Advanced Chiral Catalysis for High-Purity Levetiracetam Manufacturing and Commercial Scale-Up
Advanced Chiral Catalysis for High-Purity Levetiracetam Manufacturing and Commercial Scale-Up
The pharmaceutical industry continuously seeks robust synthetic routes for antiepileptic agents that balance high stereochemical purity with operational simplicity. Patent CN113511994B, published in March 2023, introduces a transformative preparation method for levetiracetam, a critical active pharmaceutical ingredient (API). This innovation addresses the longstanding challenge of racemization often encountered in traditional alkylation processes by employing a specialized chiral BINOL-derived phosphoric acid ligand. The technology enables the direct reaction of 2-halobutanamide with 2-pyrrolidone, bypassing the need for complex resolution steps while achieving exceptional enantiomeric excess. For R&D directors and process chemists, this represents a significant leap forward in asymmetric organocatalysis, offering a pathway to >99% chiral purity and yields exceeding 85%. 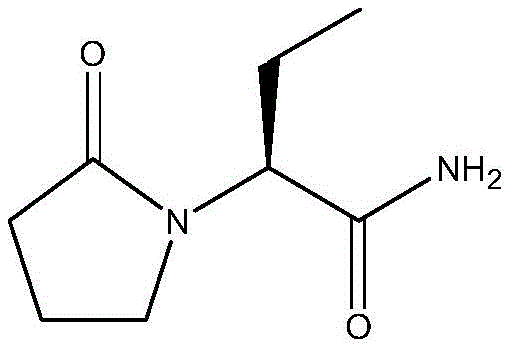
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of levetiracetam has relied heavily on chemical resolution methods or routes starting from expensive chiral amino acids, both of which impose significant economic and operational burdens. Traditional nucleophilic substitution reactions using 2-bromobutanamide and 2-pyrrolidone often necessitate strong alkali catalysts like sodium methoxide. While effective for bond formation, these harsh basic conditions frequently induce racemization at the chiral center, leading to a mixture of (R) and (S) enantiomers that requires costly and yield-reducing resolution steps to separate. Furthermore, alternative routes involving the activation of carboxyl groups via mixed anhydrides introduce additional unit operations, increasing the risk of impurity generation and complicating the supply chain with multiple intermediate handling requirements. These legacy processes often struggle to consistently meet the stringent purity specifications demanded by global regulatory bodies without sacrificing overall process efficiency.
The Novel Approach
The methodology disclosed in CN113511994B fundamentally reengineers this transformation by replacing non-selective strong bases with a sophisticated chiral phosphoric acid catalyst system. This novel approach facilitates a direct one-pot reaction between 2-halobutanamide (specifically 2-chlorobutanamide or 2-bromobutanamide) and 2-pyrrolidone in the presence of an acid-binding agent. By utilizing a chiral BINOL-derived phosphate ligand, the reaction achieves high chiral conversion rates directly, effectively transferring the chiral information from the catalyst to the product without the need for post-reaction resolution. 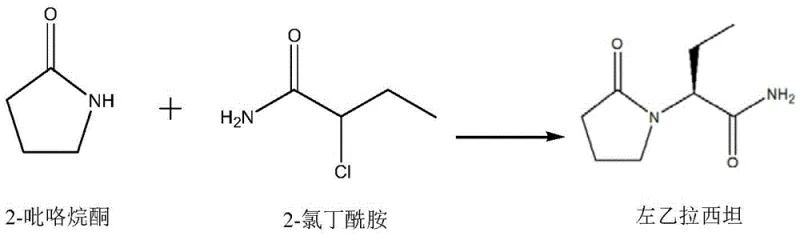 This streamlined process not only simplifies the operational workflow but also drastically reduces the generation of waste materials associated with multi-step activation and resolution protocols. The use of relatively low-toxicity raw materials and the ability to operate in common organic solvents like toluene further enhance the safety profile and environmental compliance of the manufacturing process.
This streamlined process not only simplifies the operational workflow but also drastically reduces the generation of waste materials associated with multi-step activation and resolution protocols. The use of relatively low-toxicity raw materials and the ability to operate in common organic solvents like toluene further enhance the safety profile and environmental compliance of the manufacturing process.
