Advanced Arylthiourea Synthesis for High-Potency VEGFR-2 Oncology Intermediates
The pharmaceutical landscape for oncology therapeutics is continuously evolving, driven by the urgent need for more effective and safer tyrosine kinase inhibitors. Patent CN101723890B introduces a groundbreaking class of arylthiourea compounds that demonstrate exceptional potency in inhibiting Vascular Endothelial Growth Factor Receptor-2 (VEGFR-2). This specific patent data highlights a strategic shift in molecular design, moving beyond traditional urea-based scaffolds to leverage the unique electronic properties of the thiourea moiety. For R&D directors and procurement specialists, understanding the synthesis and biological profile of these intermediates is critical for securing a competitive edge in drug development pipelines. The disclosed compounds not only show superior inhibition rates against tumor cell lines but also present a markedly improved safety profile regarding hepatotoxicity. This report analyzes the technical merits and commercial viability of integrating these arylthiourea intermediates into large-scale manufacturing processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis pathways for kinase inhibitors often rely on complex multi-step sequences involving harsh reagents and expensive transition metal catalysts. Many existing methods for generating urea-linked scaffolds require phosgene or its equivalents, which pose significant safety hazards and environmental compliance challenges during commercial production. Furthermore, conventional routes frequently suffer from poor atom economy and generate substantial quantities of hazardous waste, driving up the overall cost of goods sold. The purification of these intermediates can be notoriously difficult, often requiring extensive chromatographic separation that limits throughput and scalability. Additionally, older generation inhibitors like Sorafenib, while effective, are associated with notable off-target toxicities that limit their therapeutic window in clinical settings. These factors collectively create bottlenecks for supply chain managers seeking reliable and cost-efficient sources of high-quality oncology intermediates.
The Novel Approach
The methodology described in the patent offers a streamlined alternative by utilizing a direct condensation reaction between substituted isothiocyanates and a specific aminopyridine intermediate. This approach eliminates the need for hazardous phosgene reagents, replacing them with safer coupling conditions that proceed efficiently in polar aprotic solvents. The reaction tolerates a wide range of functional groups, allowing for the rapid generation of diverse analog libraries without compromising yield or purity. By operating at moderate temperatures ranging from 20°C to 100°C, the process reduces energy consumption and simplifies reactor requirements for scale-up. This synthetic flexibility enables manufacturers to adapt quickly to changing market demands for specific substituted derivatives. The robustness of this chemistry ensures consistent batch-to-batch quality, which is paramount for meeting stringent regulatory standards in pharmaceutical manufacturing.
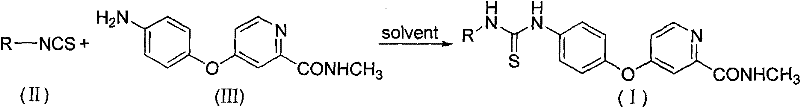
Mechanistic Insights into VEGFR-2 Inhibition by Arylthiourea Derivatives
The core pharmacophore of these compounds centers on the arylthiourea structure, which acts as a potent hydrogen bond donor and acceptor within the ATP-binding pocket of the VEGFR-2 kinase domain. Structural analysis suggests that the sulfur atom in the thiourea linkage provides distinct electronic interactions compared to oxygen in ureas, potentially enhancing binding affinity and selectivity. The molecules are designed to compete with ATP for the active site, thereby blocking downstream signaling pathways responsible for tumor angiogenesis and proliferation. Substituents on the aryl ring, such as halogens or electron-withdrawing groups, further modulate the lipophilicity and steric fit within the enzyme cleft. This precise molecular engineering results in inhibition rates that significantly outperform reference standards in enzymatic assays. Understanding this mechanism is vital for researchers aiming to optimize lead compounds for next-generation anticancer therapies.
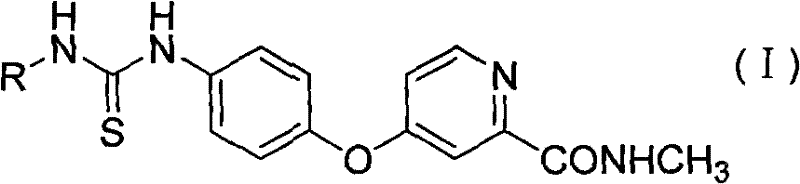
Beyond enzymatic inhibition, the impurity profile of these synthesized intermediates is tightly controlled through the specificity of the condensation reaction. The use of well-defined starting materials minimizes the formation of side products that could complicate downstream purification or introduce toxicological risks. The patent data indicates that the resulting compounds maintain high stability under physiological conditions, ensuring they reach their target site effectively. Rigorous control over the substitution pattern on the nitrogen atoms prevents the formation of unwanted regioisomers. This level of chemical precision translates to a cleaner final API, reducing the burden on quality control laboratories during release testing. For supply chain stakeholders, a predictable and clean synthesis route means fewer production delays and lower costs associated with waste disposal and reprocessing.
