Advanced Strained-Ring Phosphine Catalysts for Efficient Pharmaceutical Intermediate Manufacturing
Advanced Strained-Ring Phosphine Catalysts for Efficient Pharmaceutical Intermediate Manufacturing
The landscape of organophosphorus catalysis is undergoing a significant transformation with the introduction of novel high-strain cyclic structures that offer superior reactivity profiles compared to traditional acyclic analogues. Patent CN108250239B discloses a groundbreaking class of amino-substituted pentavalent four-membered small ring phosphine compounds, specifically designed to leverage ring strain for enhanced catalytic performance. These compounds, characterized by a phosphetane 1-oxide core substituted with various amine groups, represent a critical advancement in the field of organocatalysis, particularly for redox-neutral transformations. For R&D directors and process chemists, the ability to utilize a catalyst that combines the stability of a P(V) oxide with the latent reactivity of a strained ring opens new avenues for synthesizing complex pharmaceutical intermediates with higher atom economy. The structural versatility allows for fine-tuning of electronic and steric properties by modifying the amine substituents, making this platform technology adaptable to a wide range of synthetic challenges.
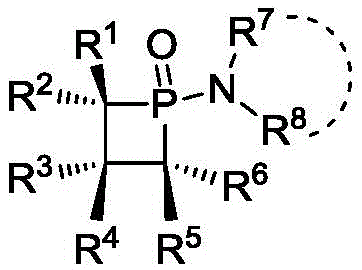
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional phosphine-mediated reactions, such as the classic Wittig reaction, have long relied on stoichiometric amounts of reagents like triphenylphosphine (PPh3). While effective, this approach generates equimolar amounts of triphenylphosphine oxide (TPPO) as a byproduct, which is notoriously difficult to remove from the final product mixture, often requiring extensive chromatography or recrystallization steps that reduce overall yield and increase waste. Furthermore, the regeneration of PPh3 from TPPO is energetically demanding and typically requires harsh reducing agents, making catalytic cycles impractical for many industrial applications. The lack of tunability in simple aryl phosphines also limits their ability to induce stereocontrol or accelerate reaction rates in sterically hindered substrates. For procurement managers, the reliance on stoichiometric phosphines translates to higher raw material costs and significant downstream processing expenses associated with waste disposal and purification, creating a bottleneck in the cost reduction in pharmaceutical intermediates manufacturing.
The Novel Approach
The innovative strategy presented in the patent data utilizes a four-membered phosphetane ring system, which introduces substantial angle strain around the phosphorus atom. This strain energy effectively lowers the activation barrier for the reduction of the P(V) oxide back to the active P(III) species, facilitating a true catalytic cycle when paired with appropriate terminal reductants like silanes. Unlike the planar and rigid structure of triphenylphosphine, the puckered four-membered ring offers a unique steric environment that can enhance selectivity and reaction rates. The incorporation of amino substituents further modulates the electron density at the phosphorus center, stabilizing the transition states involved in the catalytic cycle. This approach not only minimizes phosphine oxide waste but also allows for the use of catalytic loadings, drastically reducing the material intensity of the process. For supply chain heads, this shift from stoichiometric to catalytic processes implies a more streamlined workflow with fewer unit operations, directly contributing to reducing lead time for high-purity pharmaceutical intermediates.
Mechanistic Insights into Phosphetane-Catalyzed Intramolecular Wittig Reactions
The catalytic efficacy of these amino-substituted phosphetane oxides stems from their ability to undergo reversible redox cycling between the pentavalent oxide state and the trivalent phosphine state. In the proposed mechanism, the P(V) oxide precursor is first reduced in situ by a silicon-based reductant to generate the active P(III) phosphetane species. This highly reactive trivalent phosphorus center then attacks the carbonyl substrate to form an oxaphosphetane intermediate, which subsequently collapses to release the olefin product and regenerate the P(V) oxide catalyst. The key differentiator here is the ring strain; the four-membered ring in the catalyst destabilizes the P=O bond relative to acyclic analogues, making the reduction step thermodynamically more favorable. This mechanistic feature ensures that the catalyst turnover number (TON) remains high throughout the reaction, preventing the accumulation of inactive oxidized species. Understanding this redox potential is crucial for process optimization, as it dictates the choice of reductant and the reaction temperature required to maintain steady-state catalysis.
Furthermore, the presence of the amino group adjacent to the phosphorus atom plays a pivotal role in stabilizing the catalyst structure and influencing its solubility profile. The nitrogen lone pair can engage in hyperconjugative interactions with the phosphorus d-orbitals, fine-tuning the nucleophilicity of the P(III) species. This electronic modulation is essential for controlling the chemoselectivity of the reaction, ensuring that the catalyst reacts preferentially with the intended carbonyl group over other electrophilic sites present in complex drug molecules. From an impurity control perspective, the robust nature of the phosphetane ring prevents premature decomposition or side reactions that often plague sensitive phosphine catalysts. This stability translates to a cleaner reaction profile with fewer unidentified impurities, simplifying the purification process and ensuring that the final API intermediate meets stringent regulatory specifications for residual metals and organic volatiles.
