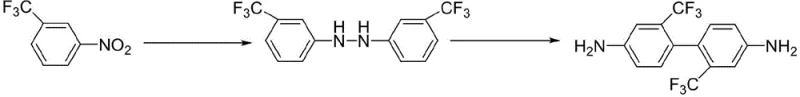
Scalable Production of 2,2'-Bis(trifluoromethyl)-4,4'-diaminobiphenyl via Hydrazine Reduction
The chemical industry is constantly seeking more efficient and safer pathways for producing high-performance fluorinated monomers, and the recent disclosure in patent CN113024385A presents a significant breakthrough in the synthesis of 2,2'-bis(trifluoromethyl)-4,4'-diaminobiphenyl (TFDB). This critical intermediate is indispensable for manufacturing soluble polyimides, which are prized for their exceptional thermal stability, mechanical strength, and optical transparency in advanced electronic applications. The patented method introduces a novel two-step sequence that replaces hazardous high-pressure hydrogenation with a mild hydrazine-based reduction, fundamentally altering the safety and economic profile of TFDB manufacturing. By utilizing m-nitrobenzotrifluoride as a starting material and employing a palladium-carbon catalytic system in a low-boiling alcohol solvent, the process achieves high conversion rates while maintaining a remarkably clean reaction profile. This technological advancement addresses long-standing challenges in the fluorine materials sector, offering a robust alternative to traditional nitration-coupling routes that often suffer from poor selectivity and difficult purification steps.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of fluorinated biphenyl diamines has been plagued by significant operational hurdles and environmental concerns associated with legacy synthesis routes. Traditional methods often rely on the coupling of halogenated nitro compounds using expensive transition metal catalysts or the reduction of dinitro precursors using zinc powder in alkaline conditions. The zinc powder reduction pathway, in particular, generates massive quantities of solid zinc oxide waste and highly alkaline wastewater, creating a substantial burden for waste treatment facilities and driving up disposal costs. Furthermore, alternative catalytic hydrogenation routes using molecular hydrogen require specialized high-pressure reactors and rigorous safety protocols to mitigate explosion risks, which drastically increases the capital expenditure required for plant construction. These conventional processes also frequently struggle with selectivity issues, leading to complex mixtures of azo, azoxy, and amine by-products that are difficult to separate, ultimately compromising the purity required for high-end polyimide applications.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the innovative process described in the patent utilizes hydrazine hydrate as a liquid reducing agent, effectively bypassing the need for gaseous hydrogen and high-pressure infrastructure. This method operates under atmospheric pressure in standard reaction vessels, significantly lowering the barrier to entry for manufacturers and enhancing overall process safety. The reaction proceeds through a selective reduction of the nitro group to a hydrazine intermediate, followed by an acid-catalyzed rearrangement that efficiently constructs the biphenyl backbone. A key feature of this approach is the use of a single low-boiling alcohol solvent, such as methanol or ethanol, which simplifies the workup procedure by eliminating the need for complex solvent exchanges or liquid-liquid separations between steps. The result is a streamlined, continuous workflow that not only improves yield and selectivity but also aligns perfectly with modern green chemistry principles by minimizing waste generation and energy consumption.
Mechanistic Insights into Pd/C-Catalyzed Hydrazine Reduction
The core of this synthetic strategy lies in the precise control of the catalytic reduction step, where palladium on carbon facilitates the transfer of hydrogen from hydrazine to the nitro substrate. Under the influence of an inorganic base like potassium hydroxide or sodium hydroxide, the hydrazine molecule is activated on the catalyst surface, allowing for the selective reduction of the nitro group to a hydrazino group without over-reduction to the amine at this stage. This selectivity is crucial, as it prevents the formation of m-trifluoromethylaniline impurities that typically plague zinc-based reductions. The reaction temperature is carefully maintained between 40°C and 76°C, a range that ensures sufficient kinetic energy for the transformation while preventing the decomposition of the sensitive hydrazine intermediate. The presence of the trifluoromethyl group exerts a strong electron-withdrawing effect, which influences the electronic density of the aromatic ring and necessitates the optimized catalytic conditions described to achieve the reported 95% to 97% selectivity.
Following the reduction, the process leverages a classic benzidine-type rearrangement driven by strong inorganic acids such as sulfuric or hydrochloric acid. This step involves the protonation of the hydrazine nitrogen atoms, triggering a [5,5]-sigmatropic rearrangement that forms the new carbon-carbon bond between the two aromatic rings. The reaction is conducted at low temperatures, ranging from -10°C to 20°C, to control the exothermic nature of the acid addition and to suppress side reactions that could lead to semidine by-products. The use of a polar alcohol solvent proves advantageous here, as it promotes the dissolution of the organic intermediate in the acidic medium, ensuring homogeneous reaction conditions. Upon completion, the product precipitates out of the aqueous phase upon neutralization, a physical property that allows for easy isolation via simple filtration, thereby avoiding the need for energy-intensive distillation or chromatographic purification steps.
