Advanced Synthesis of 3-Nitro-7-Azaindole Triazole Glycosides for Oncology Applications
Introduction to Novel Azaindole-Based Therapeutics
The pharmaceutical landscape is continuously evolving with the demand for more potent and selective therapeutic agents, particularly in the fields of oncology and antiviral therapy. Patent CN112110969B, published in July 2022, introduces a significant advancement in this domain by disclosing a novel class of triazole glycoside derivatives based on a 3-nitro-1-ethyl formate-7-azaindole scaffold. This specific chemical architecture represents a strategic fusion of the biologically privileged 7-azaindole core with a sugar-modified triazole moiety, designed to optimize pharmacokinetic profiles. The 7-azaindole unit is well-known for its presence in numerous bioactive natural products and synthetic drugs, offering a robust platform for interacting with various biological targets including kinases and viral enzymes. By functionalizing this core with a nitro group and subsequently linking it to a galactose-derived triazole, the inventors have created a molecule that exhibits promising inhibitory activity against tumor cell lines such as HepG2 and SGC7901, as well as anti-HBV potential. This development underscores the critical role of precise molecular engineering in modern drug discovery, where subtle structural modifications can lead to substantial improvements in efficacy and safety profiles.
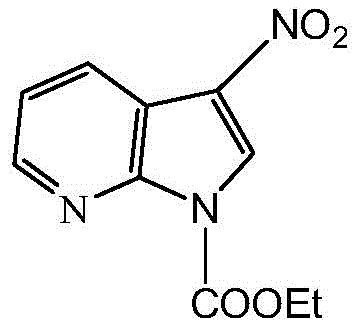
For procurement specialists and supply chain managers, understanding the structural complexity of such intermediates is vital for assessing sourcing risks and production feasibility. The core intermediate, 3-nitro-1-ethyl formate-7-azaindole, serves as the pivotal electrophilic partner in the subsequent coupling reactions. Its stability and reactivity profile dictate the success of the entire synthetic sequence. As a reliable pharmaceutical intermediate supplier, recognizing the nuances of this scaffold allows for better forecasting of raw material requirements and potential bottlenecks in the supply chain. The patent details a method that not only achieves high purity but also maintains the integrity of the sensitive nitro and ester functionalities, which is often a challenge in large-scale heterocyclic synthesis. This level of control is essential for ensuring consistent quality in the final active pharmaceutical ingredient (API), thereby reducing the risk of batch failures and costly delays in clinical development timelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the functionalization of the 7-azaindole ring system, particularly at the 3-position, has been fraught with challenges regarding regioselectivity and reaction conditions. Conventional methods often rely on harsh electrophilic substitution conditions that can lead to over-nitration or degradation of the sensitive pyrrole nitrogen. Furthermore, introducing sugar moieties onto heterocyclic scaffolds typically requires multi-step protection and deprotection strategies that utilize stoichiometric amounts of heavy metal catalysts, such as copper(I) in classic Huisgen cycloadditions. These traditional approaches not only increase the environmental footprint due to heavy metal waste but also complicate the purification process, necessitating rigorous scavenging steps to meet stringent regulatory limits for residual metals in pharmaceuticals. Additionally, the poor solubility of many intermediate species in standard organic solvents can hinder reaction kinetics, leading to prolonged reaction times and lower overall yields. For a procurement manager, these inefficiencies translate directly into higher cost of goods sold (COGS) and extended lead times, making the final drug product less competitive in the market.
The Novel Approach
The methodology outlined in patent CN112110969B offers a transformative solution to these historical bottlenecks by employing a streamlined, four-step synthetic route that maximizes efficiency and minimizes waste. The process begins with a mild N-protection of 7-azaindole using ethyl chloroformate, followed by a controlled nitration that selectively targets the 3-position without compromising the rest of the molecule. The cornerstone of this innovation is the subsequent [3+2] dipolar cycloaddition, which proceeds under thermal reflux in methanol without the need for exogenous metal catalysts. This "click chemistry" approach ensures high atom economy and exceptional regioselectivity, directly yielding the triazole-linked intermediate with minimal byproduct formation. The final deprotection step utilizes sodium methoxide, a cost-effective and readily available reagent, to reveal the free hydroxyl groups of the sugar moiety. This novel approach significantly simplifies the downstream processing, as the absence of transition metals eliminates the need for complex chelation and filtration steps. Consequently, this route facilitates cost reduction in API manufacturing by reducing both material consumption and processing time, while simultaneously enhancing the sustainability profile of the production process.
