Advanced Arylthiourea Synthesis for Next-Generation VEGFR-2 Targeted Oncology Therapeutics
The pharmaceutical landscape for oncology therapeutics is constantly evolving, driven by the urgent need for agents that offer higher efficacy with reduced systemic toxicity. Patent CN101723890B introduces a significant advancement in this field through the disclosure of a novel class of arylthiourea derivatives designed as potent inhibitors of Vascular Endothelial Growth Factor Receptor-2 (VEGFR-2). These compounds represent a strategic evolution from traditional urea-based kinase inhibitors, addressing critical limitations in current treatment protocols. The core innovation lies in the specific structural modification of the thiourea scaffold, which has been demonstrated to retain high inhibitory potency against tumor cell proliferation while exhibiting a markedly improved safety profile regarding normal hepatocyte viability. For research and development teams focused on next-generation anti-angiogenic drugs, understanding the synthetic accessibility and biological potential of these intermediates is paramount for accelerating pipeline development.
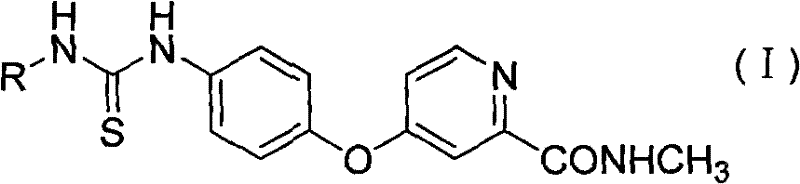
In the context of modern drug discovery, the transition from lead identification to clinical candidate requires robust chemical scaffolds that can be optimized for both potency and pharmacokinetic properties. The arylthiourea motif described in this patent serves as a highly versatile pharmacophore. Unlike earlier generations of kinase inhibitors that often suffered from off-target effects or severe hepatic toxicity, these new derivatives leverage the unique hydrogen-bonding capabilities of the thiourea linkage to achieve selective binding within the ATP pocket of the kinase domain. This structural precision allows for the modulation of biological activity through systematic variation of the aryl substituents, providing medicinal chemists with a powerful toolkit for structure-activity relationship (SAR) studies. The ability to fine-tune electronic and steric properties without compromising the core binding interaction is a distinct advantage in the competitive landscape of targeted cancer therapy development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional approaches to synthesizing kinase inhibitors, particularly those based on urea linkages like Sorafenib, often involve complex multi-step sequences that require harsh reaction conditions or expensive coupling reagents. A significant drawback of many existing VEGFR inhibitors is their narrow therapeutic index, where the dosage required to inhibit tumor growth also induces substantial toxicity in healthy tissues, particularly the liver. From a manufacturing perspective, conventional routes may rely on transition metal catalysts that necessitate rigorous purification steps to meet stringent regulatory limits for residual metals in active pharmaceutical ingredients (APIs). Furthermore, the stability of certain urea linkages under physiological conditions can sometimes be suboptimal, leading to metabolic instability that reduces the effective half-life of the drug in vivo. These factors collectively increase the cost of goods sold (COGS) and extend the timeline for clinical translation, creating bottlenecks for pharmaceutical companies aiming to bring new therapies to market efficiently.
The Novel Approach
The methodology outlined in the patent data presents a streamlined alternative that directly addresses these manufacturing and biological challenges. By utilizing a direct condensation reaction between specific isothiocyanates and a functionalized aminopyridine intermediate, the synthesis bypasses the need for complex activation steps or hazardous reagents. This approach not only simplifies the operational workflow but also enhances the overall atom economy of the process. Biologically, the substitution of the urea oxygen with sulfur in the thiourea linkage appears to confer distinct advantages in terms of receptor binding affinity and selectivity. The resulting compounds demonstrate superior inhibition of VEGFR-2 mediated signaling pathways compared to reference standards, while critically showing reduced cytotoxicity against normal liver cells. This dual benefit of simplified synthesis and enhanced safety margin makes this chemical series an attractive candidate for further development and commercialization in the oncology sector.
