Advanced Metal-Catalyzed Synthesis of 1'-Beta-Methyl Azetidinone Intermediates for Commercial Scale-Up
Advanced Metal-Catalyzed Synthesis of 1'-Beta-Methyl Azetidinone Intermediates for Commercial Scale-Up
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex antibiotic scaffolds, particularly carbapenems, which remain critical in treating resistant bacterial infections. Patent CN1277819C discloses a groundbreaking process for producing azetidinone compounds that serve as vital intermediates in the synthesis of 1β-methyl carbapenem-type antibacterial agents. This technology addresses long-standing challenges in stereocontrol and process stability by utilizing a specific metal-catalyzed coupling reaction. Unlike traditional methods that rely on unstable intermediates or harsh conditions, this novel approach enables the selective formation of the desired 1'-beta configuration under mild reaction parameters. By leveraging transition metal catalysis, manufacturers can achieve superior yields and purity profiles, directly impacting the cost-efficiency and reliability of the supply chain for high-purity pharmaceutical intermediates.
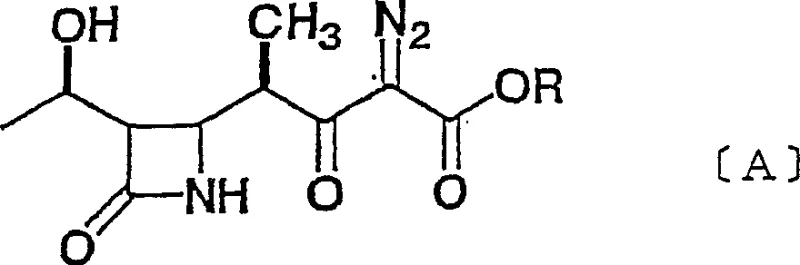
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1'-beta-methyl azetidinone intermediates has been plagued by significant technical and economic hurdles. Prior art methods, such as those described in Japanese Patent Publications No. 57-123182 and No. 64-25779, often necessitate the use of prohibitively expensive starting materials that drive up the overall cost of goods. Furthermore, these conventional routes typically involve multi-step sequences that are operationally cumbersome, leading to cumulative yield losses at each stage. Another critical drawback found in existing literature, specifically in methods resembling Reaction Scheme 4, is the reliance on unstable silyl enol ethers. These intermediates are notoriously difficult to handle on an industrial scale due to their sensitivity to moisture and thermal instability, posing risks to both process safety and batch-to-batch consistency. Consequently, these factors render many traditional processes economically unviable for large-scale commercial production.
The Novel Approach
The methodology outlined in CN1277819C represents a paradigm shift by streamlining the synthetic route into a direct coupling reaction. This innovative process reacts a specific azetidinone derivative (Formula 2) directly with a diazo compound (Formula 3) in the presence of a defined metal compound (Formula 4) and a base. This strategy effectively bypasses the need for generating unstable silyl enol ether intermediates, thereby simplifying the workflow and reducing the number of unit operations. The reaction proceeds under mild conditions, typically between -70°C and 0°C, which minimizes thermal degradation and side reactions. By optimizing the choice of metal catalyst and base, the process achieves high stereoselectivity, preferentially forming the therapeutically relevant 1'-beta isomer over the alpha isomer. This direct approach not only enhances operational simplicity but also significantly improves the overall atom economy and throughput of the manufacturing process.
Mechanistic Insights into Metal-Catalyzed Stereoselective Coupling
The core of this technological advancement lies in the precise interaction between the diazo species and the metal catalyst. The metal compound, selected from Group 4, 12, 13, or 14 metals such as Titanium (Ti), Zinc (Zn), or Aluminum (Al), acts as a Lewis acid to activate the diazo carbonyl system. When combined with a base such as tributylamine or N-ethylpiperidine, the system generates a reactive metal-carbenoid or activated complex that facilitates the nucleophilic attack on the azetidinone ring. This mechanism is crucial for controlling the stereochemistry at the 1'-position. Experimental data from the patent indicates that the ratio of the desired beta-isomer to the alpha-isomer can exceed 95:5 under optimized conditions. For instance, the use of Titanium Tetrachloride (TiCl4) in conjunction with specific amines has demonstrated exceptional control over the stereochemical outcome, ensuring that the final product meets the rigorous purity specifications required for downstream antibiotic synthesis.
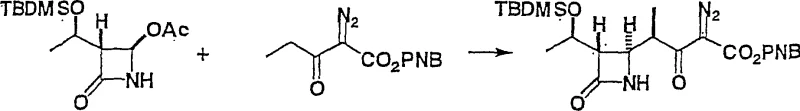
Impurity control is another critical aspect managed by this catalytic system. In conventional non-catalyzed or poorly catalyzed reactions, the formation of byproducts such as dimerization products of the diazo compound or hydrolysis of the beta-lactam ring can be prevalent. However, the specific combination of metal halides and organic bases described in this patent creates a buffered environment that suppresses these deleterious side reactions. The base serves not only to neutralize acidic byproducts generated during the reaction but also to modulate the reactivity of the metal center, preventing overly aggressive conditions that could compromise the integrity of the sensitive four-membered azetidinone ring. This delicate balance ensures that the impurity profile remains clean, reducing the burden on downstream purification steps like chromatography or crystallization, which is a key factor in reducing manufacturing costs.
