Advanced Copper-Catalyzed Synthesis of 4-Acetoxy-2-Methyl-2-Butenal for Commercial Vitamin A Production
The pharmaceutical and fine chemical industries are constantly seeking robust, scalable pathways for critical Vitamin A intermediates, specifically 4-acetoxy-2-methyl-2-butenal. Patent CN108997117B introduces a transformative synthetic methodology that leverages tiglic aldehyde as a cost-effective starting material, diverging significantly from traditional, hazard-prone routes. This innovation addresses long-standing challenges in impurity control and environmental compliance, offering a streamlined three-step sequence that achieves a total yield exceeding 82 percent. For R&D Directors and Procurement Managers, this patent represents a pivotal shift towards sustainable manufacturing, replacing unstable intermediates and heavy metal catalysts with a precise copper-catalyzed oxidation system. The technical breakthrough lies not only in the yield but in the operational safety and the drastic reduction of three wastes, positioning this method as a superior choice for reliable Vitamin A intermediate supplier partnerships aiming for long-term supply chain stability.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-acetoxy-2-methyl-2-butenal has been plagued by significant technical and environmental hurdles that compromise commercial viability. Traditional routes, such as those utilizing isoprene and sodium hypochlorite, generate substantial volumes of saline wastewater, creating severe disposal challenges and increasing operational costs for environmental compliance. Other methods relying on ethylene oxide involve highly unstable intermediates that pose safety risks during storage and handling, while dimethoxyacetone routes suffer from lengthy reaction sequences and low overall yields, often ranging between 35 to 45 percent. These inefficiencies result in higher raw material consumption and complex purification processes that erode profit margins. Furthermore, the reliance on noble metal catalysts in some legacy processes introduces supply chain vulnerabilities due to the fluctuating cost and availability of metals like rhodium or palladium. The visual complexity of these older pathways is evident in prior art diagrams, which often depict multi-step sequences with poor atom economy.
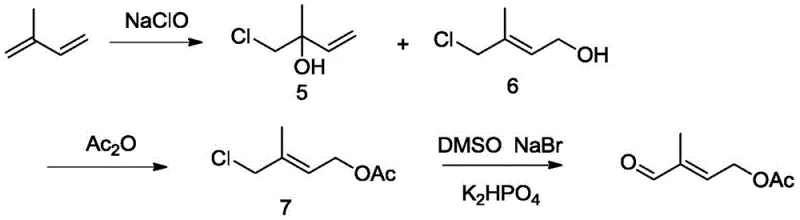
The Novel Approach
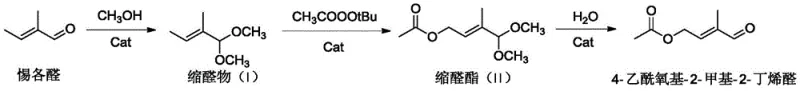
In stark contrast, the novel methodology disclosed in the patent utilizes a concise three-step strategy starting from tiglic aldehyde, a readily available and inexpensive feedstock. This approach bypasses the need for hazardous chlorination reagents and unstable epoxide intermediates, fundamentally altering the safety profile of the manufacturing process. The core innovation involves a highly selective allylic oxidation step catalyzed by a copper salt and nitrogen ligand system, which operates under mild conditions to preserve the integrity of the molecular structure. By protecting the aldehyde group as an acetal prior to oxidation, the process minimizes side reactions and ensures high regioselectivity, leading to a total yield that significantly outperforms conventional methods. This streamlined workflow reduces the number of unit operations required, thereby lowering energy consumption and simplifying equipment requirements for commercial scale-up of complex pharmaceutical intermediates. The result is a cleaner, more efficient process that aligns with modern green chemistry principles while delivering superior economic value.
Mechanistic Insights into Copper-Catalyzed Allylic Oxidation
The heart of this synthetic advancement lies in the second step, where a specialized copper catalyst system facilitates the allylic oxidation of the acetal intermediate. Unlike non-selective oxidants that may attack multiple sites on the molecule, the copper salt, when coordinated with specific nitrogen-containing ligands such as 2,2'-bipyridine or 1,10-phenanthroline, directs the oxidation exclusively to the allylic position. This coordination chemistry creates a reactive environment that activates the tert-butyl peroxyacetate oxidant precisely where needed, preventing over-oxidation to carboxylic acids or degradation of the sensitive acetal protecting group. The mechanistic precision ensures that the reaction proceeds with high conversion rates even at low temperatures, typically between 0 and 20°C, which further suppresses thermal decomposition pathways. For technical teams, understanding this catalytic cycle is crucial, as it demonstrates how ligand tuning can transform a base metal into a highly efficient tool for complex bond formation, replacing expensive noble metals without sacrificing performance or selectivity in the synthesis of high-purity OLED material or pharmaceutical precursors.
Impurity control is inherently built into this mechanism through the strategic use of protection and deprotection groups. The initial conversion of tiglic aldehyde to its dimethyl acetal shields the reactive carbonyl group from the oxidizing conditions, preventing the formation of aldol condensation byproducts or polymerization issues that often plague aldehyde chemistry. Following the oxidation, the final acidic hydrolysis step cleanly removes the protecting group to reveal the target aldehyde functionality without generating persistent organic pollutants. This sequence ensures that the final impurity profile is dominated by easily separable byproducts rather than structurally similar isomers that are difficult to purge. Consequently, the downstream purification burden is drastically reduced, allowing for the production of material that meets stringent purity specifications with minimal chromatographic intervention. This level of control is essential for maintaining batch-to-batch consistency in the commercial scale-up of complex polymer additives or active pharmaceutical ingredients.
