Scalable Synthesis of Pyrrolotriazine Intermediates for Next-Generation Kinase Inhibitors
Introduction to Advanced Pyrrolotriazine Synthesis
The development of potent kinase inhibitors, particularly those targeting the c-Met receptor, represents a critical frontier in oncology therapeutics. Patent CN1993130A discloses a sophisticated and highly efficient methodology for preparing pyrrolotriazine intermediates, which serve as the foundational scaffolds for these life-saving medications. This intellectual property outlines a robust chemical pathway that addresses many of the historical challenges associated with constructing fused heterocyclic systems. By leveraging specific organometallic additions and controlled oxidative rearrangements, the disclosed process enables the production of high-purity intermediates essential for drug discovery and development. For pharmaceutical companies seeking a reliable pharmaceutical intermediate supplier, understanding the nuances of this patented technology is vital for securing a stable supply chain for next-generation cancer therapies.
The significance of this technology extends beyond mere academic interest; it provides a tangible route to compounds that modulate signal transduction pathways involved in tumor invasion and metastasis. The patent details multiple embodiments, including the preparation of various substituted pyrrolotriazines that can be further functionalized into active pharmaceutical ingredients. The strategic design of these synthetic routes ensures that critical functional groups are introduced with high regioselectivity, minimizing the formation of unwanted byproducts. This level of precision is paramount when manufacturing compounds intended for human consumption, where impurity profiles are strictly regulated. Consequently, adopting such validated processes can significantly de-risk the development timeline for new kinase inhibitor programs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional methods for synthesizing fused heterocyclic kinase inhibitors often rely on convergent strategies that can suffer from poor atom economy and difficult purification steps. Conventional routes frequently involve harsh reaction conditions that may degrade sensitive functional groups or lead to the formation of complex mixtures of regioisomers that are challenging to separate. For instance, direct halogenation or nitration of pre-formed heterocyclic cores can result in unpredictable substitution patterns, necessitating extensive chromatographic purification which is not feasible on a large commercial scale. Furthermore, many legacy processes utilize expensive transition metal catalysts that require rigorous removal to meet residual metal specifications, adding significant cost and complexity to the manufacturing workflow. These inefficiencies can lead to prolonged lead times and increased costs in API manufacturing, creating bottlenecks for procurement teams.
The Novel Approach
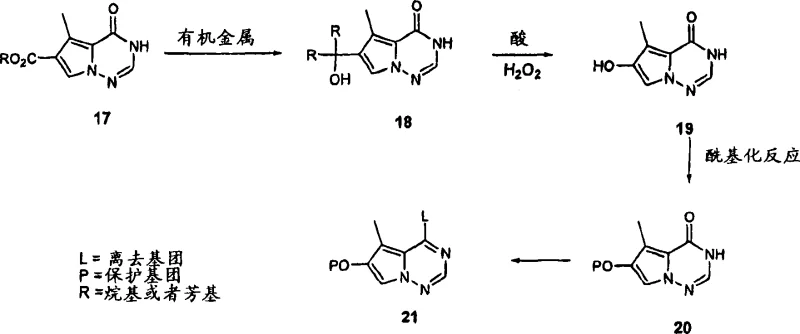
In contrast, the novel approach detailed in CN1993130A utilizes a linear yet highly controlled sequence starting from readily available carboxylate precursors. The core innovation lies in the use of a Grignard reagent to install alkyl groups followed by a specific acid-catalyzed oxidative rearrangement. This strategy bypasses the need for difficult direct functionalization of the aromatic core by building the necessary substitution pattern through skeletal rearrangement. The process employs common reagents such as methylmagnesium chloride and methanesulfonic acid, which are cost-effective and widely available in the global chemical market. By shifting the complexity to well-understood transformation steps like nucleophilic addition and esterification, the novel approach simplifies the overall synthetic tree. This simplification translates directly into cost reduction in API manufacturing by reducing the number of unit operations and improving overall yield consistency.
Mechanistic Insights into Oxidative Rearrangement and Functionalization
The heart of this synthetic strategy is the transformation of the tertiary alcohol intermediate into the hydroxypyrrolotriazine core via oxidative rearrangement. Mechanistically, this involves the activation of the hydroperoxide species by a strong acid catalyst, such as methanesulfonic acid, which facilitates the migration of the alkyl group and the subsequent restoration of aromaticity. This step is critical because it establishes the correct oxidation state and substitution pattern required for biological activity. The reaction conditions are meticulously controlled, typically maintaining temperatures between -5°C and -0.7°C to prevent over-oxidation or decomposition of the sensitive peroxide intermediates. The quenching of the reaction with reducing agents like sodium bisulfite ensures the safe removal of excess oxidants, thereby enhancing process safety and environmental compliance. Understanding this mechanism allows chemists to fine-tune reaction parameters to maximize yield and minimize impurity formation.
Following the formation of the hydroxyl core, the process employs a protection-chlorination sequence to generate a versatile electrophilic intermediate. The hydroxyl group is first protected as a pivalate ester using pivaloyl chloride, which serves to stabilize the molecule during subsequent harsh chlorination steps. The introduction of the chlorine atom at the 4-position using phosphorus oxychloride (POCl3) creates a highly reactive handle for nucleophilic aromatic substitution. This chloro-intermediate is pivotal as it allows for the attachment of various aniline or phenol derivatives in later stages, enabling the rapid generation of diverse analog libraries for structure-activity relationship (SAR) studies. The use of amine bases like DIEA during chlorination helps to scavenge generated acid and drive the reaction to completion, ensuring high conversion rates. This mechanistic understanding underscores the robustness of the pathway for producing high-purity kinase inhibitor intermediates.