How to Synthesize 1'-Beta-Methyl Azetidinone Efficiently
Implementing this synthesis route requires careful attention to reaction parameters to maximize yield and selectivity. The process begins with the preparation of the catalytic system in an anhydrous organic solvent, followed by the controlled addition of substrates. The patent provides detailed guidance on molar ratios, suggesting a slight excess of the diazo compound (0.7 to 4 equivalents) relative to the azetidinone substrate to drive the reaction to completion. Temperature control is paramount, with the reaction typically initiated at low temperatures (e.g., -40°C) to manage the exotherm and ensure high stereoselectivity. Detailed standardized synthesis steps for replicating this high-efficiency pathway are provided in the guide below.
- Prepare the reaction system under an inert atmosphere (nitrogen or argon) using a suitable organic solvent such as dichloromethane or toluene.
- Mix the diazo compound (Formula 3) with a specific metal compound (Formula 4, e.g., TiCl4) and a base (e.g., tributylamine) at low temperatures ranging from -70°C to 0°C.
- Add the azetidinone substrate (Formula 2) to the mixture, maintain the temperature for 1 to 3 hours, and isolate the product via extraction and chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-catalyzed process offers tangible strategic benefits beyond mere technical feasibility. The elimination of unstable intermediates like silyl enol ethers translates directly into enhanced supply chain reliability, as the process is less susceptible to disruptions caused by reagent degradation or specialized storage requirements. Furthermore, the use of commodity chemicals such as titanium tetrachloride and common amines ensures that raw material sourcing is robust and cost-effective, mitigating the risk of supply bottlenecks associated with exotic or proprietary reagents. This stability allows for more accurate forecasting and inventory management, crucial for maintaining continuous production lines in the competitive pharmaceutical market.
- Cost Reduction in Manufacturing: The streamlined nature of this process inherently drives down manufacturing costs by reducing the total number of processing steps. By avoiding the isolation and purification of unstable intermediates, manufacturers save on solvent usage, energy consumption, and labor hours. Additionally, the high stereoselectivity reduces the loss of material to unwanted isomers, improving the overall yield of the valuable beta-configured intermediate. While specific percentage savings depend on plant-specific variables, the qualitative reduction in waste generation and the avoidance of expensive specialty reagents contribute to a substantially lower cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate a range of solvents including dichloromethane, toluene, and acetonitrile, provides flexibility in sourcing. If one solvent becomes scarce or expensive due to market fluctuations, the process can be adapted to use alternatives without compromising performance, as evidenced by the patent's data on solvent screening. This flexibility ensures that production schedules remain uninterrupted, safeguarding against the volatility of the global chemical supply chain and ensuring consistent delivery of high-purity pharmaceutical intermediates to downstream partners.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method aligns well with modern green chemistry principles. The reaction operates at relatively mild temperatures compared to high-energy thermal processes, reducing the carbon footprint associated with heating and cooling. Moreover, the high selectivity minimizes the generation of complex waste streams that require expensive treatment. The ability to scale this reaction from gram-scale laboratory experiments to multi-kilogram production batches without significant loss in efficiency makes it an ideal candidate for commercial scale-up of complex pharmaceutical intermediates, ensuring long-term viability and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this azetidinone synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a reliable basis for decision-making. Understanding these nuances is essential for R&D teams evaluating process transfer and procurement teams assessing vendor capabilities.
Q: What is the primary advantage of this metal-catalyzed method over conventional routes?
A: This method eliminates the need for unstable silyl enol ethers and expensive starting materials used in prior art, allowing for a shorter synthesis route with higher stereoselectivity (beta:alpha ratios up to 98:2) under mild conditions.
Q: Which metal compounds are most effective for this transformation?
A: The patent identifies titanium tetrachloride (TiCl4), zinc chloride (ZnCl2), zirconium tetrachloride (ZrCl4), and aluminum chloride (AlCl3) as highly effective catalysts, with TiCl4 showing particularly high yields and selectivity.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process utilizes commercially available solvents and reagents, operates at manageable temperatures (-40°C to 5°C), and avoids hazardous or unstable intermediates, making it robust for industrial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azetidinone Compound Supplier
The technological potential of this metal-catalyzed synthesis route is immense, offering a clear pathway to more affordable and accessible carbapenem antibiotics. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into industrial reality. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of verifying the critical beta:alpha isomer ratios and impurity profiles required for this sensitive chemistry. We understand the complexities of handling reactive metal catalysts and diazo species, and our engineering teams are adept at designing safe, efficient reactors for these specific transformations.
We invite global partners to collaborate with us to leverage this advanced manufacturing capability. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage potential clients to contact us to obtain specific COA data from our pilot batches and comprehensive route feasibility assessments. Let us help you secure a stable, high-quality supply of these critical intermediates, driving efficiency and innovation in your antibiotic development pipeline.
