Advanced Synthesis of Alkoxyiminoacetamide Derivatives for Scalable Agrochemical Production
The global demand for high-efficacy agricultural fungicides continues to drive innovation in intermediate synthesis, particularly for strobilurin-class compounds. Patent CN1095830C introduces a groundbreaking methodology for preparing alkoxyiminoacetamide derivatives, which serve as critical precursors in the manufacturing of potent agricultural fungicides. This technology addresses long-standing challenges in selectivity and process safety, offering a robust pathway for producing 2-(2-hydroxymethylphenyl)-2-alkoxyiminoacetamide derivatives. The core innovation lies in a transamination strategy that bypasses the limitations of traditional alkylation, ensuring higher purity and operational efficiency. By leveraging this advanced synthetic route, manufacturers can achieve superior control over stereochemistry, specifically targeting the biologically active E-isomer essential for fungicidal performance. The versatility of this process allows for the seamless integration into existing production lines, making it a valuable asset for any reliable agrochemical intermediate supplier seeking to optimize their portfolio. Furthermore, the downstream utility of these intermediates is well-documented, as they can be efficiently converted into final active ingredients with proven field efficacy. 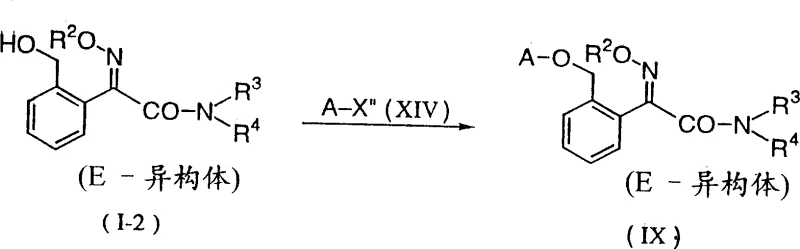
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-substituted alkoxyiminoacetamides relied heavily on direct alkylation of primary amides using aggressive alkylating agents such as dimethyl sulfate or methyl halides. This conventional approach suffers from inherent chemoselectivity issues, often yielding complex mixtures containing unreacted starting materials, the desired mono-alkylated product, and unwanted di-alkylated byproducts. Separating these structurally similar compounds requires energy-intensive purification steps, such as repeated recrystallization or preparative chromatography, which drastically reduces overall yield and increases production costs. Moreover, the use of toxic alkylating agents poses significant safety risks and environmental compliance burdens, necessitating specialized handling protocols and waste treatment facilities. The inability to selectively control the degree of substitution means that process engineers must constantly balance conversion rates against purity specifications, leading to inconsistent batch quality. Additionally, conventional methods often struggle to maintain the desired E/Z stereochemical ratio, requiring additional isomerization steps that further complicate the manufacturing workflow. These cumulative inefficiencies create bottlenecks in the supply chain, limiting the ability to scale production to meet global agricultural demands without compromising on cost or safety standards.
The Novel Approach
In stark contrast, the methodology disclosed in CN1095830C employs a transamination reaction between a hydroxyiminoacetamide precursor and a specific amine compound to achieve precise N-substitution. This novel approach eliminates the formation of mixed alkylation byproducts by utilizing the amine reactant itself to define the substitution pattern, thereby ensuring high selectivity for either mono-methyl or di-methyl derivatives depending on the chosen amine. The reaction proceeds under mild conditions, typically in alcoholic solvents at temperatures ranging from 0°C to 80°C, which significantly reduces energy consumption and thermal stress on the equipment. A remarkable feature of this process is its ability to simultaneously drive the formation of the thermodynamically stable E-isomer during the transamination step, often negating the need for a separate isomerization procedure. This dual functionality streamlines the synthesis workflow, reducing the number of unit operations and minimizing solvent usage. 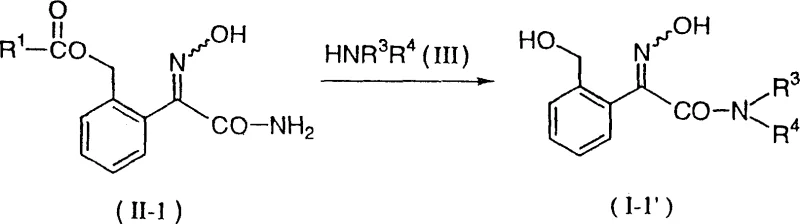
Mechanistic Insights into Transamination and Isomerization Dynamics
