Advanced Stereoselective Alkylation Process for High-Purity Pharmaceutical Intermediates Manufacturing
The pharmaceutical industry continuously seeks robust synthetic routes for complex chiral intermediates, particularly those serving as precursors for potent therapeutic agents like CCR5 receptor antagonists. Patent CN100343240C discloses a groundbreaking methodology for the stereoselective alkylation of chiral 2-alkyl-4-protected piperazines, addressing critical challenges in stereochemical purity and process efficiency. This technology enables the synthesis of Formula VIII compounds, which are vital intermediates in the production of HIV treatments and anti-inflammatory drugs, with unprecedented control over diastereomeric ratios. By shifting from traditional organic base catalysts to inexpensive inorganic bases, this process not only enhances the chemical outcome but also aligns with modern green chemistry principles, making it a highly attractive option for reliable pharmaceutical intermediates supplier networks aiming to optimize their manufacturing portfolios.
The limitations of conventional methods for amine alkylation often stem from the reliance on bulky organic bases such as 2,2,6,6-tetramethylpiperidine (TMP) or diisopropylethylamine (DIPEA). As illustrated in prior art like Scheme 1 described by Tagat et al., these traditional approaches typically suffer from moderate yields ranging between 50% and 65%, coupled with poor stereoselectivity where the desired isomer ratio is often no better than 3:1. This lack of selectivity necessitates extensive downstream purification processes, such as chromatography or repeated crystallizations, which drastically increase production costs and extend lead times. Furthermore, the use of stoichiometric amounts of expensive organic amines generates significant waste streams, complicating environmental compliance and adding to the overall burden of cost reduction in pharmaceutical intermediates manufacturing.
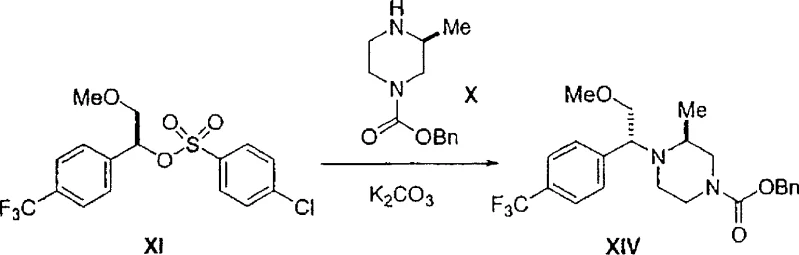
The novel approach presented in this patent fundamentally alters the reaction landscape by utilizing inorganic bases such as potassium carbonate, sodium bicarbonate, or trisodium phosphate to catalyze the alkylation of chiral amines. This strategic substitution results in a dramatic improvement in process performance, with yields consistently reaching approximately 85% and diastereomeric ratios exceeding 95:5 in favor of the desired stereoisomer. The mechanism likely involves a different transition state geometry facilitated by the inorganic cation, which favors the inversion or retention of configuration required for the specific (R,S) or (S,S) product depending on the substrate. This enhancement eliminates the need for complex separation of unwanted isomers, thereby streamlining the workflow and ensuring a high-purity pharmaceutical intermediate output that meets stringent regulatory specifications without additional processing burdens.
Mechanistic Insights into Inorganic Base-Catalyzed Stereoselective Alkylation
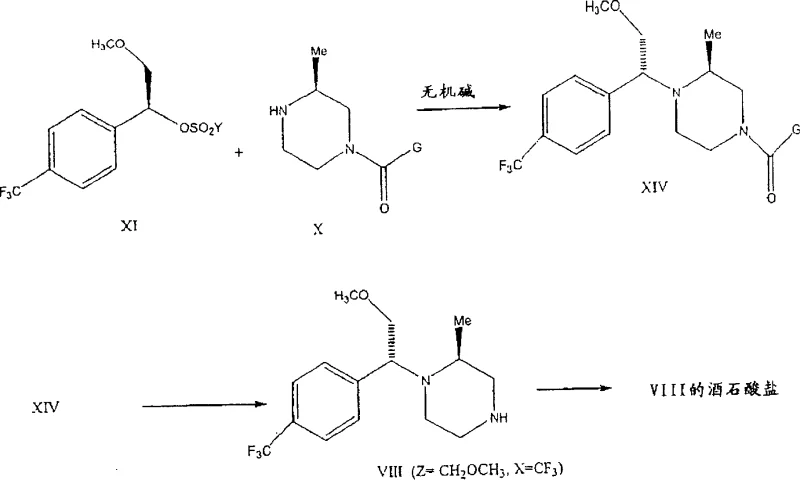
The core of this technological advancement lies in the interaction between the chiral sulfonate alkylating agent (Formula XI) and the mono-protected piperazine (Formula X) under the influence of inorganic bases. The reaction proceeds efficiently in polar aprotic or mixed solvent systems, such as toluene and acetonitrile, at elevated temperatures ranging from 80°C to 110°C. The inorganic base acts not merely as an acid scavenger but plays a pivotal role in modulating the nucleophilicity of the piperazine nitrogen while minimizing racemization at the chiral center of the alkylating agent. Detailed experimental data indicates that when Z equals CH2OMe, the process achieves an RS/SS stereochemical ratio of greater than 90:10, a significant leap from the roughly 82:18 ratio observed with organic bases. This high level of stereocontrol is critical for ensuring the biological efficacy of the final CCR5 antagonist, as the wrong enantiomer could be inactive or even toxic.
