Advanced Electrochemical Synthesis of 4-Acetoxy-2-methyl-2-butenal for Commercial Scale-up
The synthesis of 4-acetoxy-2-methyl-2-butenal, widely known in the industry as 'pentaldehyde,' represents a critical bottleneck in the global supply chain for Vitamin A production. As a key C5 building block, its efficient manufacture dictates the cost and availability of downstream retinoids. Patent CN111270261A introduces a paradigm-shifting approach to this challenge by leveraging electrochemical oxidation technology. Unlike traditional thermal processes that rely on harsh oxidants or expensive transition metals, this invention utilizes electrons as the primary reagent, mediated by N-hydroxysuccinimide (NHS) and stabilized by fatty acid iron salts. This technological leap not only achieves a remarkable yield of over 95% but also operates under exceptionally mild conditions, typically between 10°C and 40°C. For R&D directors and procurement strategists, this signals a move away from volatile chemical markets towards a more controlled, electricity-driven manufacturing model that promises enhanced safety and reduced environmental footprint.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of pentaldehyde has been plagued by inefficient multi-step sequences and severe environmental liabilities. One prominent legacy route, disclosed in patent US5453547, initiates with dimethoxyacetone and proceeds through acetylene addition, hydrogenation, and copper-catalyzed isomerization. As illustrated in the reaction scheme below, this pathway is inherently cumbersome, involving at least four distinct chemical transformations before hydrolysis yields the target aldehyde. The cumulative effect of these steps results in a dismal overall yield ranging merely from 35% to 45%, representing a massive loss of raw material value. Furthermore, the reliance on acetylene introduces significant safety hazards due to its explosive nature, while the copper catalysts require rigorous removal protocols to meet pharmaceutical purity standards.
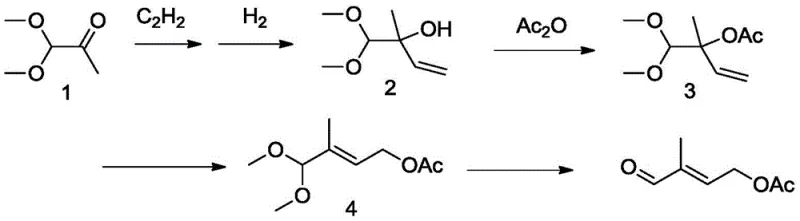
Alternative pathways have attempted to streamline this synthesis but introduced different critical failures. For instance, the isoprene-based route described in US5424478 utilizes sodium hypochlorite for chlorohydrin formation followed by DMSO oxidation. While chemically feasible, this method generates voluminous quantities of saline wastewater—specifically noted as producing ten times the mass of the product in waste effluent. This high COD and salt load creates an insurmountable burden for modern wastewater treatment facilities, rendering the process economically unviable in regions with strict environmental compliance. Similarly, the rhodium-catalyzed carbonylation of butenediol derivatives, while effective, ties the production cost directly to the fluctuating market prices of precious group metals, creating unacceptable supply chain volatility for bulk commodity chemicals.
The Novel Approach
The electrochemical strategy presented in CN111270261A fundamentally重构 s the synthesis logic by bypassing stoichiometric oxidants entirely. Instead of consuming chemical oxidants that become waste, this method uses molecular oxygen from the air as the terminal oxidant, driven by an electric current. The core innovation lies in the use of N-hydroxysuccinimide as a redox mediator. In this system, the substrate, prenol acetate, does not react directly at the electrode surface, which would require high voltages and lead to decomposition. Instead, the mediator is oxidized at the anode to form a stable nitroxyl radical species. This radical then selectively abstracts a hydrogen atom from the allylic position of the prenol acetate in the bulk solution. This indirect electrolysis mechanism ensures high selectivity and prevents the over-oxidation of the sensitive aldehyde product, a common failure mode in direct anodic oxidation. The result is a streamlined, single-step conversion that eliminates the need for hazardous reagents like chlorine or high-pressure synthesis gas.
