Advanced Synthesis of 2-Amino-4-Acetamino Anisole for High-Performance Dye Manufacturing
Advanced Synthesis of 2-Amino-4-Acetamino Anisole for High-Performance Dye Manufacturing
The global demand for high-performance disperse dyes, particularly for polyester fiber applications, necessitates a robust and reliable supply of key intermediates such as 2-amino-4-acetamino anisole. Patent CN107903182B introduces a transformative synthetic methodology that addresses long-standing challenges in purity and environmental impact associated with traditional manufacturing processes. This innovative approach leverages a strategic sequence of etherification, catalytic hydrogenation, acetylation, and directed nitration to produce 2-amino-4-acetamino anisole with exceptional purity levels ranging from 99.5% to 99.8%. For R&D directors and procurement specialists in the fine chemical sector, this patent represents a significant leap forward, offering a pathway to reduce impurity profiles while enhancing the overall sustainability of the supply chain for critical dye intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-amino-4-acetamino anisole has relied heavily on routes starting from 2,4-dinitrochlorobenzene or 2,4-diaminoanisole, which present substantial technical and environmental hurdles. Traditional methods often employ stoichiometric reducing agents such as iron powder, sodium sulfide, or hydrazine hydrate, which generate massive quantities of hazardous solid waste and wastewater, complicating disposal and increasing regulatory compliance costs. Furthermore, when starting from 2,4-diaminoanisole, the presence of two reactive amino groups creates significant selectivity issues during the acylation step; the amino groups are prone to oxidation, leading to complex byproduct mixtures and the formation of unwanted double-acylated species. These side reactions not only lower the overall yield but also make the purification process arduous, often resulting in final products with purity levels that struggle to consistently exceed 98%, which is insufficient for high-end textile applications requiring precise color consistency.
The Novel Approach
In stark contrast, the methodology disclosed in CN107903182B adopts a clever "acylation-first, then nitration, then reduction" strategy that fundamentally alters the reaction landscape to favor high selectivity and purity. By initially converting p-nitroanisole to p-anisidine and immediately protecting the amino group via acetylation, the process effectively masks the nucleophilic site before introducing the nitro group. This protective acetyl group directs the subsequent nitration specifically to the ortho-position relative to the acetamido group, preventing random substitution and minimizing side reactions. 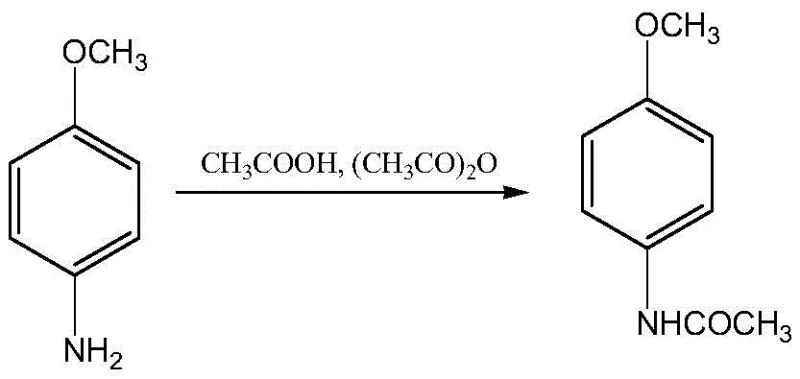
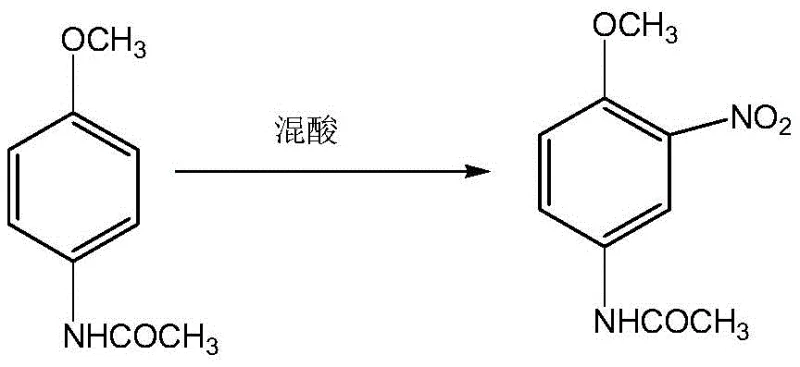 This sequence ensures that the sensitive amino functionality is never exposed to harsh oxidative conditions until the very final step, thereby virtually eliminating the formation of oxidation byproducts and double-acylation impurities. The result is a streamlined process that delivers a crystalline product with superior quality metrics, directly addressing the pain points of difficult separation and low purity inherent in older technologies.
