Advanced TFMB Synthesis: Scalable Polyimide Monomer Manufacturing for High-Performance Applications
The global demand for high-performance polyimides in aerospace and microelectronics sectors has intensified the search for efficient monomer synthesis routes. Patent CN112939796B introduces a groundbreaking synthetic method for 2,2'-bis(trifluoromethyl)-4,4'-diaminobiphenyl (TFMB), a critical monomer known for imparting exceptional thermal stability and chemical inertness to polymer matrices. This technology represents a significant leap forward in fine chemical engineering, addressing long-standing inefficiencies in traditional manufacturing protocols. By leveraging a strategic three-step sequence involving amidation protection, reductive coupling, and deamidation, the process achieves total yields ranging from 60% to 80%, substantially outperforming legacy methods. For R&D directors and procurement specialists, this patent offers a viable pathway to secure high-purity electronic chemical intermediates while mitigating the environmental burdens associated with heavy metal waste and harsh acidic conditions.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of TFMB has been plagued by severe technical and economic bottlenecks that hinder scalable manufacturing. The diphenyl hydrazine rearrangement method, for instance, relies heavily on large quantities of inorganic acids, generating massive volumes of waste acid that are difficult and costly to treat due to complex byproduct mixtures. Furthermore, this route suffers from poor atom economy, with total yields stagnating around 50%, rendering it economically unviable for large-scale operations. Alternatively, the Ullmann coupling route, while chemically direct, necessitates extreme reaction temperatures and excessive catalyst consumption, leading to significant metal contamination and environmental pollution. These conventional pathways not only inflate production costs through waste management and energy consumption but also fail to meet the stringent purity requirements demanded by advanced optoelectronic applications, often resulting in yields below 40% that cannot support consistent supply chains.
The Novel Approach
In stark contrast, the methodology disclosed in CN112939796B revolutionizes TFMB production by implementing a mild, catalytic reductive coupling strategy protected by an amide group. This innovative approach utilizes 2-chloro-5-amino benzotrifluoride, a commercially abundant and low-cost precursor, effectively bypassing the need for expensive or hard-to-source starting materials like 4-nitro-3-trifluoromethylaniline. The reaction conditions are remarkably gentle, operating at moderate temperatures between 60°C and 70°C, which drastically reduces energy expenditure compared to the high-heat requirements of Ullmann coupling. By employing a palladium-carbon catalyst in conjunction with safe reducing agents such as hydroxylamine hydrochloride, the process minimizes hazardous waste generation and simplifies downstream purification. This streamlined synthetic logic not only enhances the overall yield to an impressive 60-80% range but also ensures the production of TFMB with purity levels exceeding 99.7%, making it ideally suited for reliable polyimide monomer supplier networks seeking consistency and quality.
Mechanistic Insights into Pd/C-Catalyzed Reductive Coupling
The core of this synthetic breakthrough lies in the sophisticated use of temporary amide protection to facilitate selective carbon-carbon bond formation. Initially, the amino group of the starting material is protected via amidation with a cyclic anhydride, such as succinic anhydride, forming a stable intermediate that prevents unwanted side reactions during the subsequent coupling phase. This protection strategy is crucial for maintaining the integrity of the aromatic system and ensuring that the reductive coupling occurs specifically at the chloro-substituted positions. The presence of the electron-withdrawing trifluoromethyl groups further activates the aromatic ring towards nucleophilic attack, yet the amide group balances this reactivity to prevent over-reduction or decomposition. Following the coupling event, the robust nature of the amide linkage allows the molecule to withstand the rigorous conditions of the palladium-catalyzed reduction without degradation, ensuring high selectivity for the biphenyl backbone formation.
Subsequent deamidation under basic reflux conditions cleanly removes the protecting group to reveal the free amine functionalities essential for polyimide polymerization. This final step is highly efficient, driven by the thermodynamic stability of the resulting aromatic diamine and the solubility differences that facilitate easy isolation of the product. The entire mechanistic pathway is designed to minimize impurity profiles, particularly avoiding the formation of azo or hydrazo byproducts common in older reduction methods. By controlling the stoichiometry of the reducing agent and the base, the process effectively suppresses the formation of mono-coupled species or over-reduced cyclohexyl derivatives. This precise control over the reaction trajectory is what enables the achievement of high-purity TFMB, satisfying the rigorous specifications required for high-purity OLED material and semiconductor applications where trace impurities can compromise device performance.
