Scalable Synthesis of Novel Triazole Glycoside Azaindole Derivatives for Antiviral Applications
Scalable Synthesis of Novel Triazole Glycoside Azaindole Derivatives for Antiviral Applications
The pharmaceutical landscape is constantly evolving towards more complex heterocyclic scaffolds that offer superior pharmacokinetic profiles. Patent CN112110969B introduces a groundbreaking methodology for synthesizing a specific triazole glycoside derivative of 3-nitro-1-ethyl formate-7-azaindole. This innovation addresses critical challenges in drug design by merging the biological potency of the 7-azaindole core with the solubility-enhancing properties of a galactose-triazole hybrid. For R&D directors and procurement specialists, this represents a significant opportunity to access high-value intermediates for anticancer and antiviral pipelines. The strategic combination of these moieties results in a molecule with an optimized lipid-water partition coefficient, crucial for membrane permeability.
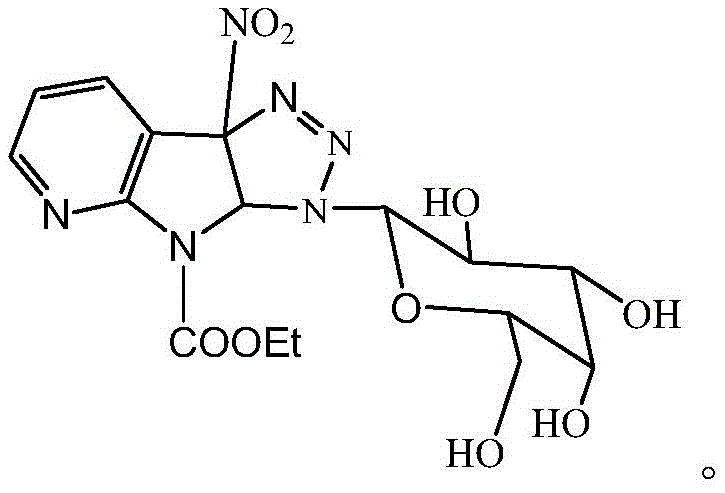
This technical insight report dissects the proprietary synthesis route detailed in the patent, highlighting its feasibility for commercial scale-up. By leveraging a metal-free thermal cycloaddition strategy, the process eliminates the need for costly transition metal catalysts often associated with click chemistry. This not only simplifies the purification workflow but also aligns with stringent regulatory requirements regarding heavy metal residues in active pharmaceutical ingredients (APIs). As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is vital for ensuring supply chain continuity and cost efficiency in the production of next-generation therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to functionalizing the 7-azaindole scaffold often rely on harsh electrophilic substitution reactions that suffer from poor regioselectivity and limited substrate tolerance. Conventional glycosylation methods frequently require protecting group manipulations that add multiple steps, reducing overall yield and increasing waste generation. Furthermore, direct coupling of sugars to electron-deficient heterocycles can be kinetically sluggish, necessitating high temperatures or aggressive Lewis acids that compromise the integrity of sensitive functional groups like the nitro group. These inefficiencies create bottlenecks in the supply chain, leading to higher costs and longer lead times for high-purity pharmaceutical intermediates.
The Novel Approach
The methodology outlined in CN112110969B circumvents these issues through a clever application of 1,3-dipolar cycloaddition chemistry. By utilizing an azido-sugar and an electron-deficient azaindole dipolarophile, the reaction proceeds with high atom economy and excellent stereocontrol. This route avoids the use of toxic copper catalysts typically required for azide-alkyne cycloadditions, thereby streamlining the downstream processing. The subsequent deprotection step employs mild basic conditions using sodium methoxide, ensuring the stability of the triazole ring while efficiently revealing the hydrophilic hydroxyl groups of the sugar. This streamlined approach offers substantial cost savings in API manufacturing by reducing solvent consumption and purification complexity.
Mechanistic Insights into Thermal [3+2] Dipolar Cycloaddition
The core transformation in this synthesis is the formation of the triazole ring via a thermal [3+2] dipolar cycloaddition. The 3-nitro-1-ethyl formate-7-azaindole acts as a dipolarophile, where the electron-withdrawing nitro group at the 3-position activates the adjacent double bond for nucleophilic attack by the azide. Unlike metal-catalyzed variants, this thermal process relies on the intrinsic electronic properties of the reactants, driven by reflux conditions in methanol. This mechanism ensures that the resulting triazole ring is formed with high fidelity, creating a robust linkage between the heterocyclic core and the carbohydrate unit. The absence of metal coordination complexes simplifies the reaction profile, making it highly predictable and easier to control on a large scale.
