Advanced Stereoselective Alkylation Process for High-Purity Pharmaceutical Intermediates
Introduction to Advanced Stereoselective Alkylation Technology
The pharmaceutical industry continuously seeks robust synthetic routes for complex chiral intermediates, particularly those serving as key building blocks for potent therapeutic agents. Patent CN1642930A discloses a groundbreaking methodology for the stereoselective alkylation of chiral 2-alkyl-4-protected piperazines, a critical structural motif found in CCR5 receptor antagonists used for treating HIV and inflammatory diseases. This technology represents a significant paradigm shift from traditional organic base-catalyzed reactions to a more efficient inorganic base-catalyzed system. By leveraging inexpensive and readily available inorganic catalysts, this process addresses long-standing challenges in stereocontrol and yield optimization. For R&D teams and supply chain leaders, understanding this innovation is vital for securing a reliable pharmaceutical intermediate supplier capable of delivering high-purity materials with reduced environmental impact and lower production costs.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the stereoselective alkylation of chiral amines has relied heavily on organic bases such as tertiary amines, including pyridine, triethylamine, and notably 2,2,6,6-tetramethylpiperidine (TMP). As illustrated in the prior art reaction schemes, these conventional methods often suffer from suboptimal performance metrics that hinder commercial viability. Specifically, reactions utilizing organic bases typically result in moderate chemical yields ranging from 50% to 65%, coupled with poor stereoselectivity where the ratio of desired to undesired isomers is often as low as 3:1. This lack of selectivity necessitates additional downstream purification steps, such as chromatography or recrystallization, which drastically increase manufacturing costs and extend lead times. Furthermore, the use of bulky organic bases can introduce steric hindrance issues that limit reaction efficiency, making the scale-up of complex polymer additives or API intermediates technically challenging and economically burdensome.
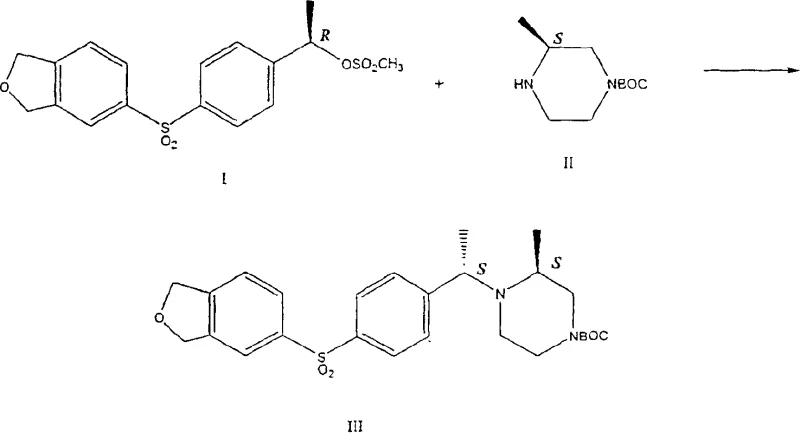
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes inorganic bases to catalyze the alkylation reaction, resulting in a dramatic improvement in both yield and stereochemical purity. By replacing expensive organic amines with inorganic salts such as potassium carbonate, sodium bicarbonate, or trisodium phosphate, the process achieves yields exceeding 85% and diastereomeric ratios that can surpass 90:10 or even 95:5 depending on the substrate. This method is particularly effective when the substituent Z is a methoxymethyl group, where the inorganic catalyst facilitates a highly selective transformation that minimizes the formation of unwanted stereoisomers. The transition to inorganic catalysis not only simplifies the reaction workup but also enhances the overall atom economy of the synthesis. This breakthrough offers a compelling solution for cost reduction in electronic chemical manufacturing and pharmaceutical production, providing a scalable pathway that aligns with modern green chemistry principles.
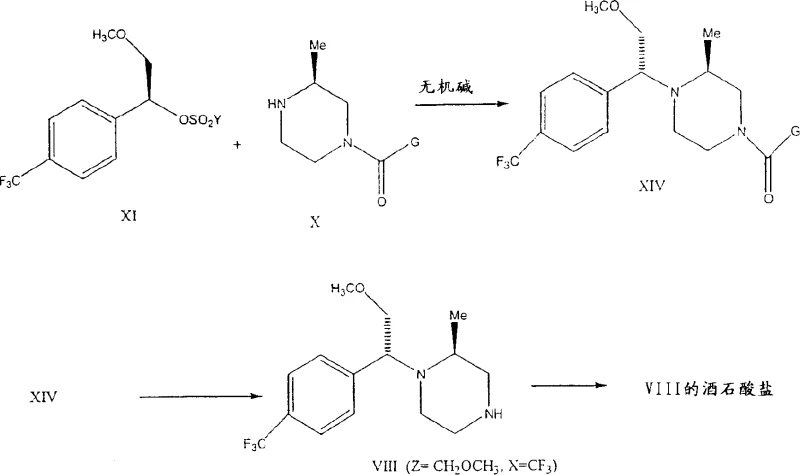
Mechanistic Insights into Inorganic Base-Catalyzed Alkylation
The mechanistic advantage of using inorganic bases lies in their ability to facilitate the nucleophilic attack of the piperazine nitrogen on the chiral alkylating agent without the steric bulk associated with large organic amine molecules. In the reaction involving chiral 2-methyl-4-protected piperazines, the inorganic base acts as a proton scavenger, neutralizing the acid by-product generated during the displacement of the leaving group, such as a mesylate or tosylate. This efficient acid removal drives the equilibrium towards product formation while maintaining a reaction environment that favors the retention or specific inversion of configuration at the chiral center. Detailed analysis of the patent examples reveals that the nature of the substituent Z plays a crucial role in determining the stereochemical outcome; for instance, when Z is a methyl group, the S,S isomer is predominantly formed, whereas a methoxymethyl group favors the R,S configuration. This tunability allows chemists to precisely engineer the stereochemistry of the final API intermediate, ensuring compliance with stringent regulatory requirements for chiral purity.
