Advanced Non-Ether Reduction Technology for High-Purity Thiazolidinedione API Intermediates
The pharmaceutical industry is constantly seeking robust, scalable, and economically viable pathways for the production of critical antihyperglycemic agents, particularly within the thiazolidinedione class. Patent CN1875018A introduces a transformative methodology for the preparation of compounds such as pioglitazone, rosiglitazone, and troglitazone by addressing the limitations of traditional reduction techniques. This innovation centers on a novel reduction step that converts thiazolidinedione precursors possessing an exocyclic double bond at the 5-position into their active saturated forms. Unlike conventional approaches that rely heavily on catalytic hydrogenation under high pressure or the use of ecologically questionable reagents, this disclosed process utilizes a dithionite source within a specifically engineered non-ether solvent medium. For R&D directors and process chemists, this represents a significant leap forward in managing impurity profiles and reaction safety, while simultaneously offering procurement teams a route that relies on commercially abundant and cost-effective reagents. The strategic shift away from high-pressure hydrogenation not only mitigates operational risks but also streamlines the regulatory pathway by avoiding heavy metal catalysts often associated with older methodologies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of key antihyperglycemic thiazolidinediones has been plagued by significant technical and economic hurdles inherent to catalytic hydrogenation processes. Traditional methods frequently necessitate the use of supported catalysts, such as those involving cobalt ions or precious metals, which introduce complex downstream purification challenges to meet stringent residual metal specifications. Furthermore, these reactions typically require high-pressure hydrogen gas, mandating the use of specialized, expensive reaction equipment and rigorous safety protocols that inflate capital expenditure and operational overhead. The reliance on ether solvents in some prior art, such as the processes disclosed in Japanese Patent Application 9-213107, presents additional ecological and safety liabilities, as ethers are often prone to peroxide formation and possess unfavorable environmental profiles. From a supply chain perspective, the dependency on high-pressure infrastructure creates bottlenecks in manufacturing capacity, limiting the ability to rapidly scale production to meet global demand for diabetes medications without substantial investment in hazardous operation facilities.
The Novel Approach
The methodology outlined in CN1875018A circumvents these industrial bottlenecks by employing a chemical reduction strategy using dithionite sources, such as sodium dithionite, in a non-ether solvent environment. This approach operates effectively at ambient atmospheric pressure, thereby eliminating the need for high-pressure hydrogenation reactors and the associated safety risks. A critical component of this innovation is the selection of the solvent medium; the patent explicitly identifies aqueous N,N-dimethylformamide (DMF) as a preferred system that outperforms traditional ether-based mixtures. By avoiding ether solvents, the process achieves superior solubility characteristics for the precursors, preventing the unwanted precipitation of unreacted starting materials that often leads to irreproducible results in other systems. This technological pivot allows for a simpler, more cost-effective workflow that is inherently safer and more attractive from both an industrial and ecological standpoint, directly addressing the pain points of modern pharmaceutical manufacturing.
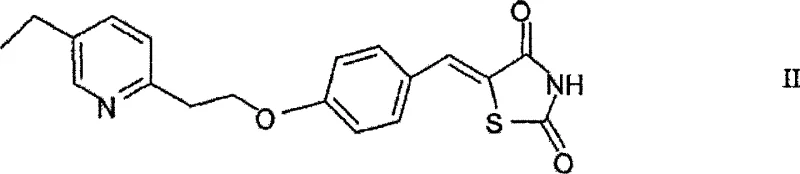
Mechanistic Insights into Dithionite-Mediated Exocyclic Double Bond Reduction
The core chemical transformation in this patent involves the selective reduction of the exocyclic carbon-carbon double bond located at the 5-position of the thiazolidinedione ring system. While the inventors do not wish to be bound by a specific theory, the mechanism is understood to involve the dithionite ion acting as a potent reducing agent in the presence of a base. The reaction is typically conducted by preparing a solution or suspension of the thiazolidinedione precursor, such as 5-(4-(2-(5-ethyl-2-pyridyl)ethoxy)phenyl)methine-2,4-thiazolidinedione, in a solvent medium containing a base like sodium carbonate or potassium carbonate. Upon heating this mixture to temperatures ranging from 40°C to 100°C, the addition of the dithionite source initiates the reduction. The presence of the base is crucial for maintaining the appropriate pH and facilitating the electron transfer required to hydrogenate the double bond without affecting other sensitive functional groups within the complex molecular architecture. This selectivity is paramount for ensuring that the final API intermediate retains its structural integrity and biological activity potential.
Impurity control is a defining feature of this mechanistic pathway, particularly regarding the minimization of unreacted precursor. In many solvent systems, the poor solubility of the unsaturated precursor leads to its precipitation, making it difficult to drive the reaction to completion and resulting in high levels of residual starting material in the final product. However, the use of the preferred DMF and water solvent mixture fundamentally alters the solubility dynamics, allowing the unsaturated precursors to remain in solution and react fully. Experimental data within the patent demonstrates that this specific solvent combination enables the unsaturated precursors to be present in the final product in amounts generally below 0.1% by weight. This level of control is practically impossible to achieve economically with known purification methods in other solvent systems, highlighting the mechanistic advantage of the aqueous DMF system in driving the equilibrium towards the desired saturated product and ensuring a high-purity profile suitable for pharmaceutical applications.