Mechanistic Insights into Chiral BINOL-Derived Phosphoric Acid Catalysis
The core of this technological breakthrough lies in the unique structural properties of the chiral BINOL-derived phosphoric acid ligand, which functions as a dual-function catalyst. Unlike simple Brønsted acids or bases, this ligand possesses a rigid binaphthyl backbone where the phosphorus atom is locked within a ring structure, preventing rotation and maintaining a prominent stereo-configuration throughout the reaction. This rigidity is crucial for inducing asymmetry, as it creates a well-defined chiral pocket that interacts selectively with the transition state of the nucleophilic substitution. The catalyst likely operates by simultaneously activating the nucleophile (2-pyrrolidone derivative) and organizing the electrophile (2-halobutanamide) through hydrogen bonding or ion-pairing interactions, thereby lowering the activation energy for the desired enantiomeric pathway while sterically hindering the formation of the unwanted (R)-enantiomer.
Furthermore, the specific substitution pattern on the ligand plays a decisive role in determining the level of stereoselectivity. The patent highlights the use of aryl groups, particularly the 3,5-dimethoxybenzyl substituent at the 3,3'-position of the BINOL framework. 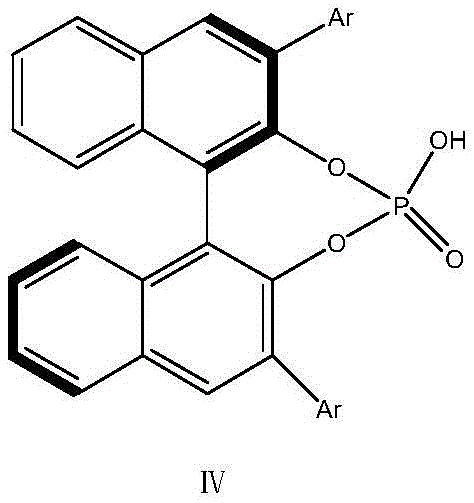 These bulky substituents exert a profound space effect during the formation of the intermediate transition state. The large volume of the chiral ligand creates significant steric hindrance that differentiates between the pro-chiral faces of the reacting species, ensuring that the reaction proceeds with high chiral selectivity. This precise steric control allows the process to achieve chiral purities greater than 99% directly from the reaction mixture, eliminating the thermodynamic drive towards racemization that plagues conventional base-catalyzed methods. The result is a highly efficient catalytic cycle that maximizes the utilization of raw materials while minimizing the formation of difficult-to-remove diastereomeric impurities.
These bulky substituents exert a profound space effect during the formation of the intermediate transition state. The large volume of the chiral ligand creates significant steric hindrance that differentiates between the pro-chiral faces of the reacting species, ensuring that the reaction proceeds with high chiral selectivity. This precise steric control allows the process to achieve chiral purities greater than 99% directly from the reaction mixture, eliminating the thermodynamic drive towards racemization that plagues conventional base-catalyzed methods. The result is a highly efficient catalytic cycle that maximizes the utilization of raw materials while minimizing the formation of difficult-to-remove diastereomeric impurities.
How to Synthesize Levetiracetam Efficiently
The implementation of this catalytic system offers a straightforward protocol suitable for immediate scale-up in GMP environments. The process begins with the formation of the nucleophilic species by treating 2-pyrrolidone with an inorganic base such as potassium carbonate or sodium hydroxide in a high-boiling, water-insoluble solvent like toluene. Following this activation, the 2-halobutanamide and the chiral catalyst are introduced, and the mixture is heated to reflux temperatures between 100°C and 130°C. The detailed standardized synthesis steps, including specific molar ratios, catalyst loading percentages, and crystallization parameters, are outlined in the guide below.
- Combine 2-pyrrolidone, a water-insoluble organic solvent like toluene, and an inorganic base such as potassium hydroxide in a reactor, stirring at ambient temperature to form the nucleophilic species.
- Add 2-chlorobutanamide or 2-bromobutanamide along with a catalytic amount (1.0-3.0% mass ratio) of the chiral BINOL-derived phosphoric acid ligand, then heat to reflux (100-130°C) for 12-16 hours.