How to Synthesize 2,2'-Bis(trifluoromethyl)-4,4'-diaminobiphenyl Efficiently
Implementing this synthesis requires careful attention to the stoichiometric ratios of hydrazine and base, as well as strict temperature control during the acid rearrangement phase. The patent outlines a straightforward protocol where the crude hydrazine solution from the first step can be telescoped directly into the second step without isolation, maximizing throughput and minimizing solvent usage. Operators must ensure the protective gas atmosphere, typically nitrogen, is maintained throughout to prevent oxidation of the sensitive hydrazine species. For detailed operational parameters including specific mixing rates and quenching procedures, please refer to the standardized guide below.
- Perform catalytic reduction of m-nitrobenzotrifluoride using hydrazine hydrate and Pd/C catalyst in alcohol solvent at 40-76°C to form 3,3'-bis(trifluoromethyl)diphenylhydrazine.
- Filter off the palladium carbon catalyst and proceed directly to the rearrangement step without solvent removal.
- Add the hydrazine solution to inorganic acid at -10 to 20°C for rearrangement, then neutralize and recrystallize to obtain high-purity TFDB.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift to this hydrazine-based methodology represents a strategic opportunity to optimize both operational expenditures and supply reliability. By eliminating the requirement for high-pressure hydrogenation reactors, manufacturers can utilize existing standard glass-lined or stainless-steel equipment, thereby avoiding the long lead times and high costs associated with procuring specialized pressure vessels. This flexibility allows for faster capacity expansion and reduces the risk of production bottlenecks caused by equipment maintenance or regulatory inspections specific to high-pressure systems. Furthermore, the ability to recycle the palladium catalyst multiple times without significant loss of activity translates into substantial long-term savings on precious metal consumption, a critical factor given the volatility of palladium prices in the global market.
- Cost Reduction in Manufacturing: The elimination of zinc powder removes the costly and logistically challenging disposal of heavy metal sludge, while the simplified solvent system reduces the energy load associated with solvent recovery and distillation. The process avoids the need for expensive phase transfer catalysts and co-catalysts often required in competing technologies, leading to a leaner bill of materials. Additionally, the high selectivity of the reaction minimizes the loss of raw materials to by-products, ensuring that a greater proportion of the input m-nitrobenzotrifluoride is converted into saleable final product. These factors combine to create a significantly lower cost of goods sold, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on readily available commodity chemicals like hydrazine hydrate and methanol ensures a stable supply of raw materials, reducing the risk of shortages associated with specialized reagents. The mild reaction conditions and robust nature of the catalyst system contribute to consistent batch-to-batch reproducibility, which is essential for maintaining long-term contracts with downstream polyimide producers. The simplified purification process, which relies on pH adjustment and crystallization rather than complex chromatography, shortens the overall cycle time per batch, allowing for increased production frequency and faster response to market demand fluctuations.
- Scalability and Environmental Compliance: The absence of heavy metal waste streams and the reduction in organic solvent complexity make this process inherently more compliant with increasingly stringent environmental regulations. Scaling up from pilot to commercial production is facilitated by the use of conventional agitation and heating systems, removing the engineering complexities of managing high-pressure gas-liquid interfaces. The green profile of the synthesis, characterized by atom economy and waste minimization, aligns with the sustainability goals of major multinational corporations, potentially qualifying the manufacturer for green supply chain incentives and preferential vendor status.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on process capabilities and product quality. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this method for large-scale production.
Q: Why is hydrazine hydrate preferred over hydrogen gas for this reduction?
A: Using hydrazine hydrate eliminates the need for high-pressure hydrogenation equipment, significantly reducing capital expenditure and safety risks associated with handling explosive hydrogen gas in industrial settings.
Q: How does this method control impurities compared to zinc powder reduction?
A: The Pd/C catalytic system offers high selectivity (95-97%) and avoids the generation of large amounts of zinc oxide solid waste and alkaline wastewater, resulting in a cleaner reaction profile with total impurities controlled within 3%.
Q: Is the palladium catalyst reusable in this process?
A: Yes, the patent data confirms that the palladium carbon catalyst can be recovered by filtration and successfully reused in subsequent batches without significant loss of catalytic activity, further optimizing production costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-Bis(trifluoromethyl)-4,4'-diaminobiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity fluorinated diamines play in the next generation of advanced polymer materials. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our state-of-the-art facilities are equipped to handle the specific requirements of fluorine chemistry, including corrosion-resistant reactor trains and rigorous QC labs capable of verifying stringent purity specifications down to the trace impurity level. We are committed to delivering not just a chemical product, but a reliable supply solution that supports your R&D and manufacturing timelines.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive value and efficiency in your supply chain.
