Revolutionizing Azo Dye Intermediates: Continuous Synthesis of 2-Amino-4-Acetamino Anisole
The chemical manufacturing landscape for azo dye intermediates is undergoing a significant transformation driven by the urgent need for greener, more efficient production methodologies. Patent CN114436876B introduces a groundbreaking continuous synthesis method for 2-amino-4-acetamino anisole, a critical coupling component widely used in the production of disperse dark blue HGL and other high-performance textile dyes. This technology represents a paradigm shift from traditional batch processing to a streamlined, continuous flow architecture that leverages ether-based solvent systems and robust nickel catalysis. For R&D directors and process engineers, this patent offers a compelling solution to long-standing challenges regarding product purity, impurity profiles, and operational continuity. By replacing hazardous or inefficient solvents like DMF and methanol with safer ether alternatives, the process not only enhances safety but also drastically improves the solubility of key intermediates, facilitating a seamless transition between reaction stages without the need for intermediate isolation. This technical advancement positions the method as a highly viable candidate for large-scale industrial adoption, promising substantial improvements in both economic efficiency and environmental compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 2-amino-4-acetamino anisole has relied on cumbersome multi-step pathways that are increasingly untenable in modern regulatory and economic environments. The traditional chlorobenzene method, depicted in the reaction scheme below, involves a six-step sequence including nitration, methoxylation, and multiple reduction and acylation steps, resulting in low overall yields and significant environmental burdens.
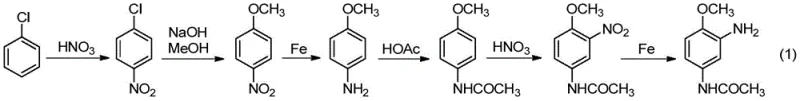
Furthermore, even the improved dinitrochlorobenzene method, which shortens the route to three steps, suffers from critical discontinuities. In conventional methanol-based systems, the intermediate 2,4-dinitroanisole exhibits poor solubility, necessitating filtration, washing, and drying before the subsequent hydrogenation step. This interruption not only increases equipment costs and labor intensity but also generates large volumes of saline wastewater. Additionally, the reliance on expensive precious metal catalysts like Pd/C or Pt/C inflates production costs, while the use of DMF as a solvent in some variations leads to the formation of difficult-to-remove by-products like 2,4-dinitro-N,N-dimethylaniline, compromising the final product quality and yield.
The Novel Approach
The innovative methodology disclosed in CN114436876B overcomes these structural inefficiencies by implementing a truly continuous three-step process anchored by a specialized ether solvent system. As illustrated in the reaction scheme below, the process flows directly from methoxylation to hydrogenation and finally to selective acetylation without isolating the unstable intermediates.
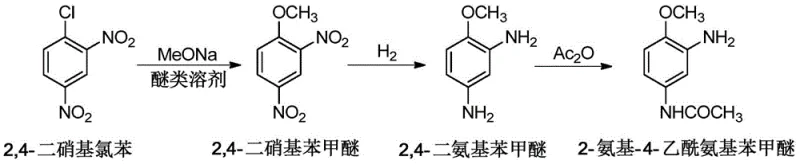
By utilizing ether solvents such as tetrahydrofuran or ethylene glycol dimethyl ether, the process ensures excellent solubility for 2,4-dinitroanisole, allowing the reaction mixture to be transferred directly to the hydrogenation reactor after simple filtration of salt by-products. This eliminates the drying and handling steps that typically bottleneck production. Moreover, the substitution of precious metals with cost-effective nickel-based catalysts or Pt-Sn bimetallic systems significantly reduces raw material expenses while maintaining high catalytic activity and selectivity. The entire operation is conducted under an inert atmosphere, effectively mitigating the risk of oxidative degradation of the sensitive diamino intermediate, thereby ensuring consistent high purity and color quality in the final dye intermediate.
Mechanistic Insights into Ether-Solvent Mediated Continuous Synthesis
The core mechanistic advantage of this technology lies in the synergistic interaction between the ether solvent matrix and the specific catalytic conditions employed during the hydrogenation phase. In the initial methoxylation step, the use of sodium methoxide in an ether medium facilitates a clean nucleophilic aromatic substitution, converting 2,4-dinitrochlorobenzene to 2,4-dinitroanisole with high selectivity. Crucially, the residual alkalinity from the methoxylation reagent carries over into the hydrogenation stage, naturally maintaining the reaction system pH between 8 and 10. This intrinsic pH buffering capability is vital, as it suppresses the coking and polymerization of the reactive 2,4-diaminoanisole intermediate on the catalyst surface, a common failure mode in acidic or neutral hydrogenation environments. The nickel-based catalysts, whether boride, Raney nickel, or modified ternary systems, operate efficiently under mild temperatures of 50-65°C and moderate hydrogen pressures, ensuring complete conversion while minimizing side reactions.