How to Synthesize Arylthiourea Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these high-value intermediates with reproducibility. The process begins with the preparation of the key amine precursor, followed by a straightforward coupling step that can be monitored via thin-layer chromatography. Detailed operational parameters regarding solvent selection, molar ratios, and workup procedures are critical for achieving the reported yields. Implementing this route requires attention to moisture control and temperature regulation to maximize efficiency. The standardized nature of the reaction steps facilitates technology transfer from laboratory scale to pilot plant operations. Please refer to the structured guide below for the specific procedural breakdown.
- Preparation of Key Intermediate: Synthesize [4-(4-aminobenzyloxy)(2-pyridyl)]-N-methylformamide via acylation and etherification steps under controlled nitrogen atmosphere.
- Condensation Reaction: Mix the amine intermediate with substituted isothiocyanates in polar aprotic solvents like DMF or DMSO at temperatures ranging from 20°C to 100°C.
- Purification and Isolation: Quench the reaction, extract with organic solvents such as ethyl acetate, and purify the crude product using silica gel column chromatography to achieve pharmaceutical grade purity.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this synthetic route offers substantial strategic benefits for organizations focused on cost reduction and supply chain resilience in pharmaceutical manufacturing. The elimination of hazardous reagents like phosgene simplifies regulatory compliance and reduces the need for specialized containment infrastructure. This inherently safer chemistry lowers insurance premiums and operational overheads associated with handling dangerous materials. Furthermore, the use of commodity solvents and readily available starting materials mitigates the risk of supply disruptions caused by raw material shortages. The high yields reported in the patent examples suggest a material-efficient process that maximizes output per batch. These factors combine to create a more robust and economically viable supply chain for critical oncology intermediates.
- Cost Reduction in Manufacturing: The simplified reaction sequence reduces the number of unit operations required, leading to significant savings in labor and utility costs. By avoiding expensive transition metal catalysts, the process eliminates the need for costly metal scavenging steps and residual metal testing. The high atom economy of the condensation reaction ensures that raw materials are converted efficiently into the desired product, minimizing waste generation. These cumulative efficiencies drive down the overall cost of production, allowing for more competitive pricing in the global market. Ultimately, this economic advantage supports sustainable growth and investment in further R&D initiatives.
- Enhanced Supply Chain Reliability: Sourcing strategies benefit from the use of common chemical feedstocks that are widely available from multiple global suppliers. This diversity in the supply base reduces dependency on single-source vendors and enhances negotiation leverage. The robustness of the chemistry ensures that production schedules can be maintained even under varying operational conditions. Reduced complexity in the manufacturing process also shortens the lead time required for batch completion and release. Consequently, procurement teams can maintain lower inventory levels while ensuring continuous availability of critical intermediates for drug formulation.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard reaction conditions and equipment commonly found in multipurpose chemical plants. The absence of heavy metals and hazardous gases aligns with increasingly strict environmental regulations and corporate sustainability goals. Waste streams are easier to treat and dispose of, reducing the environmental footprint of the manufacturing facility. This compliance advantage facilitates faster regulatory approvals and smoother audits from international health authorities. Scalability ensures that the supply can grow in tandem with clinical demand, supporting the successful commercialization of new therapeutic agents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these arylthiourea compounds. Answers are derived directly from the experimental data and specifications provided in the patent documentation. This section aims to clarify uncertainties regarding safety, scalability, and biological performance. Stakeholders are encouraged to review these insights to inform their sourcing and development strategies. Comprehensive technical support is available for partners seeking deeper engagement with this technology.
Q: How does the toxicity profile of these arylthiourea compounds compare to Sorafenib?
A: According to patent data, these novel arylthiourea derivatives exhibit significantly lower cytotoxicity towards normal human liver cells (LO-2) compared to Sorafenib, while maintaining or exceeding VEGFR-2 inhibition efficacy.
Q: What are the optimal reaction conditions for scaling this synthesis?
A: The process utilizes mild conditions with reaction temperatures between 20°C and 100°C and common solvents like DMF or acetonitrile, facilitating easier scale-up and reduced energy consumption compared to high-pressure alternatives.
Q: Can the substituents on the aryl ring be modified for specific potency?
A: Yes, the general formula allows for diverse substitutions including halogens, trifluoromethyl groups, and esters, enabling medicinal chemists to fine-tune pharmacokinetic properties and binding affinity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylthiourea Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, offering unparalleled expertise in scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications required for oncology intermediates. We understand the critical nature of supply continuity in the pharmaceutical sector and have built our operations to ensure reliability and consistency. Our team of experienced chemists is ready to adapt the patented synthesis routes to meet your specific volume and quality requirements. Partnering with us ensures access to high-quality materials that accelerate your drug development timelines.
We invite you to contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your project needs. Our experts can provide specific COA data and route feasibility assessments to demonstrate how we can optimize your supply chain. Engaging with us early in your development process allows for seamless technology transfer and risk mitigation. Let us support your mission to bring life-saving therapies to patients worldwide with our commitment to excellence and innovation. Reach out today to discuss how we can become your trusted partner in chemical manufacturing.