Mechanistic Insights into Thiourea-Mediated Kinase Inhibition
The biological efficacy of these arylthiourea derivatives stems from their precise interaction with the intracellular tyrosine kinase domain of VEGFR-2. Structurally, the kinase domain contains a critical ATP-binding cleft located between two protein lobes, which serves as the primary target for small molecule inhibitors. The thiourea moiety acts as a key hydrogen bond donor and acceptor, anchoring the molecule within this cleft and effectively competing with ATP for binding sites. This competitive inhibition prevents the phosphorylation of downstream substrates, thereby halting the signal transduction cascade that drives endothelial cell proliferation and new blood vessel formation essential for tumor growth. The flexibility of the linker region connecting the thiourea core to the heterocyclic pyridine ring allows the molecule to adapt to the conformational nuances of the binding pocket, maximizing van der Waals contacts and enhancing binding affinity. This mechanistic understanding is crucial for R&D directors aiming to predict the in vivo behavior of these compounds and design subsequent generations of analogs with even greater potency.
From a purity and impurity control perspective, the robustness of the thiourea formation reaction offers significant advantages. The direct condensation of amines with isothiocyanates is generally a clean reaction with few side products, primarily limited to unreacted starting materials which are easily removed during workup. This contrasts with coupling reactions that might generate difficult-to-remove urea byproducts or racemization issues. The stability of the thiourea bond under the reaction conditions described (20-100°C in polar solvents) ensures that the final product maintains its structural integrity without degradation. For quality control teams, this translates to a simpler impurity profile, facilitating easier validation of analytical methods and ensuring consistent batch-to-batch quality. The ability to produce high-purity intermediates with minimal chromatographic purification steps is a key factor in reducing production costs and ensuring supply chain reliability for downstream API manufacturing.
How to Synthesize Arylthiourea VEGFR Inhibitors Efficiently
The synthesis of these high-value pharmaceutical intermediates relies on a convergent strategy that couples two readily available building blocks. The process begins with the preparation of the key amine component, [4-(4-aminobenzyloxy)(2-pyridyl)]-N-methylformamide, which serves as the constant scaffold across the compound library. This amine is then reacted with a diverse array of aryl isothiocyanates, allowing for rapid exploration of chemical space. The reaction is typically conducted in polar aprotic solvents such as DMF or DMSO, which facilitate the dissolution of both reactants and promote the nucleophilic attack of the amine on the electrophilic carbon of the isothiocyanate group. The mild thermal requirements, ranging from ambient temperature to moderate heating, make this process energy-efficient and safe to operate on a large scale. Detailed standardized synthesis steps for optimizing yield and purity are provided in the guide below.
- Preparation of the key amine intermediate [4-(4-aminobenzyloxy)(2-pyridyl)]-N-methylformamide via nucleophilic substitution.
- Selection of appropriate aryl isothiocyanate derivatives based on desired substituent patterns for structure-activity optimization.
- Direct condensation of the amine and isothiocyanate in polar aprotic solvents like DMF or DMSO at temperatures ranging from 20°C to 100°C.