How to Synthesize Pyrrolotriazine Intermediates Efficiently
The synthesis of these valuable intermediates follows a logical progression that balances reactivity with selectivity. The process begins with the preparation of the tertiary alcohol via Grignard addition, followed by the critical oxidative rearrangement to form the heterocyclic core. Subsequent steps involve protection of the phenolic hydroxyl group and activation of the ring system through chlorination. Each step has been optimized in the patent examples to demonstrate feasibility on a meaningful scale, providing a clear blueprint for process chemists. The detailed experimental procedures offer specific guidance on solvent choices, temperature profiles, and workup techniques that are essential for reproducibility. For those looking to implement this chemistry, the detailed standardized synthesis steps see the guide below provide the necessary operational framework.
- Perform nucleophilic addition of methylmagnesium chloride to ethyl 5-methyl-4-oxo-3,4-dihydro-pyrrolo[2,1-f][1,2,4]triazine-6-carboxylate in THF at controlled temperatures below 35°C.
- Execute oxidative rearrangement of the resulting tertiary alcohol using methanesulfonic acid and hydrogen peroxide at sub-zero temperatures to form the hydroxypyrrolotriazine core.
- Protect the hydroxyl group via acylation with pivaloyl chloride followed by chlorination using phosphorus oxychloride to yield the reactive chloro-intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the processes described in this patent offer substantial advantages for supply chain stability and cost management. The reliance on commodity chemicals rather than exotic reagents means that raw material sourcing is less susceptible to market volatility. The ability to perform reactions in common solvents like THF and ethyl acetate simplifies solvent recovery and recycling efforts, contributing to a more sustainable manufacturing footprint. Moreover, the isolation of intermediates as crystalline solids, as demonstrated in the examples, facilitates easy handling, storage, and transportation, reducing the risk of degradation during logistics. These factors collectively enhance supply chain reliability, ensuring that critical drug substances can be manufactured without interruption. For procurement managers, this translates to a more predictable supply of high-quality materials.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of straightforward crystallization for purification significantly lower the cost of goods sold. By avoiding complex chromatographic separations, the process reduces solvent consumption and waste disposal costs, leading to substantial cost savings. The high yields reported in the kilogram-scale examples further amplify these economic benefits by maximizing the output from each batch of raw materials. Additionally, the telescoping of certain steps or the use of crude intermediates where purity allows can streamline the workflow, reducing labor and equipment time. These efficiencies make the production of these complex heterocycles economically viable for large-scale commercial applications.
- Enhanced Supply Chain Reliability: The synthetic route utilizes starting materials that are commercially available from multiple global suppliers, mitigating the risk of single-source dependency. The robustness of the reaction conditions, which tolerate minor variations in temperature and stoichiometry without significant loss of quality, ensures consistent production output. This reliability is crucial for maintaining continuous manufacturing schedules and meeting the demanding delivery timelines of the pharmaceutical industry. Furthermore, the stability of the protected intermediates allows for inventory buffering, providing a safety stock that can absorb fluctuations in demand. This resilience strengthens the overall supply chain against external disruptions.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of the process through examples involving multi-kilogram quantities, proving its readiness for industrial adoption. The waste streams generated are primarily aqueous salts and organic solvents that can be treated using standard effluent management protocols, ensuring compliance with environmental regulations. The avoidance of heavy metals simplifies the regulatory filing process by reducing the burden of elemental impurity testing. As the industry moves towards greener chemistry, this process aligns well with sustainability goals by optimizing atom economy and reducing the environmental impact of pharmaceutical production. This makes it an attractive option for companies committed to responsible manufacturing practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these pyrrolotriazine intermediates. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. They are intended to clarify the capabilities of the process and its suitability for various drug development stages. Understanding these details helps stakeholders make informed decisions about integrating this technology into their pipelines. The responses cover aspects of purity, scalability, and chemical versatility.
Q: What is the primary advantage of the oxidative rearrangement step in this synthesis?
A: The oxidative rearrangement allows for the precise migration of substituents to form the specific hydroxypyrrolotriazine core required for c-Met inhibition, avoiding complex isomer separation.
Q: Can this process be scaled for commercial API production?
A: Yes, the patent explicitly demonstrates kilogram-scale synthesis (e.g., 1.9 kg starting material), indicating robust scalability for industrial manufacturing.
Q: What purity levels are achievable with this method?
A: The described crystallization and workup procedures consistently yield intermediates with purity ranging from 96% to 99%, suitable for downstream pharmaceutical processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pyrrolotriazine Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis, possessing the technical expertise to translate complex patent methodologies into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can grow seamlessly from clinical trials to market launch. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of pyrrolotriazine intermediate meets the highest quality standards required for oncology drug development. Our commitment to excellence ensures that you receive materials that are ready for the next stage of your synthesis without the need for additional remediation.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how we can support your supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall manufacturing expenses. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to deliver high-purity kinase inhibitor intermediates reliably. Partner with us to secure a competitive advantage in the rapidly evolving landscape of cancer therapeutics.