This sequence ensures that the sensitive amino functionality is never exposed to harsh oxidative conditions until the very final step, thereby virtually eliminating the formation of oxidation byproducts and double-acylation impurities. The result is a streamlined process that delivers a crystalline product with superior quality metrics, directly addressing the pain points of difficult separation and low purity inherent in older technologies.
Mechanistic Insights into Multi-Element Catalytic Hydrogenation
A cornerstone of this advanced synthesis is the utilization of sophisticated multi-element heterogeneous catalysts that drive the reduction steps with high efficiency and minimal environmental footprint. The first reduction stage, converting p-nitroanisole to p-anisidine, employs a quaternary catalyst system comprising Nickel, Molybdenum, Cobalt, and Lithium (or Iron) supported on modified alumina. This specific metallic combination creates a synergistic electronic environment on the catalyst surface that facilitates the rapid and selective transfer of hydrogen to the nitro group while leaving the methoxy ether linkage intact. 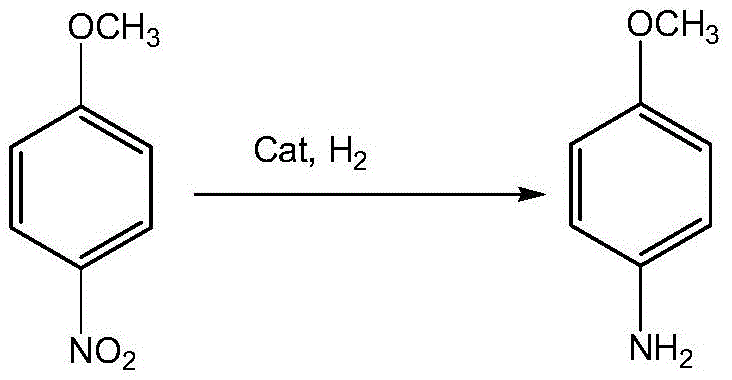 The use of such a robust catalyst allows the reaction to proceed under moderate temperatures and pressures, significantly reducing energy consumption compared to high-severity thermal processes. Moreover, because the catalyst is heterogeneous, it can be easily separated from the reaction mixture via filtration, allowing for multiple reuse cycles which drastically lowers the cost of goods sold (COGS) over time.
The use of such a robust catalyst allows the reaction to proceed under moderate temperatures and pressures, significantly reducing energy consumption compared to high-severity thermal processes. Moreover, because the catalyst is heterogeneous, it can be easily separated from the reaction mixture via filtration, allowing for multiple reuse cycles which drastically lowers the cost of goods sold (COGS) over time.
The final reduction step, transforming 2-nitro-4-acetamino anisole into the target 2-amino-4-acetamino anisole, utilizes a ternary catalyst system of Nickel, Molybdenum, and Boron (or Cobalt and Phosphorus). This catalyst is specifically tuned to reduce the aromatic nitro group in the presence of the acetamido moiety without causing hydrolysis of the amide bond, a common side reaction in acidic or overly aggressive reduction environments. 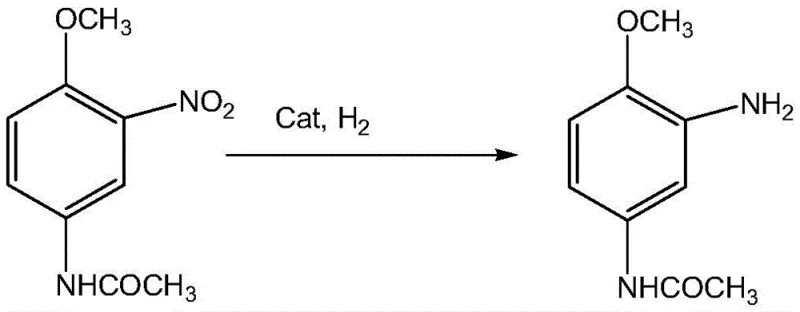 The mechanistic precision of this catalyst ensures that the final product retains its structural integrity, contributing to the reported purity of over 99%. From an impurity control perspective, the combination of these catalytic systems with the protective group strategy ensures that the impurity spectrum is exceptionally clean, simplifying downstream processing and quality control assays for pharmaceutical and dye manufacturers alike.
The mechanistic precision of this catalyst ensures that the final product retains its structural integrity, contributing to the reported purity of over 99%. From an impurity control perspective, the combination of these catalytic systems with the protective group strategy ensures that the impurity spectrum is exceptionally clean, simplifying downstream processing and quality control assays for pharmaceutical and dye manufacturers alike.
