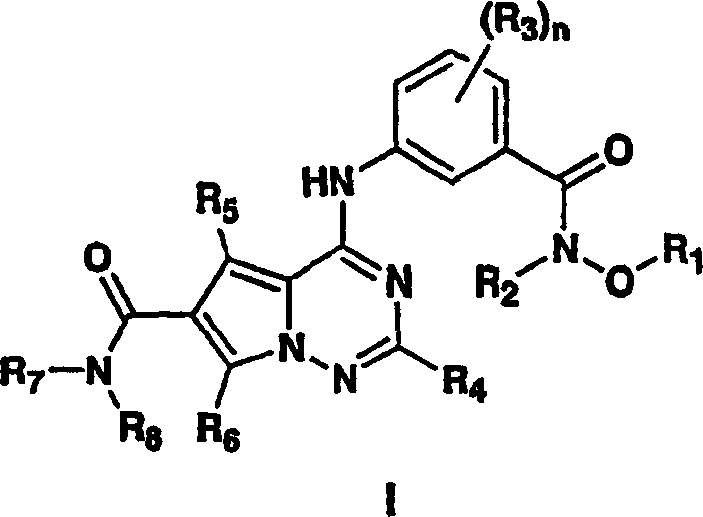
Advanced Manufacturing of Pyrrolotriazine Kinase Inhibitor Intermediates for Global Pharma Applications
The pharmaceutical industry's relentless pursuit of potent anti-inflammatory agents has placed pyrrolotriazine kinase inhibitors at the forefront of therapeutic research, particularly for treating diseases mediated by p38α and p38β enzymes. Patent CN1771234A discloses a groundbreaking improvement in the preparation of these critical pharmaceutical intermediates, addressing long-standing challenges in yield, safety, and scalability. Unlike previous methodologies that relied on hazardous reagents and complex purification techniques, this invention introduces a novel pyrrolotriazine ring-forming procedure that streamlines the synthesis of the bicyclic heterocycle core. By utilizing commercially available starting materials such as 6-alkyl-1,2,4-triazin-5-ones and 3-substituted pyruvates, the process significantly reduces reliance on toxic reactants while maintaining high product integrity. This technological leap not only enhances the purity profile of the resulting kinase inhibitors but also offers a robust pathway for cost reduction in pharmaceutical intermediate manufacturing. For R&D directors and procurement specialists alike, understanding the mechanistic nuances and commercial implications of this patent is essential for securing a competitive edge in the development of next-generation anti-inflammatory therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods for synthesizing pyrrolotriazine cores, such as those described in WO00/71129, were fraught with significant operational and safety drawbacks that hindered efficient commercial scale-up. These traditional routes typically involved the reaction of Michael acceptors with tosylmethyl isocyanide (TosMIC), a reagent known for its instability and handling difficulties. Furthermore, the subsequent steps often required the use of hazardous aminating agents like diphenylphosphorylhydroxylamine, posing severe risks to personnel and requiring specialized containment infrastructure. Perhaps most critically for supply chain managers, these legacy processes frequently necessitated expensive chromatographic separation to isolate the desired product from complex reaction mixtures, drastically inflating production costs and extending lead times. The generation of toxic byproducts, such as cyanides in alternative acetaldehyde condensation methods, further complicated waste management and environmental compliance, making these routes unsustainable for modern high-purity API intermediate production.
The Novel Approach
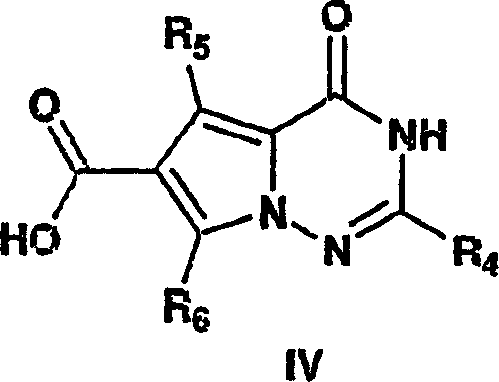
The methodology outlined in CN1771234A represents a paradigm shift by replacing these perilous steps with a direct and efficient condensation reaction. The core innovation lies in reacting a 6-alkyl-1,2,4-triazin-5-one directly with a 3-substituted pyruvate, such as 3-bromopyruvate, to construct the pyrrolotriazine ring system in a single, high-yielding step. This approach completely bypasses the need for TosMIC and dangerous aminating reagents, thereby simplifying the safety profile of the manufacturing process. Moreover, the resulting intermediates can often be purified through straightforward crystallization and filtration rather than chromatography, which is a game-changer for commercial scale-up of complex pharmaceutical intermediates. By leveraging common solvents like water and ethanol, the process aligns with green chemistry principles, reducing the environmental footprint and facilitating easier regulatory approval for the final drug substance.
Mechanistic Insights into Pyrrolotriazine Ring Formation
The heart of this synthetic breakthrough is the nucleophilic cyclization that forms the fused bicyclic system. In the initial step, the nitrogen atom of the 1,2,4-triazin-5-one acts as a nucleophile, attacking the electrophilic carbon of the 3-substituted pyruvate. The presence of a suitable leaving group (X) at the 3-position of the pyruvate, such as a halogen or sulfonate, is critical for driving this transformation forward. Following the initial addition, an intramolecular cyclization occurs, facilitated by the elimination of the leaving group, to close the pyrrole ring onto the triazine scaffold. This mechanism is highly advantageous because it avoids the formation of unstable intermediates common in older routes. The reaction conditions are mild yet effective, typically proceeding in aqueous or alcoholic media at elevated temperatures, which ensures that the reaction kinetics favor the desired product over potential side reactions.
