global demand with improved cost structures.
For procurement and supply chain leaders, this represents a significant opportunity to secure a more reliable pharmaceutical intermediate supplier capable of meeting global demand with improved cost structures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1H-4(5)-aminoimidazole-5(4)-carboxamide (AICA) and its derivatives has been plagued by cumbersome multi-step sequences that hinder industrial scalability. Traditional routes often rely on the Hofmann rearrangement of cyanoimidazoles, a reaction notorious for its low yields and the requirement for hazardous halogenated reagents. Other methods involve the catalytic reduction of nitroimidazoles or the decomposition of purine nuclei, both of which suffer from poor atom economy and difficult purification profiles. Furthermore, earlier attempts to utilize DAMN often resulted in impractical yields, such as the mere 2% reported in older literature when reacting with formamidine acetate. These legacy processes frequently necessitate the use of environmentally damaging solvents like chloroform and require cryogenic conditions, imposing heavy burdens on waste management and energy consumption. Consequently, the final products often require extensive recrystallization to meet purity standards, driving up production costs and extending lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a direct and efficient condensation of DAMN with readily available nitriles or orthoformates to generate key amidine intermediates. 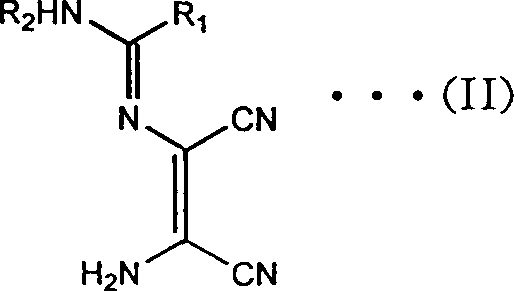 This strategy bypasses the need for dangerous rearrangement reactions and instead employs a straightforward cyclization in alkaline media. The process allows for the use of common industrial solvents like methanol or ethanol, facilitating easier recovery and recycling. By operating at moderate temperatures ranging from room temperature to reflux, the method significantly reduces energy expenditure and safety risks associated with exothermic low-temperature reactions. The ability to isolate intermediates as stable salts further enhances the robustness of the supply chain, allowing for flexible production scheduling. This modernization of the synthetic route ensures cost reduction in API manufacturing by minimizing unit operations and maximizing the throughput of valuable active pharmaceutical ingredients.
This strategy bypasses the need for dangerous rearrangement reactions and instead employs a straightforward cyclization in alkaline media. The process allows for the use of common industrial solvents like methanol or ethanol, facilitating easier recovery and recycling. By operating at moderate temperatures ranging from room temperature to reflux, the method significantly reduces energy expenditure and safety risks associated with exothermic low-temperature reactions. The ability to isolate intermediates as stable salts further enhances the robustness of the supply chain, allowing for flexible production scheduling. This modernization of the synthetic route ensures cost reduction in API manufacturing by minimizing unit operations and maximizing the throughput of valuable active pharmaceutical ingredients.
Mechanistic Insights into Alkaline Cyclization and Hydrolysis
The heart of this technological advancement is the base-mediated transformation of the acyclic amidine precursor into the target imidazole ring system. When the compound of formula (II) is subjected to an alkaline aqueous solution, typically using sodium or potassium hydroxide, a concerted cyclization and hydrolysis mechanism is initiated. The hydroxide ions attack the electrophilic nitrile carbons, promoting intramolecular nucleophilic attack by the adjacent amino group to close the five-membered imidazole ring. Simultaneously, the remaining nitrile functionality undergoes hydrolysis to form the primary carboxamide group, completing the structural framework of formula (I). 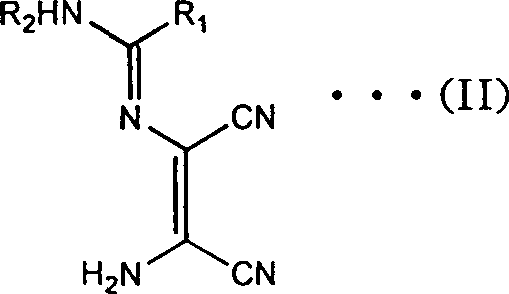 This dual transformation occurs efficiently under reflux conditions, driven by the thermodynamic stability of the aromatic imidazole core. The use of water as the primary reaction medium not only serves as a reactant for hydrolysis but also acts as a heat sink, ensuring precise temperature control throughout the exothermic process.
This dual transformation occurs efficiently under reflux conditions, driven by the thermodynamic stability of the aromatic imidazole core. The use of water as the primary reaction medium not only serves as a reactant for hydrolysis but also acts as a heat sink, ensuring precise temperature control throughout the exothermic process.