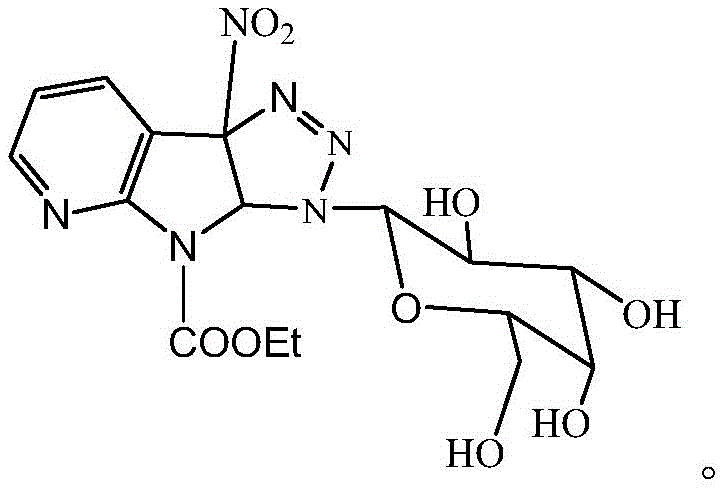
Mechanistic Insights into 1,3-Dipolar Cycloaddition
The chemical elegance of this synthesis lies in the mechanistic details of the 1,3-dipolar cycloaddition between the electron-deficient 3-nitro-7-azaindole and the azido-sugar. The presence of the strong electron-withdrawing nitro group at the 3-position of the azaindole ring significantly enhances the electrophilicity of the adjacent double bond, effectively activating it as a dipolarophile. This activation allows the reaction to proceed smoothly with the 1-azido-peracetylgalactose, which acts as the 1,3-dipole, under relatively mild thermal conditions. Unlike uncatalyzed reactions that might require extreme temperatures, this specific substrate pairing lowers the activation energy barrier, enabling the formation of the 1,2,3-triazole ring with high fidelity. The resulting triazole ring is not merely a linker; it serves as a bioisostere for amide bonds, offering enhanced metabolic stability and resistance to enzymatic hydrolysis, which is crucial for the in vivo half-life of the drug candidate. Furthermore, the stereochemical information inherent in the galactose starting material is faithfully preserved throughout the cycloaddition and deprotection steps, ensuring that the final product possesses the correct (2S,3S,4R,5S) configuration required for optimal biological recognition and binding affinity.
From a quality control perspective, understanding this mechanism is paramount for establishing robust specifications for high-purity azaindole derivatives. The reaction's high selectivity minimizes the formation of regioisomers, which are often difficult to separate and can act as genotoxic impurities. The patent data indicates that the final compound exhibits a LogP value of approximately 2.87, striking an ideal balance between lipophilicity and hydrophilicity. This physicochemical property is a direct result of the triazole glycoside architecture, which improves aqueous solubility compared to the parent azaindole while retaining sufficient lipid solubility to traverse cell membranes. For R&D directors, this implies that the synthetic route is not only chemically sound but also strategically aligned with drug-likeness criteria, reducing the likelihood of late-stage attrition due to poor ADME (Absorption, Distribution, Metabolism, and Excretion) properties. The ability to tune these properties through precise chemical synthesis demonstrates the sophistication of the underlying technology and its potential for generating a diverse library of analogs for structure-activity relationship (SAR) studies.
How to Synthesize Triazole Glycoside Derivative Efficiently
The synthesis of this complex heterocyclic system is achieved through a logical sequence of transformations that prioritize operational simplicity and scalability. The process initiates with the protection of the pyrrolic nitrogen of 7-azaindole, a critical step to prevent unwanted side reactions during the subsequent nitration. Following the introduction of the nitro group, the key carbon-nitrogen bond formation occurs via the cycloaddition reaction, which constructs the central triazole bridge. The final stage involves the removal of the acetyl protecting groups from the sugar moiety to restore the hydrophilic character of the molecule. Each step has been optimized to ensure high conversion rates and ease of isolation, typically involving standard workup procedures such as aqueous extraction and solvent evaporation. The detailed standardized synthesis steps for replicating this high-value intermediate are provided in the guide below.
- Synthesize N-ethyl formate-7-azaindole by reacting 7-azaindole with sodium hydride and ethyl chloroformate in DMF under ice bath conditions.
- Perform nitration using concentrated nitric acid in acetic anhydride to introduce the nitro group at the 3-position of the azaindole ring.
- Execute a [3+2] dipolar cycloaddition between the nitro-azaindole intermediate and 1-azido-peracetylgalactose in refluxing methanol.