Impurity control is another area where this mechanism excels, providing R&D teams with a cleaner crude product profile. In conventional syntheses, side reactions involving the aminating agent or the Michael acceptor often led to difficult-to-remove impurities that required rigorous chromatographic cleanup. In contrast, the direct condensation described here generates fewer byproducts, and those that do form are often structurally distinct enough to be removed via simple recrystallization. The use of specific leaving groups like bromine allows for precise control over the reaction rate and selectivity, minimizing the formation of regioisomers. This inherent purity is vital for downstream processing, as it reduces the burden on purification units and ensures that the final high-purity pharmaceutical intermediates meet stringent quality specifications without excessive reprocessing.
How to Synthesize Pyrrolotriazine Kinase Inhibitors Efficiently
The synthesis of these potent kinase inhibitors follows a logical, five-step sequence designed for maximum efficiency and yield. The process begins with the formation of the core carboxylic acid, followed by esterification to protect the functionality and activate the molecule for subsequent transformations. Chlorination of the triazine ring then serves as a crucial activation step, enabling the introduction of diverse aniline substituents. The final amidation step allows for the modular attachment of various amine groups, facilitating the rapid exploration of structure-activity relationships. Detailed standard operating procedures for each stage, including specific molar ratios, temperature controls, and workup protocols, are essential for replicating the high yields reported in the patent literature. For a comprehensive guide on executing this synthesis in a GMP environment, please refer to the standardized protocol below.
- React 6-alkyl-1,2,4-triazin-5-one with 3-substituted pyruvate (e.g., 3-bromopyruvate) to form the pyrrolotriazine carboxylic acid core.
- Esterify the carboxylic acid intermediate using an alcohol and a coupling agent like HCl to generate the ester derivative.
- Chlorinate the ester using a chlorinating agent such as POCl3 in the presence of a base to activate the triazine ring.
- Couple the chlorinated intermediate with a substituted aniline (e.g., N-methoxy-3-amino-4-methylbenzamide) to form the amine-substituted scaffold.
- Perform a final amidation reaction with a primary or secondary amine to yield the target pyrrolotriazine carboxamide kinase inhibitor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this novel synthetic route offers tangible benefits that extend far beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By utilizing commodity chemicals like ethyl triazinone and bromopyruvic acid, manufacturers can mitigate the risk of supply disruptions associated with exotic or custom-synthesized reagents. This shift to widely available starting materials ensures a more stable and predictable sourcing strategy, which is critical for maintaining continuous production schedules in a volatile global market. Furthermore, the elimination of chromatography steps translates directly into reduced processing time and lower solvent consumption, contributing to significant operational cost savings.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the removal of expensive purification technologies. Traditional methods relying on column chromatography are not only costly in terms of silica gel and solvents but are also notoriously difficult to scale, often becoming bottlenecks in production. By replacing this with crystallization, the process becomes inherently more scalable and cost-effective. Additionally, the avoidance of hazardous reagents like TosMIC reduces the costs associated with special handling, storage, and hazardous waste disposal. These cumulative efficiencies result in a leaner manufacturing process that delivers substantial cost savings without compromising on the quality of the final pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Reliability is the cornerstone of a robust supply chain, and this synthesis method strengthens it by decoupling production from scarce reagents. The reliance on bulk chemicals means that suppliers can source materials from multiple vendors, reducing dependency on single-source providers. The simplified workflow also shortens the overall cycle time from raw material intake to finished goods, allowing for faster response to market demand fluctuations. This agility is particularly valuable in the fast-paced pharmaceutical sector, where reducing lead time for high-purity intermediates can accelerate drug development timelines and bring life-saving therapies to patients sooner.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges, but this route is designed with scalability in mind. The use of water and ethanol as primary solvents aligns with modern environmental, health, and safety (EHS) standards, minimizing the generation of volatile organic compounds (VOCs). The ability to perform key steps in aqueous media reduces the fire hazard potential and simplifies wastewater treatment protocols. Consequently, facilities can achieve higher throughput with a smaller environmental footprint, ensuring compliance with increasingly stringent global regulations while maintaining the flexibility to ramp up production volumes as clinical trials progress to commercial stages.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and background analysis provided in the patent documentation, offering clarity on how this technology compares to existing standards. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own pipeline projects.
Q: How does this new process improve safety compared to conventional methods?
A: Conventional methods often rely on hazardous reagents like TosMIC or generate toxic cyanide byproducts. This patented process utilizes commercially available 3-halopyruvates and avoids dangerous aminating agents, significantly reducing workplace hazards and waste disposal complexity.
Q: What are the scalability advantages of this synthetic route?
A: The process eliminates the need for expensive and difficult chromatographic purification steps. Instead, it relies on crystallization and filtration, which are far more amenable to large-scale commercial production (100 kgs to 100 MT), ensuring consistent supply chain reliability.
Q: Can this method be adapted for different kinase inhibitor analogs?
A: Yes, the modular nature of the synthesis, particularly the final amidation step, allows for the introduction of diverse amine substituents. This flexibility supports the rapid generation of structure-activity relationship (SAR) libraries for drug discovery programs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolotriazine Kinase Inhibitor Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust and scalable synthetic routes in the development of advanced therapeutics. Our team of expert chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. We are committed to delivering stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of pyrrolotriazine intermediate meets the highest industry standards. Our state-of-the-art facilities are equipped to handle the specific solvent systems and reaction conditions required by this innovative process, providing you with a secure and reliable supply partner.
We invite you to collaborate with us to leverage these technological advancements for your drug development programs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and accelerate your path to market.
