Optimizing Carbapenem Antibiotic Intermediates via Controlled Seed Crystallization for Industrial Scale-Up
Introduction to Advanced Crystallization Technologies for Beta-Lactam Synthesis
The pharmaceutical industry continuously seeks robust methodologies to enhance the quality and stability of critical antibiotic intermediates, particularly those serving as the backbone for broad-spectrum carbapenem antibiotics. Patent CN101432289B introduces a groundbreaking approach to the crystallization of azetidinone compounds, which are indispensable precursors in the synthesis of 1-beta-methylcarbapenem derivatives. This technology addresses long-standing challenges in the production of these high-value intermediates by fundamentally altering the crystallization dynamics through the strategic use of seed crystals and hydrocarbon solvents. By shifting the paradigm from traditional precipitation methods to a controlled growth mechanism, manufacturers can achieve unprecedented levels of purity and physical stability. This report analyzes the technical merits of this patented process, offering deep insights for R&D directors and supply chain leaders looking to optimize their production of reliable pharmaceutical intermediate supplier materials. The implications of this technology extend beyond mere chemical purity, touching upon critical operational metrics such as filtration efficiency and long-term storage stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
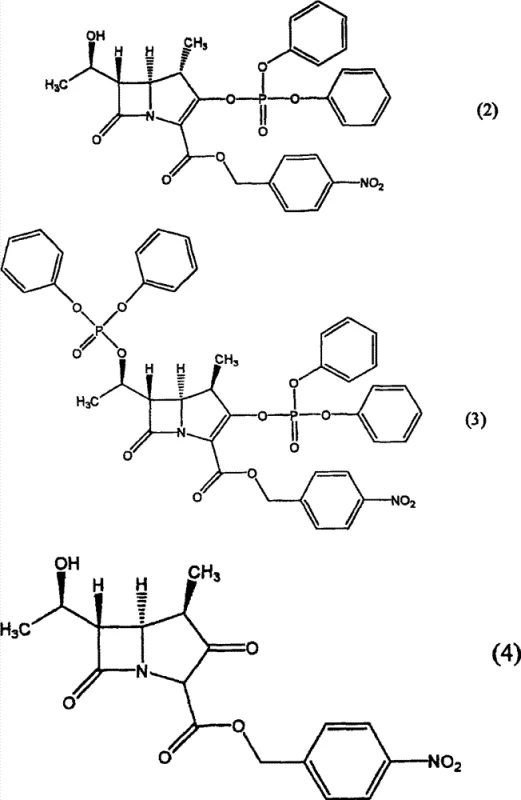
Historically, the isolation of azetidinone intermediates has been plagued by significant technical hurdles that compromise both product quality and manufacturing efficiency. Traditional crystallization techniques, often involving the simple addition of anti-solvents like n-hexane to dichloromethane solutions without precise seed control, frequently result in crystals with suboptimal physical properties. A primary concern is the entrapment of structurally related impurities, specifically those represented by general formulas (2), (3), and (4), which often persist at levels exceeding 0.4% in the final product. These impurities not only complicate downstream synthetic steps but also pose regulatory risks for the final antibiotic drug substance. Furthermore, conventional methods typically yield crystals with poor filterability due to small particle sizes and irregular morphology, leading to prolonged filtration times that bottleneck production lines. The instability of these conventionally produced crystals under accelerated storage conditions, such as elevated temperatures, further exacerbates supply chain risks by limiting shelf life and requiring stringent climate-controlled logistics.
The Novel Approach
The innovative methodology described in the patent data revolutionizes this landscape by introducing a seeded crystallization process that meticulously controls nucleation and crystal growth. Instead of relying on spontaneous nucleation, this approach involves adding a hydrocarbon solvent to a solution of the azetidinone compound in the presence of a specific quantity of seed crystals, ranging up to 200% by weight relative to the dissolved compound. This excess of seed material ensures that supersaturation is relieved primarily through the growth of existing crystals rather than the formation of new, imperfect nuclei. The result is a crystalline product with a significantly improved particle size distribution, where the proportion of larger crystals (greater than 350μm) is markedly increased. This structural optimization directly translates to enhanced filterability, allowing for rapid solid-liquid separation that dramatically improves manufacturing throughput. Moreover, the controlled growth environment effectively excludes impurity molecules from the crystal lattice, yielding a high-purity product with impurity levels consistently maintained below 0.3%, thereby setting a new standard for quality in carbapenem intermediate manufacturing.