- Upon completion, cool the mixture, perform aqueous workup to remove inorganic salts, distill off the solvent, and recrystallize the residue from an alcohol solvent like methanol or ethanol to isolate pure levetiracetam.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic technology translates into tangible strategic benefits regarding cost structure and supply reliability. The elimination of the chiral resolution step is perhaps the most significant economic driver, as resolution typically consumes up to 50% of the theoretical yield in the discarded enantiomer and requires additional solvents and processing time. By achieving high enantiomeric excess directly, this method effectively doubles the potential yield from the same amount of starting material compared to racemic synthesis followed by resolution. Additionally, the one-pot nature of the reaction reduces the number of isolation and purification stages, leading to substantial savings in solvent consumption, energy usage, and labor costs associated with multi-step batch processing.
- Cost Reduction in Manufacturing: The process utilizes inexpensive and readily available starting materials such as 2-chlorobutanamide and 2-pyrrolidone, avoiding the premium pricing associated with chiral amino acid precursors. The catalytic loading is remarkably low, ranging from 1.0% to 3.0% by mass, which minimizes the cost contribution of the sophisticated chiral ligand to the final product. Furthermore, the use of recoverable solvents like toluene and the avoidance of expensive coupling reagents or activating agents significantly lowers the variable cost of goods sold (COGS), allowing for more competitive pricing in the generic API market.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, which tolerate standard industrial heating and stirring equipment, ensures high reproducibility across different manufacturing sites. The raw materials are commodity chemicals with stable global supply chains, reducing the risk of bottlenecks associated with specialized chiral building blocks. The simplified workflow also shortens the overall production cycle time, enabling faster turnaround from order to delivery and improving the agility of the supply chain to respond to market demand fluctuations without the need for excessive safety stock.
- Scalability and Environmental Compliance: From an EHS perspective, the replacement of strong alkalis like sodium methoxide with milder inorganic bases and the use of low-toxicity reagents reduces the hazard profile of the manufacturing process. The high atom economy and reduced waste generation align with green chemistry principles, facilitating easier regulatory approval and waste disposal management. The process is inherently scalable from kilogram to multi-ton quantities, as the exothermic profile is manageable and the crystallization steps utilize common alcohol solvents, ensuring that quality remains consistent regardless of batch size.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this chiral synthesis route. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on catalyst selection, reaction parameters, and purification strategies.
Q: How does the chiral BINOL-derived phosphoric acid ligand improve enantioselectivity compared to traditional bases?
A: Unlike traditional strong bases like sodium methoxide which cause racemization, the chiral BINOL-derived phosphoric acid acts as a dual-function catalyst. Its rigid ring structure and bulky 3,5-dimethoxybenzyl substituents create a specific chiral environment that transfers chirality effectively during the nucleophilic substitution, achieving over 99% chiral purity without resolution.
Q: What are the optimal reaction conditions for maximizing yield in this process?
A: The patent specifies using a water-insoluble organic solvent with a boiling point above 100°C, preferably toluene, under reflux conditions at 100-130°C. The molar ratio of 2-pyrrolidone to 2-halobutanamide should be maintained between 1:1.0 and 1:1.3, with a catalyst loading of 1.0-3.0% relative to the halobutanamide mass.
Q: Does this method require a chiral resolution step?
A: No, one of the primary advantages of this method is the elimination of the resolution step. The asymmetric catalysis directly produces the (S)-enantiomer with high selectivity (>99% ee), significantly simplifying the downstream purification process and reducing material waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Levetiracetam Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our technical team has thoroughly analyzed the potential of the chiral BINOL-derived phosphoric acid catalysis described in CN113511994B and is prepared to integrate this high-efficiency route into our production capabilities. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including chiral HPLC analysis to guarantee >99% ee, meeting the exacting standards required for antiepileptic drug intermediates.
We invite potential partners to engage with our technical procurement team to discuss how this innovative synthesis can optimize your supply chain. By leveraging our expertise in asymmetric catalysis and process development, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our commitment to technological excellence can drive value for your organization.