- Deprotect the acetyl groups using sodium methoxide in a methanol-dichloromethane mixture, followed by neutralization and purification.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement officers, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical novelty. The primary advantage lies in the significant simplification of the manufacturing process, which directly correlates to reduced operational expenditures. By eliminating the reliance on precious metal catalysts, the process removes a major cost driver associated with both the purchase of the catalyst and the specialized equipment required for its removal and recovery. This shift towards catalyst-free thermal cycloaddition aligns perfectly with green chemistry principles, reducing the generation of hazardous waste and lowering the environmental compliance burden. Furthermore, the starting materials employed in this synthesis, such as 7-azaindole, ethyl chloroformate, and galactose derivatives, are commodity chemicals with established global supply chains. This availability ensures enhanced supply chain reliability, mitigating the risk of raw material shortages that often plague projects relying on exotic or custom-synthesized reagents. The robustness of the route also means that it is less susceptible to minor fluctuations in reaction parameters, leading to more consistent batch-to-batch quality and fewer production disruptions.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts such as copper or palladium removes the necessity for expensive ligand systems and complex metal scavenging resins, which are significant cost centers in traditional heterocyclic synthesis. Additionally, the use of common solvents like methanol, dichloromethane, and ethyl acetate allows for efficient solvent recovery and recycling, further driving down the variable costs associated with large-scale production. The high yield and selectivity of the cycloaddition step minimize the loss of valuable intermediates, ensuring that the maximum amount of raw material is converted into the desired product. This efficiency translates into substantial cost savings per kilogram of the final API, providing a competitive edge in price-sensitive markets.
- Enhanced Supply Chain Reliability: The reliance on bulk commodity chemicals ensures that the supply chain is resilient against geopolitical disruptions or single-source supplier failures. Since the reagents are widely produced for various industrial applications, procurement teams can easily source alternatives or negotiate better pricing through competitive bidding. The synthetic route's tolerance to standard laboratory and plant equipment means that it can be manufactured in existing facilities without the need for capital-intensive retrofitting or specialized reactor installations. This flexibility allows for rapid scale-up from pilot plant to commercial production, reducing lead time for high-purity intermediates and accelerating the time-to-market for the final drug product.
- Scalability and Environmental Compliance: The process avoids the generation of heavy metal waste streams, simplifying wastewater treatment and reducing the costs associated with environmental remediation and regulatory reporting. The unit operations involved, such as liquid-liquid extraction and column chromatography, are well-understood and easily scalable from gram to ton quantities using standard chemical engineering principles. The final purification steps yield a product with high purity, reducing the need for repetitive recrystallizations that can erode overall yield. This scalability ensures that the commercial scale-up of complex heterocycles can be achieved smoothly, supporting the growing demand for this potent antitumor and antiviral candidate without compromising on quality or sustainability standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this triazole glycoside derivative. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of this synthetic approach for industry stakeholders. Understanding these details is crucial for making informed decisions regarding technology transfer and partnership opportunities.
Q: What is the primary advantage of the 1,3-dipolar cycloaddition method used in this patent?
A: The 1,3-dipolar cycloaddition allows for the direct construction of the triazole ring under mild thermal conditions without requiring toxic transition metal catalysts like copper, significantly simplifying downstream purification and reducing heavy metal impurities in the final API.
Q: How does the introduction of the triazole glycoside structure affect the drug's properties?
A: Introducing the triazole glycoside structure enhances the compound's water solubility and bioavailability while maintaining lipophilicity (LogP ~2.87), which is critical for membrane permeability and effective intracellular delivery for antitumor and antiviral activity.
Q: Is this synthetic route suitable for large-scale commercial production?
A: Yes, the route utilizes commodity chemicals such as 7-azaindole, ethyl chloroformate, and galactose derivatives, and relies on standard unit operations like extraction and column chromatography, making it highly scalable for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Triazole Glycoside Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the immense potential of the 3-nitro-1-ethyl formate-7-azaindole triazole glycoside derivative in the development of next-generation therapeutics. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate this patented laboratory methodology into a robust, GMP-compliant manufacturing process. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our capability to handle complex heterocyclic chemistry, including sensitive nitration and cycloaddition reactions, positions us as the ideal partner for bringing this innovative molecule from the bench to the bedside.
We invite pharmaceutical companies and research institutions to collaborate with us to unlock the full commercial potential of this compound. By leveraging our process optimization capabilities, we can help you achieve a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data, discuss route feasibility assessments, and explore how our manufacturing solutions can accelerate your drug development pipeline. Together, we can ensure a secure and efficient supply of this critical intermediate, paving the way for breakthrough treatments in oncology and virology.