Furthermore, the impurity profile of the reaction is significantly cleaner when inorganic bases are employed. Conventional organic bases can sometimes participate in side reactions or form stable complexes with the product, leading to difficult-to-remove impurities that compromise the quality of the high-purity OLED material or pharmaceutical ingredient. The inorganic system described herein minimizes these side reactions, resulting in a crude product that often requires minimal purification before the final deprotection step. The deprotection itself can be achieved through simple acid or base hydrolysis, as demonstrated by the conversion of protected intermediates to the free base or tartrate salt forms. This streamlined sequence reduces the number of unit operations required, thereby lowering the risk of product degradation and ensuring a consistent supply of high-quality intermediates for downstream drug synthesis.
How to Synthesize Chiral 2-Methyl-4-Protected Piperazine Efficiently
The synthesis of these valuable chiral intermediates follows a logical and robust sequence that begins with the selective protection of the piperazine ring, followed by the preparation of the chiral alkylating agent, and concludes with the key alkylation step. The patent provides extensive experimental data supporting the feasibility of this route, including specific reaction conditions, solvent choices, and temperature ranges that optimize the outcome. For process chemists looking to implement this technology, the detailed examples serve as a comprehensive guide for translating laboratory success into commercial reality. The following sections outline the standardized synthesis steps derived directly from the patent disclosure, ensuring reproducibility and safety during scale-up operations.
- Prepare the protected piperazine starting material by selectively mono-protecting 2-methylpiperazine at the 4-position using acid or base catalysis.
- Synthesize the chiral alkylating agent (sulfonate ester) from the corresponding chiral alcohol using sulfonyl chloride and a base.
- React the protected piperazine with the sulfonate ester in the presence of an inorganic base catalyst (e.g., potassium carbonate) in a suitable solvent.
- Remove the protecting group from the alkylated product using acid or base hydrolysis to obtain the final chiral amine intermediate.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this inorganic base-catalyzed process offers tangible benefits that extend beyond mere technical superiority. The primary advantage lies in the substantial cost savings achieved by eliminating the need for expensive, specialized organic bases like TMP or DIPEA, which are often subject to price volatility and supply constraints. By switching to commodity inorganic chemicals such as potassium carbonate, manufacturers can significantly reduce raw material costs while simultaneously simplifying the supply chain logistics. Additionally, the higher yields and improved selectivity mean that less starting material is wasted, further enhancing the overall cost efficiency of the manufacturing process. This economic efficiency is critical for maintaining competitiveness in the global market for reliable agrochemical intermediate supplier and pharmaceutical partners.
- Cost Reduction in Manufacturing: The replacement of costly organic catalysts with inexpensive inorganic salts directly lowers the bill of materials for each batch produced. Moreover, the improved reaction selectivity reduces the burden on purification systems, leading to lower solvent consumption and waste disposal costs. This holistic reduction in operational expenses allows for more competitive pricing strategies without compromising on quality margins. The elimination of complex separation steps also translates to reduced energy consumption and shorter cycle times, contributing to a leaner and more agile manufacturing operation.
- Enhanced Supply Chain Reliability: Inorganic bases are widely available commodities with stable supply chains, unlike some specialized organic reagents that may face sourcing bottlenecks. This availability ensures continuous production capability and reduces the risk of delays caused by raw material shortages. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents, further adds to the reliability of the process, allowing for flexible scheduling and inventory management. For companies seeking reducing lead time for high-purity pharmaceutical intermediates, this stability is a key strategic asset.
- Scalability and Environmental Compliance: The process has been successfully demonstrated on a multi-kilogram scale, proving its viability for commercial scale-up of complex pharmaceutical intermediates. The use of inorganic bases generates less hazardous waste compared to organic amines, simplifying effluent treatment and ensuring compliance with increasingly strict environmental regulations. This eco-friendly profile not only mitigates regulatory risk but also enhances the corporate sustainability image, which is increasingly important for stakeholders and customers alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this stereoselective alkylation technology. These answers are derived from the specific data points and experimental results presented in the patent documentation, providing clarity on the practical aspects of the synthesis. Understanding these details helps stakeholders make informed decisions about integrating this process into their existing manufacturing portfolios.
Q: Why is the use of inorganic bases preferred over organic bases in this alkylation process?
A: The patent data demonstrates that inorganic bases significantly improve both the chemical yield and the stereoselectivity of the reaction compared to traditional organic bases like TMP or DIPEA, reducing the need for costly purification steps.
Q: What level of stereochemical purity can be achieved with this method?
A: Depending on the specific substituents, the process can achieve diastereomeric ratios exceeding 90:10 or even 95:5, which is substantially higher than the 3:1 ratio observed with conventional organic base methods.
Q: Is this synthesis route scalable for commercial production?
A: Yes, the patent includes examples performed on a multi-kilogram scale using standard reactor equipment, indicating that the process is robust and suitable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral 2-Methyl-4-Protected Piperazine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of advanced synthetic methodologies in driving the next generation of therapeutic breakthroughs. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative processes described in patent CN1642930A can be seamlessly transferred to industrial manufacturing. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of chiral intermediate performs consistently in your downstream applications. Our dedication to technical excellence makes us a trusted partner for global pharmaceutical companies seeking to optimize their supply chains.
We invite you to collaborate with us to explore how this stereoselective alkylation technology can enhance your product portfolio. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. Please contact us to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise can drive value and efficiency in your operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