Mechanistic Insights into Seed-Mediated Crystal Growth
The core mechanism driving the success of this crystallization technology lies in the thermodynamic and kinetic manipulation of the nucleation process. In a standard unseeded or lightly seeded crystallization, the system relies on primary nucleation, which is a stochastic process often leading to a wide distribution of crystal sizes and the occlusion of impurities due to rapid, uncontrolled growth. By introducing a substantial mass of seed crystals, the process shifts the energy barrier, favoring secondary nucleation and surface growth on the existing seeds. This phenomenon ensures that the solute molecules deposit onto the orderly lattice of the seeds, effectively rejecting impurity molecules that do not fit the crystal structure, such as the phosphorus-containing byproducts shown in the structural analysis. The use of hydrocarbon solvents like n-hexane or n-heptane acts as a precise anti-solvent trigger, reducing the solubility of the azetidinone compound just enough to drive deposition without causing shock precipitation. This delicate balance allows for the formation of dense, well-defined crystals with high bulk density, which is critical for efficient handling and packaging in large-scale operations.
Furthermore, the aging step incorporated into this process plays a pivotal role in Ostwald ripening, where smaller, less stable crystals dissolve and redeposit onto larger crystals, further narrowing the particle size distribution. This mechanism is essential for achieving the observed improvements in filtration rates, as larger particles create a more permeable filter cake with lower resistance to flow. The enhanced stability of the resulting crystals can be attributed to the reduced surface area-to-volume ratio and the lower internal strain within the crystal lattice, which minimizes sites for degradation initiation. For R&D teams, understanding this mechanism provides a roadmap for scaling the process, as parameters such as stirring intensity (preferably above 0.1 kW/m3) and temperature gradients become critical control points to maintain the desired growth kinetics. This level of mechanistic control transforms crystallization from a black-box unit operation into a precision engineering step that guarantees consistent product quality.
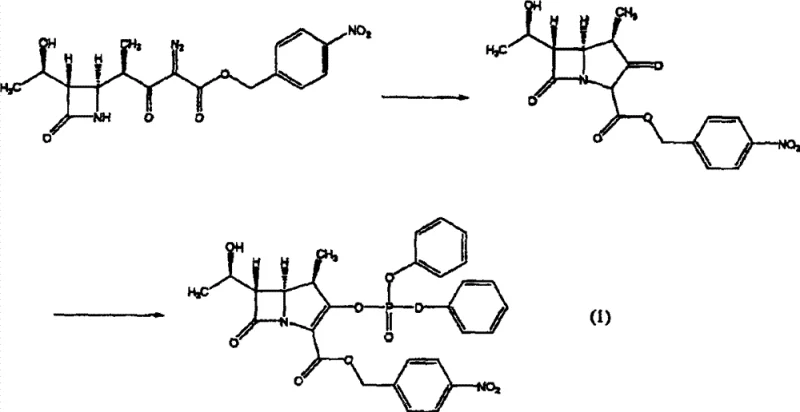
How to Synthesize Azetidinone Compound Efficiently
Implementing this advanced crystallization protocol requires a systematic approach to solvent selection and process parameter control to fully realize the benefits of improved purity and filterability. The process begins with the preparation of a 'good solvent' solution, where the azetidinone compound is dissolved in solvents such as dichloromethane, ethyl acetate, or ketones like 4-methyl-2-pentanone at concentrations typically exceeding 5 wt%. Once the solution is prepared, the critical step involves the introduction of seed crystals, which can be generated in situ or added externally, followed by the gradual addition of a hydrocarbon anti-solvent. The precise timing and rate of anti-solvent addition, coupled with controlled agitation, are vital to prevent localized supersaturation spikes that could trigger unwanted primary nucleation. Detailed standardized operating procedures for this synthesis route are essential for technology transfer and scale-up, ensuring that the laboratory-scale successes are replicated faithfully in commercial production environments.
