Scalable Electrochemical Oxidation of Thioethers for High-Purity Sulfoxide Manufacturing
Scalable Electrochemical Oxidation of Thioethers for High-Purity Sulfoxide Manufacturing
The pharmaceutical and fine chemical industries are constantly seeking greener, more efficient pathways to synthesize critical intermediates, particularly sulfoxides which are ubiquitous in bioactive molecules. Patent CN113737206A introduces a groundbreaking electrochemical strategy for the selective oxidation of thioethers to sulfoxides, addressing long-standing challenges in sustainability and cost. This innovative approach utilizes a simple undivided cell setup with sodium chloride serving a dual role as both an electrolyte and a redox mediator, effectively replacing hazardous chemical oxidants. By leveraging electricity as the primary driving force, this method achieves high conversion rates under mild conditions, typically around 20°C, using a benign acetone and water solvent system. For R&D directors and procurement specialists, this technology represents a significant shift away from reliance on precious metal catalysts, offering a robust alternative for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for preparing sulfoxides often rely heavily on stoichiometric amounts of strong chemical oxidants or expensive transition metal catalysts, which pose significant environmental and economic burdens. For instance, prior art methods, such as those utilizing chloroauric acid (HAuCl4) combined with sodium hypochlorite (NaClO), involve complex operational steps and the handling of toxic substances. 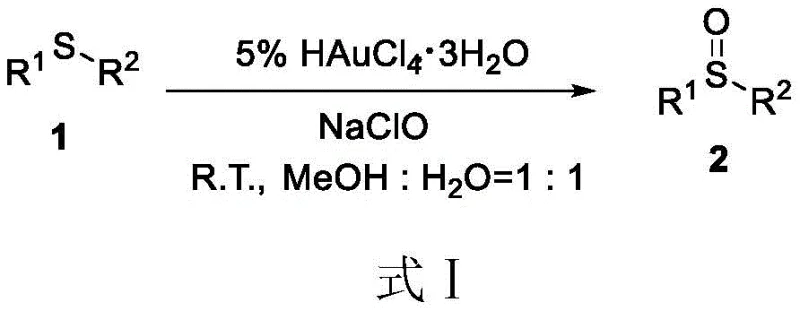 The use of heavy metals like gold not only inflates the raw material costs drastically but also necessitates rigorous downstream purification processes to ensure residual metal levels meet stringent pharmaceutical standards. Furthermore, the employment of strong oxidants like hypochlorite can lead to over-oxidation issues, where the desired sulfoxide is further oxidized to the corresponding sulfone, thereby reducing overall yield and complicating the isolation of the target molecule. These factors collectively hinder the efficiency and sustainability of conventional manufacturing protocols.
The use of heavy metals like gold not only inflates the raw material costs drastically but also necessitates rigorous downstream purification processes to ensure residual metal levels meet stringent pharmaceutical standards. Furthermore, the employment of strong oxidants like hypochlorite can lead to over-oxidation issues, where the desired sulfoxide is further oxidized to the corresponding sulfone, thereby reducing overall yield and complicating the isolation of the target molecule. These factors collectively hinder the efficiency and sustainability of conventional manufacturing protocols.
The Novel Approach
In stark contrast, the novel electrochemical methodology disclosed in the patent offers a streamlined, metal-free alternative that fundamentally simplifies the reaction engineering. As illustrated in the general reaction scheme, the process employs a constant current electrolysis in a mixture of acetone and water, utilizing inexpensive sodium chloride as the key additive. 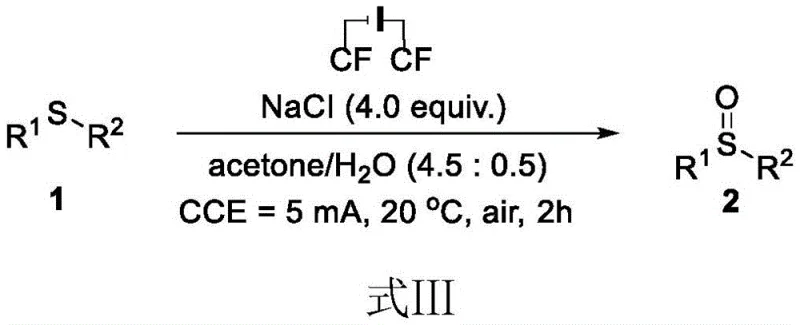 This approach eliminates the need for external chemical oxidants, as the active oxidizing species are generated in situ at the electrode surface. The reaction conditions are remarkably mild, proceeding efficiently at room temperature (20°C) with a low constant current of 5mA, which minimizes energy consumption and thermal hazards. By avoiding toxic reagents and precious metals, this method not only enhances the safety profile of the synthesis but also drastically simplifies the work-up procedure, allowing for easier product isolation via standard silica gel chromatography.
This approach eliminates the need for external chemical oxidants, as the active oxidizing species are generated in situ at the electrode surface. The reaction conditions are remarkably mild, proceeding efficiently at room temperature (20°C) with a low constant current of 5mA, which minimizes energy consumption and thermal hazards. By avoiding toxic reagents and precious metals, this method not only enhances the safety profile of the synthesis but also drastically simplifies the work-up procedure, allowing for easier product isolation via standard silica gel chromatography.
Mechanistic Insights into Electrochemical Oxidation Mediated by Chloride
The core of this technological advancement lies in the unique role of the chloride ion within the electrochemical cell, acting as a reversible redox mediator rather than a mere spectator ion. During the electrolysis process, chloride ions are oxidized at the anode to generate active chlorine species, which subsequently react with the thioether substrate to form the sulfoxide product while regenerating the chloride ion. This catalytic cycle ensures that the sodium chloride is not consumed stoichiometrically in a wasteful manner but facilitates the electron transfer necessary for oxidation. The use of graphite felt electrodes provides a high surface area for these reactions to occur, enhancing the efficiency of the electron transfer process. Furthermore, the specific solvent ratio of acetone to water (4.5:0.5) is critical; it ensures sufficient solubility for the organic substrate while maintaining the conductivity required for the electrochemical process, creating an optimized environment for selective oxidation.