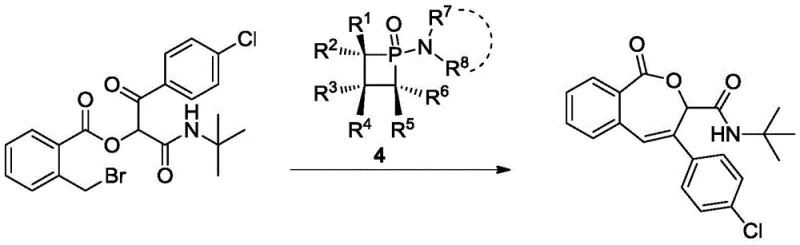
How to Synthesize 2,2,3,4,4-Pentamethylphosphetane 1-Oxide Derivatives Efficiently
The synthesis of these valuable catalysts follows a concise three-step sequence that begins with readily available polysubstituted alkenes, ensuring a robust and scalable supply chain. The process initiates with a Lewis acid-catalyzed [2+2] cycloaddition or electrophilic cyclization involving phosphorus trichloride, which constructs the strained four-membered ring skeleton with high regioselectivity. Subsequent functionalization with amines introduces the necessary nitrogen substituents, and a final mild oxidation step secures the stable P(V) oxide form suitable for storage and handling. This linear synthetic route avoids the use of expensive transition metals or cryogenic conditions, making it highly attractive for commercial production. The detailed standardized synthesis steps for producing these catalysts with consistent quality are outlined in the guide below.
- React polysubstituted alkyl propylene with PCl3 and AlCl3 in dichloromethane at 0°C to form the chlorinated intermediate.
- Perform nucleophilic substitution with amines in toluene using triethylamine at 60°C to construct the four-membered P-N ring.
- Oxidize the trivalent phosphine intermediate using hydrogen peroxide at room temperature to obtain the final pentavalent phosphetane 1-oxide.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel class of phosphetane catalysts offers substantial strategic benefits for chemical procurement and supply chain management, primarily driven by the simplification of the manufacturing process and the reduction of material costs. By shifting from stoichiometric phosphine reagents to a catalytic system, manufacturers can significantly decrease the volume of raw materials required per batch, leading to direct cost savings in reagent procurement. The elimination of stoichiometric triphenylphosphine oxide waste also reduces the burden on waste treatment facilities and lowers the environmental compliance costs associated with hazardous waste disposal. Moreover, the synthesis of the catalyst itself relies on commodity chemicals like propylene derivatives and phosphorus trichloride, which are widely available and subject to stable pricing, mitigating the risk of supply disruptions caused by niche reagent shortages.
- Cost Reduction in Manufacturing: The transition to a catalytic process fundamentally alters the cost structure of Wittig-type transformations by minimizing the consumption of high-mass phosphine reagents. Since the catalyst is regenerated in each cycle, the effective cost per mole of product is drastically lower compared to traditional methods that consume one equivalent of phosphine per equivalent of product. Additionally, the simplified workup procedures resulting from cleaner reaction profiles reduce the consumption of solvents and silica gel for purification, further driving down operational expenditures. These efficiencies compound at scale, offering a competitive margin advantage for producers of high-volume pharmaceutical intermediates without compromising on product quality.
- Enhanced Supply Chain Reliability: The synthetic route for these catalysts utilizes robust, non-proprietary starting materials that are sourced from established global supply chains, ensuring long-term availability and price stability. Unlike specialized ligands that may rely on single-source suppliers or complex multi-step syntheses, the production of these phosphetane oxides can be easily replicated by multiple contract manufacturing organizations. This redundancy in the supply base reduces the risk of bottlenecks and ensures continuity of supply even during market fluctuations. Furthermore, the stability of the final P(V) oxide products allows for extended shelf life and simplified logistics, as they do not require the stringent inert atmosphere storage conditions often needed for sensitive trivalent phosphines.
- Scalability and Environmental Compliance: The reaction conditions described in the patent, such as operating temperatures between 0°C and 60°C and the use of common solvents like dichloromethane and toluene, are fully compatible with existing standard reactor infrastructure. This compatibility eliminates the need for capital-intensive equipment upgrades, facilitating rapid technology transfer from laboratory to pilot and commercial scales. From an environmental standpoint, the catalytic nature of the process aligns with green chemistry principles by reducing the E-factor (mass of waste per mass of product), helping companies meet increasingly strict sustainability targets and regulatory requirements for solvent and heavy metal residues in fine chemical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of these amino-substituted phosphetane catalysts in industrial processes. These insights are derived directly from the experimental data and technical specifications provided in the patent literature, aiming to clarify the operational parameters and potential benefits for prospective users. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing production lines.
Q: What is the primary advantage of using strained-ring phosphetane catalysts over traditional triphenylphosphine?
A: The high ring strain in the four-membered phosphetane structure facilitates easier redox cycling between P(III) and P(V) states, significantly improving catalytic efficiency and allowing for catalytic rather than stoichiometric usage in Wittig-type reactions.
Q: Are these catalysts scalable for industrial production?
A: Yes, the synthesis utilizes readily available raw materials like polysubstituted propylene and standard reagents (PCl3, AlCl3), operating under mild temperatures (0°C to 60°C), which supports safe and cost-effective commercial scale-up.
Q: What types of reactions can these catalysts effectively promote?
A: These amino-substituted phosphetane oxides are specifically designed to catalyze intramolecular Wittig reactions efficiently, offering a greener alternative to traditional stoichiometric phosphine methods in complex molecule synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phosphetane Oxide Catalyst Supplier
As the demand for efficient and sustainable catalytic solutions grows, NINGBO INNO PHARMCHEM stands ready to support your development and production needs with our advanced capabilities in organophosphorus chemistry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from bench-scale discovery to full-scale manufacturing is seamless and risk-free. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications, guaranteeing that every batch of catalyst delivered meets the exacting standards required for GMP-compliant API synthesis. We understand the critical nature of catalyst performance in determining the success of your synthetic route and are committed to providing materials of the highest consistency and reliability.
We invite you to collaborate with our technical team to explore how these novel phosphetane catalysts can optimize your specific processes and drive down your overall manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your current production volumes and specific route requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate the tangible value our catalysts can bring to your supply chain, ensuring a partnership built on transparency, quality, and mutual success.