How to Synthesize Pioglitazone Free Base Efficiently
The synthesis of pioglitazone free base via this patented route offers a streamlined protocol that balances reaction efficiency with operational simplicity. The process begins with the preparation of a reaction mixture containing the specific thiazolidinedione precursor, a suitable base such as potassium carbonate, and the optimized non-ether solvent system. The patent details precise molar equivalents and volume ratios, emphasizing the importance of maintaining the reaction temperature between 60°C and 80°C for optimal kinetics. Following the addition of the sodium dithionite source, the mixture is maintained at this elevated temperature for a defined period, typically 1 to 3 hours, to ensure complete conversion. The subsequent workup involves cooling the reaction mixture to induce crystallization, followed by filtration and drying to isolate the free base. For a comprehensive, step-by-step technical guide including exact quantities and specific processing parameters, please refer to the standardized synthesis instructions provided below.
- Prepare a solution or suspension of the thiazolidinedione precursor in a non-ether solvent medium containing a base, such as aqueous DMF with potassium carbonate.
- Heat the mixture to an elevated temperature between 40°C and 100°C, preferably maintaining it around 60°C to 80°C for optimal reaction kinetics.
- Mix the heated solution with a dithionite source, such as sodium dithionite, and maintain the temperature for 1 to 10 hours to ensure complete reduction of the exocyclic double bond.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this dithionite-based reduction technology translates into tangible strategic benefits that extend beyond mere chemical yield. The elimination of high-pressure hydrogenation equipment significantly reduces capital expenditure requirements for manufacturing facilities, allowing for production in standard glass-lined or stainless steel reactors that are more readily available and easier to maintain. Furthermore, the substitution of expensive and potentially toxic transition metal catalysts with commodity chemicals like sodium dithionite drastically simplifies the raw material sourcing landscape. Sodium dithionite is a widely produced industrial chemical with a stable supply chain, reducing the risk of vendor lock-in or price volatility often associated with specialized catalytic reagents. This shift not only lowers the direct cost of goods sold but also enhances the overall resilience of the supply chain by relying on robust, multi-source availability for key reagents.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the removal of costly infrastructure and reagents. By operating at ambient atmospheric pressure, manufacturers avoid the significant energy costs and maintenance fees associated with high-pressure hydrogen systems. Additionally, the avoidance of noble metal or cobalt catalysts eliminates the need for expensive metal scavenging steps and the associated waste disposal costs. The use of aqueous DMF, while requiring careful solvent recovery, leverages common industrial solvents that are easier to recycle compared to complex ether mixtures. These factors combine to create a manufacturing process with a significantly lower operational cost baseline, allowing for more competitive pricing structures in the global market for antihyperglycemic intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on universally available reagents rather than specialized catalysts that may have long lead times. Sodium dithionite and common bases like potassium carbonate are produced at massive scales globally, ensuring that production schedules are not held hostage by the availability of niche chemicals. The simplified equipment requirements mean that contract manufacturing organizations (CMOs) can easily adopt this process without needing to retrofit facilities with high-pressure capabilities, thereby expanding the pool of qualified suppliers. This flexibility is crucial for mitigating supply risks, as it allows pharmaceutical companies to diversify their manufacturing base across multiple geographic regions, ensuring a steady flow of critical intermediates even during periods of regional disruption or logistical constraints.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this method offers a cleaner profile that aligns with increasingly stringent global regulations. The absence of heavy metal catalysts reduces the burden on wastewater treatment systems and minimizes the generation of hazardous solid waste, facilitating easier compliance with environmental protection standards. The process is inherently scalable because it does not rely on mass transfer limitations often encountered in gas-liquid hydrogenation reactions at large scales. Instead, the liquid-phase reduction proceeds uniformly, making the transition from pilot plant to commercial tonnage production more predictable and less prone to the engineering challenges that typically plague scale-up efforts. This ease of scale-up ensures that supply can be rapidly ramped up to meet market demand spikes without the prolonged validation cycles associated with high-pressure processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dithionite reduction technology for thiazolidinedione synthesis. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding the nuances of solvent selection, reaction conditions, and purification outcomes is essential for evaluating the feasibility of integrating this method into existing production workflows. The answers below clarify the specific advantages of the non-ether system and the quality benchmarks achievable through this innovative approach.
Q: Why is the non-ether solvent system preferred over traditional ether solvents for this reduction?
A: The patent highlights that non-ether solvents, specifically aqueous DMF mixtures, prevent the precipitation of unreacted precursors which often occurs in other solvent systems. This ensures higher reproducibility and allows the unsaturated precursors to remain below 0.1% by weight in the final product, significantly simplifying purification.
Q: What are the safety advantages of using sodium dithionite compared to catalytic hydrogenation?
A: Traditional methods often require high-pressure hydrogen gas and specialized reactors with expensive safety measures. The dithionite-based method operates at ambient atmospheric pressure, eliminating the risks associated with high-pressure hydrogen handling and reducing the need for specialized high-pressure equipment.
Q: Can this method achieve pharmaceutical-grade purity for Pioglitazone intermediates?
A: Yes, the data indicates that when using the preferred DMF/water solvent system, the resulting pioglitazone free base can achieve HPLC purity exceeding 95%, and subsequent salt formation can yield products with greater than 98% to 99.9% purity, meeting stringent pharmaceutical standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pioglitazone Intermediate Supplier
The technological advancements detailed in patent CN1875018A underscore the evolving landscape of pharmaceutical intermediate manufacturing, where safety, purity, and cost-efficiency are paramount. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, leveraging deep expertise in process chemistry to deliver high-value solutions for the global pharmaceutical industry. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive consistent, high-quality supply regardless of volume requirements. Our commitment to excellence is backed by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards, guaranteeing that our thiazolidinedione intermediates meet the exacting needs of API synthesis.
We invite forward-thinking pharmaceutical companies and procurement leaders to collaborate with us to optimize their supply chains for antihyperglycemic drugs. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities and commitment to process innovation can drive value and security for your critical drug development programs, ensuring a reliable partnership for the long term.