The mechanistic foundation of this process relies on the nucleophilic attack of the amine on the amide carbonyl, facilitated by the unique electronic environment of the adjacent oxime ether group. Unlike simple amides, the alpha-oximino structure activates the carbonyl carbon towards nucleophilic substitution while stabilizing the transition state through resonance effects. This activation allows the reaction to proceed efficiently even with weaker nucleophiles like methylamine or ethylamine, avoiding the need for strong bases that could degrade the sensitive oxime functionality. The simultaneous enrichment of the E-isomer suggests a dynamic equilibrium where the transamination event favors the formation of the sterically less hindered E-configuration, possibly through a reversible imidate intermediate. This stereoselectivity is crucial because the E-isomer exhibits significantly higher fungicidal activity compared to its Z-counterpart. Understanding this mechanism allows process chemists to fine-tune reaction parameters, such as solvent polarity and amine concentration, to maximize the E/Z ratio directly in the crude reaction mixture. Furthermore, the stability of the intermediates under acidic conditions permits an optional post-reaction isomerization step using mineral or organic acids if further enrichment is required. This flexibility ensures that manufacturers can consistently meet stringent purity specifications regardless of the initial stereochemical composition of the starting materials.
Beyond the core transamination, the synthesis of the requisite precursors involves a carefully orchestrated sequence of functional group transformations starting from readily available benzoyl cyanides. The preparation of the key intermediate, 2-(2-acyloxymethylphenyl)-2-oxoacetamide, involves the hydrolysis of the nitrile group followed by acylation of the benzylic position. 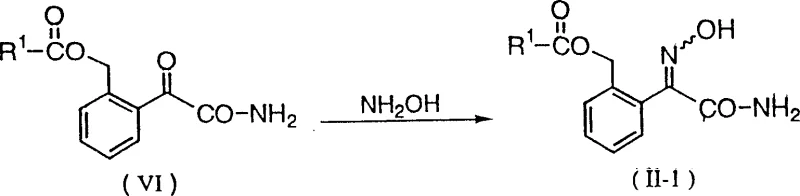
This multi-step sequence is optimized to minimize side reactions, such as the hydrolysis of the ester group, by controlling pH and temperature profiles. The use of metal carboxylate salts, such as potassium acetate, in the presence of catalytic iodides facilitates the nucleophilic displacement of benzylic halides with high efficiency. This upstream optimization ensures a steady supply of high-purity feedstock for the critical transamination step, thereby enhancing the overall robustness of the manufacturing process. The compatibility of these intermediates with various protecting groups allows for further derivatization, expanding the scope of accessible fungicidal analogs. By mastering these mechanistic nuances, production teams can achieve consistent quality across large-scale batches, ensuring reliability for downstream formulation partners.
How to Synthesize Alkoxyiminoacetamide Derivatives Efficiently
Implementing this synthesis route requires strict adherence to the reaction parameters outlined in the patent to ensure optimal yield and stereochemical purity. The process begins with the preparation of the hydroxyimino precursor, followed by the critical transamination step where the amine source is introduced. Operators must monitor reaction progress closely, typically using HPLC or NMR, to determine the endpoint and prevent over-reaction or degradation. The workup procedure involves standard extraction and washing techniques, but care must be taken to maintain the pH within a range that preserves the integrity of the oxime ether bond. For facilities aiming to produce the E-isomer exclusively, an acid-catalyzed isomerization step can be integrated seamlessly into the workflow. The following guide outlines the standardized operational steps derived from the patent examples to assist technical teams in replicating this high-efficiency process.
- React compound of formula (II-1) with an amine compound (III) in a suitable solvent such as alcohol at temperatures between 0-80°C to form the hydroxyimino derivative.
- Optionally perform an isomerization step using acid treatment to enrich the E-isomer content if the starting material has a low E/Z ratio.