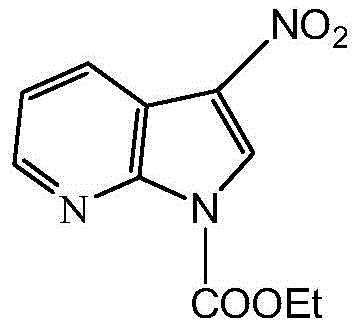
Following the cycloaddition, the deprotection mechanism involves the nucleophilic attack of methoxide ions on the carbonyl carbons of the acetyl protecting groups. This transesterification reaction cleaves the ester bonds, releasing methyl acetate and regenerating the free hydroxyl groups on the galactose ring. The use of a cation exchange resin for neutralization is a critical process control point, preventing base-catalyzed degradation of the sensitive triazole or nitro functionalities. This careful balance of reactivity ensures that the final product retains its structural integrity and biological potential, meeting the rigorous purity specifications required for clinical evaluation.
How to Synthesize Triazole Glycoside Derivative Efficiently
The synthesis protocol described in the patent provides a robust framework for producing this complex intermediate. It begins with the N-protection of 7-azaindole, followed by regioselective nitration to install the key activating group. The subsequent cycloaddition and deprotection steps are designed to maximize yield while minimizing impurities. For process chemists looking to implement this route, attention to stoichiometry and temperature control during the nitration step is paramount to prevent over-nitration or decomposition. The detailed standardized synthesis steps below outline the precise conditions required to achieve reproducible results.
- Synthesize N-ethyl formate-7-azaindole by reacting 7-azaindole with sodium hydride and ethyl chloroformate in DMF under ice bath conditions.
- Perform nitration using acetic anhydride and concentrated nitric acid to introduce the nitro group at the 3-position of the azaindole ring.
- Conduct a thermal [3+2] dipolar cycloaddition between the nitro-azaindole intermediate and 1-azido-peracetylgalactose in refluxing methanol.
- Execute deprotection of the acetyl groups using sodium methoxide in a methanol/dichloromethane mixture, followed by neutralization and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthetic route offers distinct advantages due to its reliance on commodity chemicals and straightforward unit operations. The starting materials, such as 7-azaindole, ethyl chloroformate, and galactose derivatives, are readily available from global chemical suppliers, mitigating the risk of raw material shortages. The elimination of precious metal catalysts removes a significant cost driver and simplifies the validation process for GMP manufacturing. Additionally, the use of common solvents like methanol, dichloromethane, and ethyl acetate facilitates solvent recovery and recycling, contributing to a more sustainable and cost-effective production model.
- Cost Reduction in Manufacturing: The avoidance of transition metal catalysts such as copper significantly lowers the cost of goods sold (COGS) by removing the need for expensive metal scavengers and extensive purification protocols. The thermal cycloaddition proceeds efficiently without specialized equipment, allowing for utilization of existing reactor infrastructure. Furthermore, the high atom economy of the cycloaddition step minimizes waste disposal costs, while the mild deprotection conditions reduce energy consumption associated with heating and cooling cycles.
- Enhanced Supply Chain Reliability: The synthetic pathway utilizes stable intermediates that can be stockpiled without significant degradation, ensuring a buffer against supply disruptions. The reagents involved, including sodium hydride and acetic anhydride, are bulk commodities with established supply chains, reducing the likelihood of procurement delays. This stability allows for better production planning and inventory management, ensuring consistent delivery of high-purity azaindole derivatives to downstream pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory synthesis to multi-kilogram pilot production. The absence of heavy metals simplifies environmental compliance and wastewater treatment, aligning with green chemistry principles. The use of column chromatography for purification, while effective at lab scale, can be transitioned to crystallization or preparative HPLC for larger batches, further enhancing the environmental profile and operational efficiency of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of this novel derivative. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation. Understanding these aspects is crucial for evaluating the feasibility of integrating this intermediate into your drug discovery programs.
Q: What is the primary advantage of introducing a triazole glycoside structure to 7-azaindole?
A: The introduction of the triazole glycoside moiety significantly enhances the water solubility and lipid-water partition coefficient (LogP ~2.87), improving bioavailability and membrane permeability compared to the parent azaindole scaffold.
Q: Does this synthesis require expensive transition metal catalysts?
A: No, the key cycloaddition step utilizes a thermal [3+2] dipolar reaction in methanol without the need for copper or other transition metal catalysts, reducing heavy metal contamination risks and purification costs.
Q: What biological activities does the final compound exhibit?
A: The derivative demonstrates potent in vitro inhibitory effects against HepG2 liver cancer cells and SGC7901 gastric cancer cells, along with significant anti-Hepatitis B Virus (HBV) activity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Glycoside Derivative Supplier
At NINGBO INNO PHARMCHEM, we specialize in translating complex academic innovations into commercially viable realities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. We understand the critical nature of stringent purity specifications and operate rigorous QC labs to guarantee that every batch of high-purity azaindole derivatives meets international standards. Our commitment to quality assurance makes us the preferred partner for pharmaceutical companies seeking to advance their antiviral and oncology pipelines.
We invite you to collaborate with us to explore the full potential of this technology. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your project milestones. Let us help you accelerate your time-to-market with our superior manufacturing capabilities and deep technical expertise in heterocyclic chemistry.