Impurity control is rigorously managed through precise thermal regulation during the final selective acetylation step. The process dictates a controlled addition of acetic anhydride at sub-zero temperatures, specifically between -15°C and -5°C, followed by a reaction phase at -10°C to 0°C. This tight thermal window is essential for achieving mono-acetylation selectivity, preventing the formation of the di-acetylated by-product which is difficult to separate and detrimental to dyeing performance. The absence of additional acid-binding agents in the preferred embodiment further simplifies the downstream purification process, as the reaction generates minimal solid waste. The final product is isolated via distillation and washing, yielding 2-amino-4-acetamino anisole with purity levels consistently exceeding 98%, suitable for direct application in high-end textile dyeing formulations without further recrystallization.
How to Synthesize 2-Amino-4-Acetamino Anisole Efficiently
The synthesis protocol outlined in the patent provides a robust framework for scaling this continuous process from laboratory benchtop to commercial production vessels. The procedure begins with the preparation of the methoxylation mixture under inert gas, followed by the direct transfer of the filtrate to a hydrogenation unit equipped with the specified nickel catalyst. Careful attention must be paid to the maintenance of the inert atmosphere throughout the transfer and reaction phases to prevent oxidation. The final acetylation requires precise temperature control equipment capable of sustaining sub-zero conditions to ensure the desired selectivity. For detailed operational parameters, stoichiometry, and safety guidelines, please refer to the standardized synthesis steps provided below.
- Perform methoxylation of 2,4-dinitrochlorobenzene using sodium methoxide in an ether solvent at 30-60°C to form 2,4-dinitroanisole.
- Conduct catalytic hydrogenation of the crude filtrate using a nickel-based catalyst at 50-65°C and 0.5-2.5 MPa pressure to yield 2,4-diaminoanisole.
- Execute selective acetylation by adding acetic anhydride at -15 to -5°C, followed by reaction at -10 to 0°C to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this continuous synthesis technology translates into tangible strategic benefits that extend beyond simple unit cost reductions. The elimination of intermediate isolation and drying steps fundamentally compresses the production cycle time, allowing for faster throughput and reduced work-in-progress inventory. This agility enables manufacturers to respond more rapidly to fluctuating market demands for azo dye intermediates, enhancing overall supply chain resilience. Furthermore, the shift from batch to continuous processing optimizes equipment utilization rates, meaning that existing infrastructure can potentially produce higher volumes without the need for significant capital expenditure on additional reactors or drying ovens.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the replacement of expensive precious metal catalysts with abundant nickel-based alternatives, which drastically lowers the recurring cost of catalytic materials. Additionally, the use of low-boiling ether solvents facilitates energy-efficient recovery and recycling through distillation, reducing both solvent purchase costs and waste disposal fees. The simplified workflow also reduces labor hours associated with manual filtering and transferring of solids, contributing to lower operational expenditures across the manufacturing value chain.
- Enhanced Supply Chain Reliability: By removing the dependency on complex multi-step batch operations that are prone to delays at isolation stages, the continuous nature of this synthesis ensures a more predictable and steady output of high-purity intermediates. The robustness of the nickel catalyst system against poisoning and the stability of the ether solvent matrix minimize the risk of batch failures or off-spec production runs. This reliability is critical for downstream dye manufacturers who require consistent quality and uninterrupted supply to maintain their own production schedules for polyester and blended fabric dyeing.
- Scalability and Environmental Compliance: The process design inherently supports scalability, as the continuous flow dynamics can be adapted to larger reactor volumes with minimal re-optimization. From an environmental perspective, the significant reduction in saline wastewater and solid waste generation aligns with increasingly stringent global environmental regulations. The avoidance of high-boiling, toxic solvents like DMF further simplifies waste treatment protocols, reducing the environmental footprint and potential liability associated with hazardous waste management, thereby securing long-term operational licenses.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on process feasibility and product quality assurance. Understanding these nuances is essential for technical teams evaluating the integration of this method into existing production lines.
Q: Why is the ether solvent system superior to traditional methanol in this synthesis?
A: Ether solvents provide superior solubility for the intermediate 2,4-dinitroanisole, eliminating the need for intermediate filtration and washing steps required in methanol systems, thus enabling a true continuous process.
Q: How does the new process reduce catalyst costs compared to conventional methods?
A: The process utilizes cost-effective nickel-based catalysts or Pt-Sn bimetallic systems instead of expensive palladium or platinum catalysts, significantly lowering the operational expenditure for the hydrogenation step.
Q: What measures are taken to prevent the oxidation of the diamino intermediate?
A: The entire reaction sequence is conducted under a strict inert gas environment, such as nitrogen or argon, which effectively prevents the air oxidation and blackening of the sensitive 2,4-diaminoanisole intermediate.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4-Acetamino Anisole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of continuous flow chemistry in the production of high-value dye intermediates like 2-amino-4-acetamino anisole. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN114436876B are fully realized in practical manufacturing settings. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced process control systems capable of maintaining the stringent purity specifications required by the global textile industry, guaranteeing product consistency and performance.
We invite forward-thinking partners to collaborate with us to leverage this advanced synthesis technology for their supply chains. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this continuous process can optimize your total cost of ownership. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us help you secure a reliable, cost-effective, and sustainable supply of this critical azo dye intermediate.