- Purify the resulting compound (I-1') or (I-2) using conventional methods like recrystallization or chromatography before further conversion to the final fungicide.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this novel synthesis technology offers substantial strategic benefits for procurement and supply chain management within the agrochemical sector. By shifting away from non-selective alkylation methods, manufacturers can drastically simplify their purification workflows, leading to significant reductions in solvent consumption and waste generation. This streamlined process directly translates to lower operating expenditures, as fewer distillation columns and crystallization vessels are required to achieve pharmaceutical-grade purity. The elimination of hazardous alkylating agents like dimethyl sulfate also reduces the regulatory burden associated with storage and handling, lowering insurance premiums and compliance costs. From a supply chain perspective, the reliance on commodity chemicals such as simple amines and alcohols ensures a stable and resilient raw material base, mitigating the risk of shortages that often plague specialty reagent markets. The robustness of the reaction conditions allows for flexible scheduling and easier scale-up, enabling suppliers to respond rapidly to fluctuating market demands without compromising delivery timelines. Furthermore, the high selectivity of the process minimizes the loss of valuable intermediates, maximizing the throughput of existing production assets and improving overall capital efficiency.
- Cost Reduction in Manufacturing: The selective nature of the transamination reaction eliminates the need for complex separation of mono- and di-alkylated byproducts, which traditionally consumes a large portion of the production budget. By avoiding expensive chromatographic separations and reducing solvent turnover, the overall cost of goods sold is significantly optimized. The milder reaction conditions also reduce energy costs associated with heating and cooling, contributing to a leaner manufacturing footprint. Additionally, the higher yield of the desired E-isomer reduces the amount of starting material required per kilogram of final product, further enhancing economic viability.
- Enhanced Supply Chain Reliability: The process utilizes widely available starting materials such as methylamine, ethylamine, and common alcohols, which are produced on a massive global scale with stable pricing. This contrasts sharply with older methods that depended on specialized alkylating agents subject to volatile market dynamics and strict transport regulations. The simplified workflow reduces the number of critical process steps, decreasing the probability of batch failures and ensuring consistent on-time delivery to customers. Suppliers can maintain lower safety stock levels due to the predictability of the reaction, freeing up working capital for other strategic investments.
- Scalability and Environmental Compliance: The absence of heavy metal catalysts and toxic alkylating agents simplifies wastewater treatment and reduces the environmental impact of the manufacturing process. This alignment with green chemistry principles facilitates easier permitting for capacity expansion and enhances the corporate sustainability profile. The reaction is inherently scalable from pilot plant to commercial tonnage without significant re-engineering, allowing for rapid deployment of new capacity to meet growing agricultural needs. Reduced hazardous waste generation also lowers disposal costs and minimizes the risk of environmental incidents.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific advantages and operational details highlighted in the patent documentation. Understanding these aspects is crucial for technical directors and procurement officers evaluating the feasibility of adopting this route for their specific product portfolios. The answers provide clarity on selectivity, safety, and process integration, helping stakeholders make informed decisions about their supply chain strategies.
Q: How does this process improve selectivity compared to traditional alkylation?
A: Unlike conventional alkylation which produces mixtures of mono- and di-methylated products requiring difficult separation, this transamination method selectively produces the desired mono-methyl or di-methyl compounds directly by choosing the specific amine reactant.
Q: Is a separate isomerization step always required for high E-isomer yield?
A: No, the patent highlights that the transamination reaction itself can yield high E-isomer content even from low E/Z ratio starting materials, though acid-catalyzed isomerization remains an optional optimization step.
Q: What are the safety advantages of this synthesis route?
A: The process utilizes safer solvents like alcohols and avoids harsh alkylating agents like dimethyl sulfate in the critical amination step, significantly reducing industrial hazards and waste treatment complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alkoxyiminoacetamide Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global food security landscape. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. We have rigorously validated the synthesis routes described in CN1095830C within our state-of-the-art facilities, implementing stringent purity specifications and rigorous QC labs to guarantee product excellence. Our commitment to process safety and environmental stewardship aligns perfectly with the advantages of this novel transamination technology, allowing us to offer a sustainable and cost-effective sourcing solution. We understand that every kilogram counts in the competitive agrochemical market, and our optimized processes are designed to deliver maximum value without compromising on quality.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and purity targets. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can enhance your supply chain resilience and profitability. Together, we can drive innovation in agricultural protection and ensure a stable supply of essential fungicide intermediates for the future.