Impurity control is another major advantage of this inorganic base-mediated pathway. Traditional organic base methods often lead to side reactions such as elimination or over-alkylation, generating difficult-to-remove impurities that compromise the purity profile. In contrast, the use of mild inorganic carbonates or phosphates suppresses these side pathways, resulting in a cleaner reaction profile as confirmed by HPLC analysis in the patent examples. The ability to directly use crude intermediates without extensive purification between steps further underscores the robustness of this method. For R&D teams, this means a more predictable impurity spectrum that is easier to characterize and control, facilitating faster regulatory filings and reducing the risk of batch failures during the commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Chiral Piperazine Intermediates Efficiently
The synthesis of these high-value chiral building blocks follows a logical sequence starting from readily available chiral alcohols and piperazines. The process begins with the selective protection of the piperazine nitrogen to prevent di-alkylation, followed by the activation of the chiral alcohol into a sulfonate ester. The key alkylation step then brings these two fragments together under the optimized inorganic base conditions described above. Finally, the protecting group is removed to reveal the free amine, which can be isolated as a stable salt. This streamlined approach minimizes unit operations and maximizes material throughput. For detailed operational parameters, safety considerations, and specific workup procedures, please refer to the standardized synthesis guide provided below which encapsulates the critical process parameters derived from the patent embodiments.
- Prepare the mono-protected chiral amine (Formula X) by reacting 2-methylpiperazine with a protecting group reagent (e.g., benzyl chloroformate) under acid or base catalysis to ensure high regioselectivity at the 4-position.
- Synthesize the chiral alkylating agent (Formula XI) by converting the corresponding chiral alcohol (Formula XII) into a sulfonate ester using a sulfonyl chloride and a base like DABCO.
- Perform the stereoselective alkylation by reacting Formula XI and Formula X in a solvent mixture (e.g., Toluene/Acetonitrile) using an inorganic base such as potassium carbonate at 80-110°C to achieve high diastereomeric ratio.
- Remove the protecting group from the alkylated product (Formula XIV) using acid (e.g., HCl) or base (e.g., NaOH) hydrolysis to yield the final free base (Formula VIII), optionally converting it to a stable salt like tartrate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this inorganic base-catalyzed process offers substantial strategic benefits beyond mere chemical efficiency. The replacement of costly organic bases with commodity inorganic salts like potassium carbonate represents a direct and significant reduction in raw material costs. Moreover, the improved yield and selectivity mean that less starting material is wasted, effectively lowering the cost of goods sold (COGS) per kilogram of active intermediate. The simplified workup procedure, which avoids complex chromatographic separations, reduces solvent consumption and waste disposal costs, contributing to a more sustainable and economically viable manufacturing model. These factors combined make this technology a powerful tool for achieving cost reduction in pharmaceutical intermediates manufacturing while maintaining high quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive organic amines and the reduction in purification steps lead to a leaner cost structure. By avoiding the need to separate close-eluting diastereomers, manufacturers save significantly on chromatography resins and solvent volumes. This efficiency translates into a more competitive pricing model for the final API intermediate, allowing partners to maintain healthy margins even in a price-sensitive market environment.
- Enhanced Supply Chain Reliability: Inorganic bases such as potassium carbonate and sodium bicarbonate are globally available commodity chemicals with stable supply chains, unlike specialized organic bases that may face sourcing bottlenecks. This availability ensures consistent production schedules and reduces the risk of delays caused by raw material shortages. Additionally, the robustness of the reaction conditions allows for flexible manufacturing across different sites, enhancing the overall resilience of the supply network for critical healthcare ingredients.
- Scalability and Environmental Compliance: The patent explicitly demonstrates the scalability of this process, with examples conducted in 100-gallon glass-lined reactors, proving its readiness for industrial production. The use of less hazardous inorganic reagents and the generation of simpler waste streams simplify environmental compliance and wastewater treatment. This alignment with green chemistry principles not only reduces regulatory risks but also supports corporate sustainability goals, making it an ideal choice for long-term partnerships focused on responsible chemical manufacturing.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its implementation. The following questions address common inquiries regarding the reaction conditions, scalability, and quality attributes of the process. These answers are derived directly from the experimental data and descriptions found within the patent documentation, providing a reliable foundation for technical decision-making. For further clarification on specific batch records or analytical methods, our technical team is available to provide deeper insights tailored to your project requirements.
Q: What is the primary advantage of using inorganic bases over organic bases in this alkylation?
A: According to patent CN100343240C, using inorganic bases like potassium carbonate significantly improves both yield (up to 85% vs 58%) and stereoselectivity (RS/SS ratio >95:5 vs 82:18) compared to organic bases like DIPEA, reducing the need for costly purification steps.
Q: Is this synthesis process scalable for commercial production?
A: Yes, the patent details embodiments performed in large-scale equipment, specifically mentioning a 100-gallon glass-lined reactor for the alkylation step, demonstrating that the process is robust and suitable for commercial scale-up of complex pharmaceutical intermediates.
Q: What solvents are recommended for the stereoselective alkylation step?
A: The patent identifies several effective solvent systems, including mixtures of toluene and acetonitrile, as well as NMP. These solvents facilitate the suspension of the inorganic base and ensure efficient reaction kinetics at elevated temperatures between 80°C and 110°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 2-Methyl-4-Protected Piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of life-saving medications. Our expertise in process chemistry allows us to adapt advanced methodologies like the inorganic base-catalyzed alkylation described in CN100343240C to meet your specific production needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply requirements are met with consistency and precision. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of chiral piperazine intermediate delivered meets the highest industry standards for stereochemical purity and chemical identity.
We invite you to collaborate with us to leverage this innovative technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this optimized route. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