How to Synthesize 2-Amino-4-Acetamino Anisole Efficiently
The synthesis protocol outlined in the patent provides a detailed, scalable roadmap for producing this valuable intermediate, beginning with the etherification of p-nitrochlorobenzene and proceeding through the critical catalytic and functionalization steps described above. The process is designed to maximize atom economy and solvent recovery, with specific attention paid to temperature control and molar ratios to ensure optimal conversion at each stage. For technical teams looking to implement this route, the following guide summarizes the critical operational parameters derived from the patent examples, ensuring reproducibility and high yield in a pilot or commercial setting.
- Perform etherification of p-nitrochlorobenzene with methanol under alkaline conditions to obtain high-purity p-nitroanisole.
- Execute primary catalytic hydrogenation using a quaternary Ni-Mo-Co-Li catalyst to reduce p-nitroanisole to p-anisidine.
- Conduct acetylation followed by controlled nitration to form 2-nitro-4-acetamino anisole, ensuring regioselectivity.
- Complete the synthesis via secondary catalytic hydrogenation using a ternary Ni-Mo-B catalyst to yield the final product with >99.5% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling economic and logistical benefits that extend far beyond simple yield improvements. The shift from stoichiometric reducing agents to catalytic hydrogenation fundamentally changes the cost structure of the manufacturing process by eliminating the purchase and disposal costs associated with tons of iron sludge or sulfide waste. Additionally, the ability to recover and recycle key solvents like methanol and acetic acid, as well as the expensive multi-element catalysts, creates a closed-loop system that significantly insulates the production cost from volatile raw material price fluctuations. This stability is crucial for long-term supply contracts where price predictability is a key negotiation lever.
- Cost Reduction in Manufacturing: The implementation of recoverable heterogeneous catalysts eliminates the need for costly noble metals or disposable stoichiometric reagents, leading to substantial operational expenditure savings. By enabling the recycling of both the catalyst and the solvent systems, the process minimizes raw material consumption per kilogram of finished product, effectively driving down the variable cost of production without compromising on quality standards.
- Enhanced Supply Chain Reliability: The reliance on readily available bulk chemicals such as p-nitrochlorobenzene, methanol, and acetic anhydride ensures that the supply chain is not dependent on niche or scarce reagents that could cause bottlenecks. The robustness of the catalytic steps, which tolerate standard industrial conditions, reduces the risk of batch failures and unplanned downtime, thereby guaranteeing a consistent and reliable flow of material to downstream dye manufacturers.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, utilizing standard unit operations like hydrogenation reactors and distillation columns that are common in fine chemical plants. By replacing polluting iron powder reduction with clean catalytic hydrogenation, the method drastically reduces the environmental burden, simplifying the permitting process and ensuring compliance with increasingly stringent global environmental regulations regarding heavy metal discharge and solid waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of 2-amino-4-acetamino anisole, based on the detailed specifications provided in the patent literature. Understanding these nuances is essential for stakeholders evaluating the feasibility of integrating this intermediate into their existing product portfolios or supply networks.
Q: How does this synthesis method improve product purity compared to traditional routes?
A: By employing an 'acylation-first' strategy prior to nitration, this method prevents the oxidation of amino groups and avoids the formation of difficult-to-separate double-acylation byproducts common in traditional diaminoanisole routes, achieving purities of 99.5-99.8%.
Q: What specific catalysts are utilized to ensure environmental compliance?
A: The process utilizes recoverable heterogeneous multi-element catalysts, specifically a quaternary Ni-Mo-Co-Li system for the initial reduction and a ternary Ni-Mo-B system for the final reduction, eliminating the need for polluting iron powder or sodium sulfide reagents.
Q: Is this manufacturing process scalable for industrial production?
A: Yes, the protocol is designed for large-scale implementation, featuring solvent recovery systems for methanol and acetic acid, along with catalyst recycling capabilities that significantly reduce operational costs and waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4-Acetamino Anisole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-purity intermediates play in the success of the global dye and fine chemical industries. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications. Our rigorous QC labs are equipped to verify the absence of critical impurities, such as double-acylation byproducts or oxidation derivatives, guaranteeing that every batch of 2-amino-4-acetamino anisole performs consistently in your final applications.
We invite you to collaborate with us to leverage this advanced synthesis technology for your specific business needs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing capabilities can enhance your supply chain efficiency and product quality.