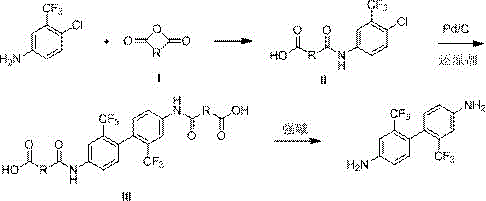
How to Synthesize 2,2'-bis(trifluoromethyl)-4,4'-diaminobiphenyl Efficiently
Implementing this patented synthesis requires careful attention to reagent stoichiometry and temperature control to maximize the benefits of the novel catalytic system. The process begins with the preparation of the amide intermediate in a solvent like dichloromethane or toluene at room temperature, ensuring complete conversion before proceeding to the critical coupling stage. Operators must maintain the reaction temperature between 60°C and 70°C during the reductive coupling phase to optimize catalyst activity while preventing thermal degradation of the sensitive intermediates. The detailed standardized synthesis steps, including specific workup procedures and purification protocols necessary to achieve the reported 99.7% purity, are outlined in the technical guide below for immediate operational reference.
- Perform amidation reaction between 2-chloro-5-amino benzotrifluoride and cyclic anhydride (e.g., succinic anhydride) in an organic solvent at room temperature to form Intermediate II.
- Conduct reductive coupling of Intermediate II using a Pd/C catalyst, a reducing agent like hydroxylamine hydrochloride, and a strong base at 60-70°C to obtain Intermediate III.
- Execute deamidation reaction on Intermediate III under reflux conditions with a strong base to yield the final 2,2'-bis(trifluoromethyl)-4,4'-diaminobiphenyl product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers transformative benefits that directly impact the bottom line and operational resilience. The shift away from complex, multi-step sequences involving hazardous reagents to a streamlined three-step process significantly simplifies the manufacturing workflow, thereby reducing the burden on quality control and logistics teams. By utilizing 2-chloro-5-amino benzotrifluoride, a commodity chemical with a stable global supply, manufacturers can decouple their production from the volatility associated with specialized, high-cost precursors. This strategic sourcing advantage ensures a more predictable raw material inventory and mitigates the risk of supply disruptions that often plague the fine chemical sector. Furthermore, the elimination of heavy metal pollutants and corrosive waste acids aligns perfectly with increasingly stringent environmental regulations, reducing compliance costs and enhancing the sustainability profile of the final product.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the substantial reduction in raw material costs and waste treatment expenses. By replacing expensive starting materials with low-cost, readily available intermediates, the overall cost of goods sold is significantly lowered without compromising product quality. The mild reaction conditions also translate to lower energy consumption, as there is no need for extreme heating or cryogenic cooling, further contributing to operational savings. Additionally, the high total yield of 60-80% means less raw material is wasted per unit of product, maximizing resource efficiency and driving down the effective cost per kilogram of TFMB produced.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method enhances supply chain continuity by reducing dependency on fragile, multi-vendor precursor chains. Since the key starting material is a common pesticide and medical intermediate, it is produced at scale by multiple suppliers globally, ensuring a buffer against regional shortages. The simplified process flow also shortens the production cycle time, allowing for faster turnaround on orders and improved responsiveness to market demand fluctuations. This agility is critical for maintaining just-in-time inventory levels and meeting the tight delivery schedules required by downstream polymer manufacturers in the electronics and aerospace industries.
- Scalability and Environmental Compliance: From a scalability perspective, the use of heterogeneous Pd/C catalysts and common organic solvents facilitates easy transition from laboratory bench scale to industrial tonnage production. The process avoids the use of exotic reagents that are difficult to source in bulk, ensuring that scale-up does not introduce new supply bottlenecks. Moreover, the environmentally friendly nature of the reaction, characterized by the absence of heavy metal waste and reduced acid usage, simplifies the permitting process for new manufacturing facilities. This compliance advantage accelerates time-to-market for new production lines and future-proofs the supply chain against tightening global environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this TFMB synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into existing manufacturing portfolios or for procurement teams assessing supplier qualifications.
Q: What are the primary advantages of this TFMB synthesis method over traditional Ullmann coupling?
A: Unlike traditional Ullmann coupling which requires high temperatures and generates significant metal pollution with yields below 40%, this novel method utilizes mild reaction conditions (60-70°C) and achieves a total yield of 60-80% with superior environmental compatibility.
Q: What starting materials are used in this patented process?
A: The process utilizes 2-chloro-5-amino benzotrifluoride as the primary starting material, which is a low-cost and readily available pesticide and medical intermediate, reacting with cyclic anhydrides such as succinic anhydride.
Q: What purity levels can be achieved with this synthetic route?
A: Experimental data from the patent indicates that the final TFMB product can achieve purity levels exceeding 99.7% (HPLC), making it highly suitable for demanding electronic and aerospace polyimide applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,2'-bis(trifluoromethyl)-4,4'-diaminobiphenyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality monomers play in the performance of next-generation polyimides and electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from patent concept to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of TFMB performs consistently in your high-value applications. Our expertise in catalytic hydrogenation and fine organic synthesis allows us to optimize this patented route for maximum yield and minimal environmental impact.
We invite you to collaborate with us to leverage this advanced technology for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized synthesis of 2,2'-bis(trifluoromethyl)-4,4'-diaminobiphenyl can enhance your supply chain efficiency and product quality.