Mechanistic Insights into NHS-Mediated Electro-Oxidation
To fully appreciate the robustness of this technology for commercial scale-up, one must understand the intricate catalytic cycle occurring within the electrochemical cell. The process begins at the anode, where N-hydroxysuccinimide undergoes a one-electron oxidation to generate the corresponding N-oxyl radical. This step is kinetically favorable and occurs at a relatively low potential, typically between 1.0V and 1.5V. Once generated, this persistent radical diffuses into the solution and interacts with the prenol acetate substrate. The mechanism involves the abstraction of an allylic hydrogen atom, generating a carbon-centered radical on the substrate and regenerating the hydroxylamine form of the catalyst. This carbon-centered radical is highly reactive towards molecular oxygen, which is continuously sparged into the reactor. The rapid addition of oxygen forms a peroxyl radical intermediate, which subsequently undergoes reduction and protonation steps at the cathode or via interaction with the electrolyte to yield the final aldehyde functionality and water as the only byproduct.
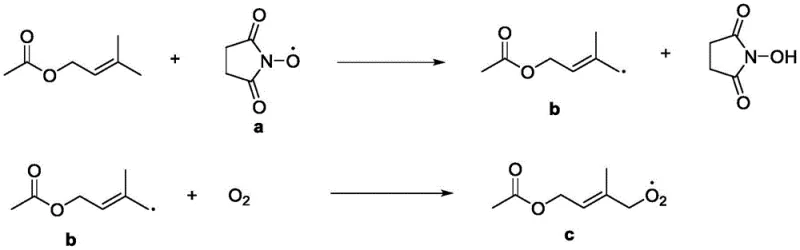
A crucial, often overlooked component of this system is the role of the fatty acid iron salt auxiliary. In many electro-organic syntheses, the accumulation of peroxyl radicals can lead to chain termination or polymerization side reactions. The patent specifies that iron salts, such as iron acetate or iron naphthenate, act as Lewis acid stabilizers for these radical intermediates. By coordinating with the peroxyl species, the iron auxiliary lowers the chemical potential energy of the transition state, effectively accelerating the rate-determining step of oxygen insertion. This synergistic effect between the organic mediator and the inorganic auxiliary allows the reaction to proceed rapidly even at ambient temperatures (20°C - 30°C). Furthermore, the cathode reaction, typically the reduction of protons to hydrogen gas or the reduction of oxygen species, is balanced to maintain charge neutrality without generating destructive reducing agents that could attack the aldehyde product. This precise control over the redox environment is what enables the reported purity levels exceeding 99%.
How to Synthesize 4-Acetoxy-2-methyl-2-butenal Efficiently
Implementing this electrochemical protocol requires careful attention to reactor design and parameter control to maximize the efficiency of the electron transfer. The process is designed to be operationally simple, utilizing a standard undivided cell configuration which reduces equipment costs compared to membrane cells. The key to success lies in maintaining the optimal ratio of acetic acid to water in the electrolyte, which influences the conductivity and the solubility of the organic substrate. Operators must ensure a steady flow of oxygen or air to prevent the depletion of the oxidant, which would stall the radical cycle. The following guide outlines the standardized procedure derived from the patent examples, ensuring reproducible high-yield results suitable for pilot and commercial operations.
- Prepare the electrolyte solution by mixing prenol acetate, acetic acid, water, N-hydroxysuccinimide catalyst, and fatty acid iron salt auxiliary in a non-diaphragm reactor.
- Introduce oxygen or air into the reactor and apply a constant voltage between 1.0V and 1.5V using a platinum anode and carbon cathode.
- Maintain the reaction temperature between 10°C and 40°C for 1 to 4 hours, then remove solvents and purify the crude product via vacuum rectification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this electrochemical methodology offers tangible strategic advantages beyond mere technical elegance. The most immediate impact is the drastic simplification of the raw material portfolio. By eliminating the need for acetylene, synthesis gas, or sodium hypochlorite, the facility removes several high-hazard materials from its inventory, significantly lowering insurance premiums and safety compliance costs. Moreover, the substitution of expensive rhodium or palladium catalysts with commodity-grade iron salts and recyclable organic mediators decouples production costs from the volatile precious metals market. This stability allows for more accurate long-term budgeting and pricing contracts with downstream Vitamin A manufacturers, enhancing the supplier's competitiveness in tender processes.