- Prepare a good solvent solution of the azetidinone compound using solvents like dichloromethane or ethyl acetate, ensuring concentration is optimized for supersaturation.
- Introduce seed crystals into the solution, ensuring the amount is between 1% and 200% by weight relative to the dissolved compound to promote crystal growth over nucleation.
- Add a hydrocarbon anti-solvent such as n-hexane or n-heptane gradually while maintaining specific temperature controls to induce crystallization and improve filterability.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this crystallization technology offers profound strategic advantages that extend well beyond the laboratory bench. The primary value proposition lies in the drastic improvement of process efficiency, driven by the enhanced filterability of the resulting crystals. In traditional manufacturing, filtration can be a rate-limiting step, often requiring hours to complete and tying up valuable reactor and filtration equipment. By producing crystals with larger particle sizes and higher bulk density, this method significantly reduces filtration time, thereby increasing the overall equipment effectiveness (OEE) and allowing for more batches to be produced within the same timeframe. This efficiency gain translates directly into cost reduction in pharmaceutical intermediate manufacturing, as it lowers the utility and labor costs associated with extended processing cycles. Additionally, the reduced processing time minimizes the duration the product spends in solution, where it is most susceptible to degradation, further safeguarding yield and quality.
- Cost Reduction in Manufacturing: The elimination of prolonged filtration steps and the reduction in reprocessing requirements due to high initial purity lead to substantial operational expenditure savings. By avoiding the need for extensive recrystallization to meet impurity specifications, manufacturers can reduce solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable production model. The ability to consistently achieve high purity in a single crystallization step simplifies the workflow, reducing the complexity and cost of quality control testing and batch release procedures.
- Enhanced Supply Chain Reliability: The improved thermal stability of the crystals ensures that the intermediate can withstand the rigors of global transportation and long-term storage without significant degradation. This robustness reduces the risk of supply disruptions caused by product spoilage during transit or warehousing, providing a more reliable supply of high-purity pharmaceutical intermediates to downstream API manufacturers. The consistent quality and physical properties of the material also facilitate smoother integration into customer production lines, reducing the likelihood of manufacturing deviations or batch failures at the client site.
- Scalability and Environmental Compliance: The use of common hydrocarbon solvents and the avoidance of exotic reagents make this process highly scalable and compatible with existing industrial infrastructure. The improved bulk density of the crystals means that more product can be stored and shipped in the same volume, optimizing logistics and reducing the carbon footprint associated with transportation. Furthermore, the higher yield and purity reduce the generation of chemical waste, aligning with increasingly stringent environmental regulations and corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this patented crystallization technology. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this method for their specific production needs.
Q: How does the seed crystal method improve impurity profiles in carbapenem intermediates?
A: By utilizing a significant amount of seed crystals (up to 200% by weight), the process promotes the growth of existing nuclei rather than forming new ones, which effectively excludes structurally related impurities like formulas (2), (3), and (4), keeping total impurities below 0.3%.
Q: What are the stability advantages of this crystallization technique?
A: The resulting crystals exhibit superior thermal stability, retaining significantly higher active content after storage at elevated temperatures (e.g., 60°C) compared to conventional methods, which reduces degradation risks during long-term storage and transport.
Q: Why is particle size distribution critical for industrial filtration?
A: Larger crystal particles with a controlled size distribution prevent clogging of filtration equipment and allow for faster solid-liquid separation, drastically reducing processing time and improving overall manufacturing throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Azetidinone Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the successful development and manufacture of life-saving antibiotics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated crystallization techniques described in patent CN101432289B can be seamlessly integrated into your supply chain. We are committed to delivering materials that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our expertise in handling complex beta-lactam chemistries allows us to navigate the nuances of seed crystal preparation and solvent management, guaranteeing a consistent supply of reliable azetidinone compound materials for your critical projects.
We invite you to collaborate with us to explore how this advanced crystallization technology can optimize your production costs and enhance your product quality. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to innovation can drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