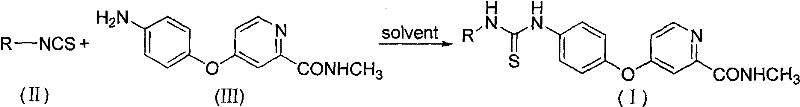
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthetic route offers tangible benefits in terms of cost structure and operational efficiency. The reliance on commodity chemicals such as substituted anilines (precursors to isothiocyanates) and common solvents like ethanol and ethyl acetate ensures a stable and diversified supply base. Unlike processes dependent on scarce noble metal catalysts or specialized reagents, this method mitigates the risk of supply disruptions and price volatility associated with exotic raw materials. The simplicity of the reaction workup, which often involves standard extraction and crystallization techniques rather than complex chromatography, significantly reduces processing time and solvent consumption. These factors collectively contribute to a more sustainable and cost-effective manufacturing model, allowing companies to allocate resources more effectively towards clinical development and market expansion strategies.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of straightforward condensation chemistry drastically simplify the production process. By avoiding the need for specialized equipment required for high-pressure or cryogenic reactions, capital expenditure is minimized. Furthermore, the high conversion rates observed in these reactions reduce the volume of waste generated, lowering disposal costs and environmental compliance burdens. The ability to perform the reaction at or near room temperature for many derivatives also results in substantial energy savings compared to processes requiring prolonged reflux or extreme cooling, directly impacting the bottom line through reduced utility costs.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, specifically the various aryl isothiocyanates and the pyridine-based amine, are derived from widely available petrochemical feedstocks. This broad availability ensures that procurement teams can source materials from multiple vendors, reducing dependency on single-source suppliers and enhancing negotiation leverage. The robustness of the chemical transformation means that minor variations in raw material quality can often be accommodated without compromising the final product specification, providing a buffer against supply chain fluctuations. This resilience is critical for maintaining continuous production schedules and meeting the demanding delivery timelines required by global pharmaceutical partners.
- Scalability and Environmental Compliance: The process is inherently scalable, having been demonstrated effectively from gram to multi-kilogram scales without loss of efficiency. The solvents used are well-understood in industrial settings, with established recovery and recycling protocols that align with green chemistry principles. The absence of heavy metal residues simplifies the regulatory filing process, as there is no need for extensive testing and validation of metal clearance steps. This streamlined path to compliance accelerates time-to-market for new drug applications. Additionally, the reduced generation of hazardous byproducts minimizes the environmental footprint of the manufacturing facility, supporting corporate sustainability goals and improving community relations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these arylthiourea intermediates. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this technology into their existing portfolios. The answers are derived directly from the experimental data and technical specifications provided in the patent literature, ensuring accuracy and relevance for decision-making purposes. Whether you are concerned with regulatory hurdles, synthesis optimization, or biological performance, these insights provide a solid foundation for further discussion with our technical team.
Q: What is the primary advantage of these arylthiourea compounds over Sorafenib?
A: While maintaining potent VEGFR-2 inhibitory activity, these novel arylthiourea derivatives demonstrate significantly lower cytotoxicity towards normal human liver cells (LO-2), offering an improved safety profile for clinical development.
Q: Are the synthesis conditions suitable for large-scale manufacturing?
A: Yes, the synthesis utilizes mild reaction conditions (20-100°C) and common industrial solvents such as DMF, ethanol, and ethyl acetate, avoiding expensive transition metal catalysts and facilitating straightforward scale-up.
Q: What is the mechanism of action for these intermediates?
A: These compounds function as ATP-competitive inhibitors that bind to the intracellular tyrosine kinase domain of VEGFR-2, effectively blocking signal transduction pathways responsible for tumor angiogenesis and cell proliferation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arylthiourea Supplier
As the demand for targeted oncology therapies continues to surge, the need for high-quality, reliably sourced intermediates has never been greater. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging decades of expertise in fine chemical synthesis to deliver superior pharmaceutical intermediates. Our state-of-the-art facilities are equipped to handle the commercial scale-up of complex organic syntheses, ranging from 100 kgs to 100 MT/annual commercial production. We pride ourselves on our stringent purity specifications and rigorous QC labs, which ensure that every batch of arylthiourea derivative meets the exacting standards required for GMP API manufacturing. Our commitment to quality and consistency makes us a trusted partner for global pharmaceutical companies seeking to secure their supply chains for critical cancer treatments.
We invite you to explore the potential of these novel VEGFR inhibitors for your drug development programs. Our technical procurement team is ready to assist you with a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We encourage you to contact us to request specific COA data for our available inventory and to discuss route feasibility assessments for custom analogs. By collaborating with NINGBO INNO PHARMCHEM, you gain access to not just a supplier, but a strategic partner dedicated to accelerating your path from discovery to clinical success through innovative chemistry and reliable execution.