- Cost Reduction in Manufacturing: The economic argument for this technology is anchored in the elimination of stoichiometric chemical oxidants and precious metal catalysts. In traditional routes, the cost of the oxidant and the subsequent waste treatment often exceeds the cost of the raw materials themselves. By using electricity and air, the variable cost of goods sold is significantly compressed. Additionally, the high atom economy means that less raw material is required to produce the same amount of product, further driving down the unit cost. The ability to reuse the electrolyte solution containing the mediator and auxiliary agent across multiple batches amortizes the cost of these additives over a much larger production volume, leading to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: Supply continuity is frequently disrupted by the logistical challenges of transporting hazardous gases like acetylene or chlorine. This electrochemical process relies on prenol acetate, a stable liquid that is easily stored and transported in standard ISO tanks. The reaction itself operates at atmospheric pressure and near-ambient temperatures, removing the need for high-pressure autoclaves or cryogenic cooling systems that are prone to maintenance downtime. This robustness ensures consistent production schedules and reduces the risk of force majeure events related to equipment failure or hazardous material transport restrictions, thereby securing the supply line for critical pharmaceutical customers.
- Scalability and Environmental Compliance: Scaling electrochemical processes is traditionally challenging due to mass transfer limitations, but the use of a soluble mediator in this specific design mitigates those issues, allowing for straightforward scale-up from laboratory to multi-ton reactors. From an environmental perspective, the reduction in wastewater generation is profound. Unlike the hypochlorite route which produces ten times its weight in saline waste, this method generates minimal aqueous waste that is primarily acetic acid and water, both of which are biodegradable and easily treated. This 'green' profile facilitates faster regulatory approvals in jurisdictions with strict environmental laws and aligns with the sustainability goals of major multinational pharmaceutical corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on performance metrics and operational requirements. Understanding these details is essential for engineering teams evaluating the feasibility of retrofitting existing facilities or designing new production lines for 4-acetoxy-2-methyl-2-butenal.
Q: Why is the electrochemical method superior to traditional Rhodium-catalyzed routes?
A: Traditional routes often rely on expensive precious metal catalysts like Rhodium and require high-pressure synthesis gas. The electrochemical method replaces these with inexpensive organic mediators and iron salts, operating at atmospheric pressure and mild temperatures, significantly lowering capital and operational expenditures.
Q: How does this process address environmental concerns associated with older synthesis methods?
A: Conventional hypochlorite oxidation methods generate massive amounts of high-salt, high-COD wastewater (up to 10 times the product mass). This electrochemical process uses oxygen as the terminal oxidant and allows for electrolyte reuse, drastically reducing liquid waste discharge and eliminating halogenated byproducts.
Q: What purity levels can be achieved with this new electrochemical protocol?
A: The patent data indicates that through optimized voltage control and the use of specific auxiliary agents, the process achieves a chemical yield exceeding 95% and a final product purity greater than 99% after standard vacuum rectification, meeting stringent pharmaceutical intermediate specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acetoxy-2-methyl-2-butenal Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of novel synthetic technologies requires a partner with deep technical expertise and proven manufacturing capabilities. Our team has extensively analyzed the electrochemical pathways described in CN111270261A and possesses the engineering know-how to translate these laboratory successes into robust commercial processes. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench to plant is seamless. Our facilities are equipped with state-of-the-art electrochemical reactors and stringent purity specifications are maintained through our rigorous QC labs, guaranteeing that every batch of 4-acetoxy-2-methyl-2-butenal meets the exacting standards required for Vitamin A synthesis.
We invite potential partners to engage with our technical procurement team to discuss how this innovative manufacturing route can optimize your supply chain. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to request specific COA data and route feasibility assessments to verify the superior quality and economic benefits of our electrochemically produced intermediates. Let us help you secure a sustainable and cost-effective supply of this critical pharmaceutical building block.