How to Synthesize 4-Acetoxy-2-Methyl-2-Butenal Efficiently
Implementing this synthesis requires careful attention to the catalytic loading and temperature control during the oxidation phase to maximize yield and selectivity. The process begins with the acid-catalyzed acetalization of tiglic aldehyde, followed by the critical copper-mediated oxidation, and concludes with a mild hydrolysis. Detailed standard operating procedures regarding reagent grades, specific catalyst ratios, and workup protocols are essential for reproducing the high yields reported in the patent data. To ensure successful technology transfer and industrial implementation, precise adherence to the reaction parameters described in the intellectual property is required.
- React tiglic aldehyde with methanol using an acidic catalyst to form the acetal intermediate (I).
- Perform copper-catalyzed allylic oxidation on the acetal using tert-butyl peroxyacetate to generate acetal ester (II).
- Hydrolyze the acetal ester under acidic conditions to yield the final 4-acetoxy-2-methyl-2-butenal product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals and supply chain leaders, the adoption of this patent-protected route offers tangible strategic advantages beyond mere technical elegance. The shift from expensive, hazard-prone raw materials to commodity chemicals like tiglic aldehyde fundamentally de-risks the supply chain, ensuring consistent availability and shielding the production schedule from volatile raw material markets. The elimination of heavy metal catalysts and hazardous chlorination reagents simplifies waste management protocols, leading to substantial cost savings in environmental compliance and disposal fees. Moreover, the high overall yield of the process means that less raw material is required to produce the same amount of finished goods, directly improving the cost of goods sold (COGS) and enhancing margin potential. These factors combine to create a more resilient manufacturing model that can withstand market fluctuations and regulatory pressures.
- Cost Reduction in Manufacturing: The replacement of noble metal catalysts with a copper-based system eliminates the need for expensive metal recovery processes and reduces the capital tied up in catalyst inventory. Additionally, the high selectivity of the reaction minimizes the loss of valuable intermediates to byproduct formation, ensuring that a greater proportion of input materials are converted into saleable product. This efficiency translates directly into lower production costs per kilogram, providing a competitive edge in price-sensitive markets without compromising on quality standards. The simplified process flow also reduces utility consumption, further contributing to overall operational expenditure reductions.
- Enhanced Supply Chain Reliability: By utilizing tiglic aldehyde, a stable and widely available starting material, the process avoids the supply bottlenecks associated with specialized or hazardous reagents like tert-butyl hypochlorite or ethylene oxide. The mild reaction conditions reduce the risk of unplanned shutdowns due to safety incidents or equipment failures related to high-pressure or high-temperature operations. This stability ensures a consistent flow of materials to downstream customers, reducing lead time for high-purity pharmaceutical intermediates and strengthening long-term supplier relationships. The robustness of the chemistry allows for flexible production scheduling to meet fluctuating demand patterns.
- Scalability and Environmental Compliance: The significant reduction in wastewater generation and the absence of halogenated waste streams simplify the environmental permitting process and reduce the burden on effluent treatment plants. This eco-friendly profile aligns with increasingly strict global regulations on industrial emissions, future-proofing the manufacturing site against regulatory changes. The process is designed for easy scale-up, with reaction parameters that can be safely translated from laboratory to pilot and finally to full commercial production scales. This scalability ensures that supply can grow in tandem with market demand for Vitamin A and related derivatives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific technical disclosures and comparative data found within the patent documentation to provide clarity on feasibility and advantages. Understanding these details is critical for stakeholders evaluating the potential for technology adoption or procurement partnerships.
Q: What are the primary advantages of the tiglic aldehyde route over isoprene-based methods?
A: The tiglic aldehyde route eliminates the use of sodium hypochlorite, significantly reducing wastewater generation and environmental pollution compared to isoprene chlorination methods. It also avoids the complex purification steps associated with unstable intermediates found in ethylene oxide routes.
Q: How does the copper catalyst system impact impurity profiles?
A: The specific copper salt and nitrogen ligand system provides high selectivity for allylic oxidation, minimizing over-oxidation byproducts. This results in a cleaner reaction profile, simplifying downstream purification and ensuring high-purity specifications suitable for pharmaceutical applications.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes mild reaction conditions (0-65°C) and readily available starting materials like tiglic aldehyde. The absence of expensive noble metals and the high overall yield (>82%) make it economically viable and scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acetoxy-2-Methyl-2-Butenal Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global supply of essential vitamins and pharmaceuticals. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN108997117B can be successfully translated into robust industrial processes. We are committed to maintaining stringent purity specifications and operating rigorous QC labs to guarantee that every batch of 4-acetoxy-2-methyl-2-butenal meets the exacting standards required by our international partners. Our infrastructure is designed to support the complex chemistry involved in copper-catalyzed oxidations, providing a secure and compliant manufacturing environment.
We invite you to collaborate with us to optimize your supply chain for Vitamin A intermediates. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate how our manufacturing capabilities can enhance your product portfolio. Let us help you secure a stable, cost-effective source of this critical chemical building block.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