From an impurity control perspective, the electrochemical method offers superior selectivity compared to traditional chemical oxidation. In conventional methods, the aggressive nature of chemical oxidants often leads to non-selective attacks on other functional groups or over-oxidation to sulfones. However, in this electrochemical system, the reaction potential can be finely tuned by adjusting the applied current. By maintaining a constant current of 5mA, the generation of the active oxidizing species is controlled, preventing the accumulation of excessive oxidant that would drive the reaction beyond the sulfoxide stage. This precise control results in high purity crude products, as evidenced by the excellent yields reported across various substrates, including those with sensitive functional groups like carboxylic acids and alkenes. Consequently, the burden on downstream purification is significantly reduced, leading to a cleaner final product profile.
How to Synthesize Sulfoxide Compounds Efficiently
The synthesis protocol described in the patent is designed for simplicity and reproducibility, making it highly attractive for laboratory optimization and subsequent pilot plant trials. The procedure involves charging a diaphragm-free electrolytic cell with the thioether substrate and four equivalents of sodium chloride, followed by the addition of the acetone-water solvent mixture. Graphite felt electrodes are immersed, and a constant current is applied for a duration of approximately two hours.
- Prepare the electrolytic cell by adding thioether compound and sodium chloride (1: 4 molar ratio) into a reactor.
- Add a mixed solvent of acetone and water (4.5: 0.5 ratio) and insert graphite felt electrodes as cathode and anode.
- Apply a constant current of 5mA at 20°C for 2 hours, then isolate the product via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this electrochemical technology translates into tangible strategic benefits regarding cost stability and supply security. The elimination of precious metal catalysts like gold removes a major source of price volatility and supply risk from the manufacturing equation. Since the process relies on commodity chemicals such as sodium chloride, acetone, and water, the raw material supply chain is inherently more robust and less susceptible to geopolitical disruptions or market fluctuations associated with rare earth or precious metal mining. This shift to abundant, low-cost reagents ensures a more predictable cost structure for long-term production contracts, allowing for better financial planning and margin protection in competitive markets.
- Cost Reduction in Manufacturing: The most immediate impact of this technology is the drastic reduction in direct material costs associated with catalysts and oxidants. By replacing expensive gold salts and hazardous hypochlorite solutions with common salt and electricity, the variable cost per kilogram of the product is significantly lowered. Additionally, the simplified work-up procedure, which avoids complex metal scavenging steps, reduces the consumption of auxiliary materials and labor hours required for purification. This leaner process flow contributes to substantial overall cost savings, making the manufacturing of sulfoxide intermediates more economically viable even at smaller scales.
- Enhanced Supply Chain Reliability: The reliance on universally available reagents enhances the resilience of the supply chain. Sodium chloride and acetone are produced in massive quantities globally, ensuring that production schedules are not held hostage by the availability of niche specialty chemicals. Furthermore, the mild reaction conditions (20°C, ambient pressure) reduce the stress on equipment and lower the risk of unplanned shutdowns due to safety incidents or equipment failure. This operational stability guarantees consistent delivery timelines, a critical factor for pharmaceutical clients who depend on just-in-time inventory strategies for their API synthesis.
- Scalability and Environmental Compliance: Electrochemical processes are inherently scalable through the numbering-up of cells or the use of larger flow reactors, facilitating a smooth transition from gram-scale R&D to ton-scale commercial production. From an environmental standpoint, the absence of heavy metal waste streams simplifies effluent treatment and helps manufacturers meet increasingly stringent environmental regulations. The use of water as a co-solvent further aligns with green chemistry principles, reducing the volume of organic waste generated. These factors collectively lower the barrier to entry for large-scale manufacturing and minimize the environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These insights are derived directly from the experimental data and beneficial effects outlined in the patent documentation, providing clarity on the practical application of the technology.
Q: What are the advantages of using electrochemistry for sulfoxide synthesis compared to traditional metal catalysis?
A: The electrochemical method eliminates the need for expensive and toxic transition metal catalysts like gold (HAuCl4). It uses electrons as clean reagents and common salt (NaCl) as a mediator, significantly reducing heavy metal contamination risks and purification costs.
Q: Can this electrochemical process be scaled for industrial production of API intermediates?
A: Yes, the process operates under mild conditions (20°C, ambient pressure) using simple undivided cells. The use of inexpensive reagents like NaCl and acetone/water solvents makes it highly suitable for large-scale commercial manufacturing without complex waste treatment.
Q: What is the selectivity of this method regarding over-oxidation to sulfones?
A: The method demonstrates high selectivity for sulfoxides. By precisely controlling the current (5mA) and reaction time, the oxidation stops primarily at the sulfoxide stage, minimizing the formation of sulfone by-products which are common in strong chemical oxidations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modern pharmaceutical manufacturing. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped to handle electrochemical reactions safely and efficiently, adhering to stringent purity specifications and operating within rigorous QC labs to guarantee the highest quality standards for every batch. We are committed to helping our partners leverage cutting-edge technologies like the one described in CN113737206A to optimize their supply chains.
We invite you to collaborate with us to explore how this metal-free electrochemical route can enhance your specific project requirements. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your target molecules, demonstrating exactly how switching to this green methodology can improve your bottom line. Please contact our technical procurement team today to request specific COA data for similar sulfoxide intermediates and to discuss detailed route feasibility assessments for your upcoming campaigns.