Purification is ingeniously managed through pH manipulation, exploiting the amphoteric nature of the resulting imidazole derivatives. Upon completion of the reaction, the solution is cooled, and the pH is carefully adjusted to the isoelectric point of the specific compound, typically within the range of 9 to 13. At this specific pH level, the solubility of the target molecule reaches its minimum, causing it to precipitate selectively as high-purity crystals while leaving inorganic salts and polar impurities in the aqueous phase. This crystallization-induced purity enhancement eliminates the need for column chromatography or extensive solvent extractions, which are often bottlenecks in fine chemical production. The result is a product with a well-defined impurity profile, meeting the rigorous specifications required for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 4(5)-Amino-5(4)-Carbamoylimidazole Efficiently
Implementing this synthesis requires strict adherence to the optimized stoichiometry and thermal profiles outlined in the patent examples to ensure maximum yield and reproducibility. The process begins with the preparation of the amidine intermediate, followed by the critical alkaline cyclization step where water and base are added in precise molar ratios. Operators must monitor the reaction progress closely, maintaining reflux temperatures for the designated duration to ensure complete conversion of the starting materials. Following the reaction, the cooling rate and pH adjustment speed are critical parameters that influence crystal morphology and filtration efficiency. Detailed standardized synthesis steps are provided below to guide technical teams in replicating this high-efficiency protocol.
- Prepare the reaction mixture by adding water and a strong base such as sodium hydroxide to the N-(2-amino-1,2-dicyanovinyl)formamidine intermediate.
- Heat the solution under reflux conditions for a specified duration to facilitate simultaneous cyclization and hydrolysis of the nitrile groups.
- Cool the reaction mixture to room temperature, adjust the pH to the isoelectric point (approximately pH 11-12) to precipitate the pure crystalline product.
Commercial Advantages for Procurement and Supply Chain Teams
For strategic sourcing professionals, the adoption of this patented methodology translates into tangible improvements in supply chain resilience and total cost of ownership. By shifting away from multi-step sequences involving hazardous reagents, manufacturers can significantly reduce the regulatory burden and insurance costs associated with handling dangerous chemicals. The reliance on commodity chemicals like DAMN and simple alkalis ensures that raw material availability remains stable even during market fluctuations, mitigating the risk of production stoppages. Furthermore, the simplified workup procedure reduces the volume of organic waste generated, aligning with increasingly strict environmental compliance standards and lowering disposal fees. These factors collectively contribute to a more sustainable and economically viable manufacturing model for high-value heterocyclic intermediates.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and halogenated solvents directly lowers the variable cost per kilogram of the final product. By utilizing aqueous reaction media and avoiding complex purification techniques like chromatography, the process minimizes solvent consumption and recovery costs. The high yield achieved in the cyclization step means less raw material is wasted, improving the overall material balance and reducing the cost of goods sold. Additionally, the ability to recycle alcohol solvents used in the intermediate synthesis further enhances the economic efficiency of the entire production line.
- Enhanced Supply Chain Reliability: Sourcing diaminomaleonitrile and basic alkalis is far more predictable than procuring specialized reagents required for Hofmann rearrangements. The robustness of the reaction conditions allows for production in a wider range of facilities, diversifying the potential manufacturing base and reducing dependency on single-source suppliers. The stability of the intermediate salts allows for inventory buffering, enabling manufacturers to respond quickly to sudden spikes in demand from downstream API producers. This flexibility is crucial for maintaining continuity of supply for critical oncology medications that rely on these intermediates.
- Scalability and Environmental Compliance: The use of water as a primary solvent inherently reduces the fire hazard and volatile organic compound (VOC) emissions associated with large-scale organic synthesis. The process generates primarily inorganic salt waste, which is easier and cheaper to treat compared to toxic organic sludge from halogenated processes. Scalability is facilitated by the homogeneous nature of the alkaline hydrolysis, which transfers easily from laboratory glassware to industrial stainless steel reactors without significant engineering hurdles. This ease of scale-up ensures that commercial production volumes can be ramped up rapidly to meet global pharmaceutical needs without compromising quality.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing supply chains. The answers are derived directly from the experimental data and process descriptions found within the patent documentation. This transparency ensures that all parties have a clear understanding of the capabilities and limitations of the method.
Q: What are the primary advantages of this DAMN-based synthesis route over traditional Hofmann rearrangement?
A: The DAMN-based route eliminates the need for hazardous halogenated solvents and complex low-temperature operations, significantly improving operational safety and reducing waste treatment costs while achieving higher overall yields.
Q: How does the pH adjustment step contribute to product purity?
A: Adjusting the pH to the specific isoelectric point of the target imidazole derivative allows for selective precipitation, effectively separating the product from soluble inorganic salts and organic impurities without extensive chromatography.
Q: Is this process scalable for commercial production of anticancer drug intermediates?
A: Yes, the process utilizes common industrial raw materials like diaminomaleonitrile and operates in aqueous media, making it highly suitable for large-scale manufacturing with simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4(5)-Amino-5(4)-Carbamoylimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 4(5)-amino-5(4)-carbamoylimidazole meets the highest international standards. Our commitment to process excellence allows us to deliver consistent quality while maintaining the flexibility required for custom synthesis projects.
We invite you to collaborate with us to optimize your supply chain for these vital pharmaceutical building blocks. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals. Let us be your partner in driving efficiency and innovation in your pharmaceutical manufacturing operations.
