Advanced Synthesis of Phenylacetic Acid Derivatives for High-Purity Pharmaceutical Intermediates
Advanced Synthesis of Phenylacetic Acid Derivatives for High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously demands robust and scalable synthetic routes for active pharmaceutical ingredient (API) intermediates, particularly for classes like cyclooxygenase (COX-2) inhibitors. Patent CN1377337A presents a comprehensive methodology for the preparation of 2-phenylamino-5-alkylphenylacetic acid derivatives, designated as Formula I compounds. These molecules serve as critical building blocks in the development of selective COX-2 inhibitors, which are vital for treating inflammation and pain. The disclosed process centers on the strategic cleavage of a lactam precursor, Formula II, using basic hydrolysis conditions. This approach offers a distinct advantage over traditional methods by leveraging stable oxindole intermediates that can be synthesized through efficient Friedel-Crafts cyclization protocols. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, understanding the mechanistic depth and operational simplicity of this patent is essential for securing a stable supply chain.
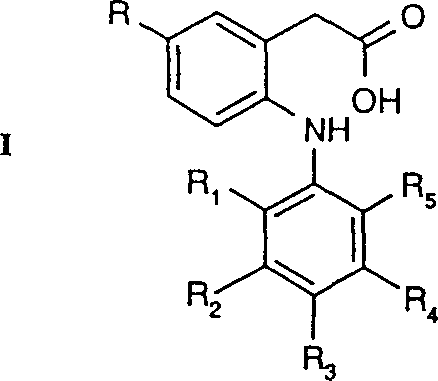
In the landscape of fine chemical manufacturing, the transition from laboratory-scale discovery to commercial production often hinges on the reliability of the synthetic pathway. Conventional methods for synthesizing substituted phenylacetic acids frequently encounter significant hurdles, including the use of hazardous reagents, complex multi-step sequences with low overall yields, and difficulties in removing trace metal impurities from catalytic cycles. Traditional approaches might rely on direct alkylation strategies that suffer from poor regioselectivity or require harsh acidic conditions that degrade sensitive functional groups on the aromatic rings. Furthermore, the purification of intermediates in older processes often necessitates extensive chromatographic separation, which is economically unfeasible for large-scale operations. These limitations result in increased production costs, extended lead times, and inconsistent batch-to-batch quality, posing substantial risks for supply chain heads managing inventory for downstream API synthesis.
The novel approach detailed in CN1377337A circumvents these historical bottlenecks by introducing a streamlined route via Formula II lactams. This strategy utilizes a Friedel-Crafts intramolecular cyclization to construct the indole core, a transformation that is highly atom-economical and generates minimal waste compared to linear syntheses. The subsequent hydrolysis of the lactam ring to the open-chain phenylacetic acid is achieved under mild basic conditions, typically employing aqueous sodium hydroxide at temperatures between 50°C and 100°C. This specific pathway ensures high purity profiles because the crystalline nature of the lactam intermediates allows for effective purification prior to the final ring-opening step. By focusing on this lactam hydrolysis tactic, manufacturers can achieve cost reduction in API manufacturing through simplified work-up procedures and the avoidance of expensive protecting group strategies that are often required in alternative synthetic designs.
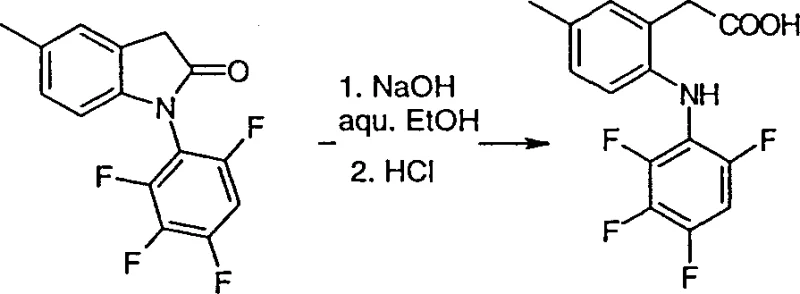
Mechanistic Insights into Friedel-Crafts Cyclization and Lactam Hydrolysis
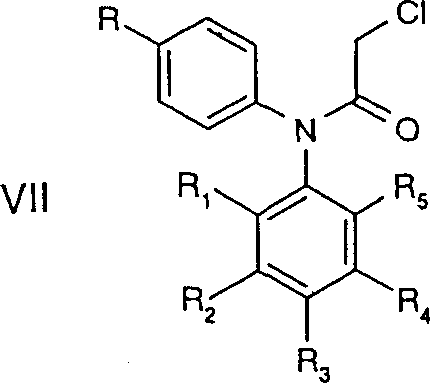
The core of this synthetic technology lies in the formation of the oxindole scaffold via a Friedel-Crafts alkylation mechanism. The process begins with an N-acylated diphenylamine derivative, Formula VII, which is subjected to Lewis acid catalysis, preferably using aluminum chloride (AlCl3). The reaction is conducted at elevated temperatures, typically ranging from 100°C to 180°C, often under solvent-free melt conditions or in high-boiling inert solvents like dichlorobenzene. Mechanistically, the Lewis acid activates the alpha-chloro carbonyl moiety, generating an electrophilic acylium-like species that undergoes intramolecular electrophilic aromatic substitution on the activated phenyl ring. This cyclization is highly regioselective, driven by the electronic properties of the substituents on the aniline ring, ensuring the formation of the desired 5-substituted oxindole isomer. The robustness of this thermal cyclization allows for the processing of diverse substrates with varying electronic demands, making it a versatile tool for generating libraries of COX-2 inhibitor precursors.
Following the construction of the heterocyclic core, the conversion to the target phenylacetic acid involves a nucleophilic acyl substitution mechanism during the hydrolysis phase. The lactam nitrogen, being part of an amide bond, is susceptible to nucleophilic attack by hydroxide ions generated from strong bases like sodium hydroxide. The reaction proceeds through a tetrahedral intermediate, which subsequently collapses to open the five-membered ring, yielding the carboxylate salt of the phenylacetic acid derivative. A critical aspect of impurity control in this stage is the management of reaction temperature and base concentration to prevent potential decarboxylation or degradation of sensitive halogen substituents on the aromatic rings. The patent specifies that the resulting reaction mixture is conveniently neutralized with mineral acids, such as hydrochloric acid, to precipitate the free acid product. This precipitation step serves as a powerful purification tool, as the target phenylacetic acids often exhibit low solubility in acidic aqueous media, allowing for isolation via simple filtration and washing, thereby ensuring high chemical purity suitable for pharmaceutical applications.
How to Synthesize 2-Phenylamino-5-alkylphenylacetic Acid Efficiently
Implementing this synthesis requires precise control over reaction parameters to maximize yield and minimize impurity formation. The process generally initiates with the preparation of the diphenylamine backbone, often achieved through palladium-catalyzed cross-coupling reactions such as the Buchwald-Hartwig amination. This step couples a substituted aniline with an aryl halide using catalysts like Pd(dba)2 and ligands such as BINAP or tri-tert-butylphosphine in solvents like toluene. Once the amine backbone is established, it is N-acylated with chloroacetyl chloride to form the cyclization precursor. The subsequent ring closure and hydrolysis steps follow the mechanistic principles previously described. For detailed operational parameters, stoichiometry, and specific work-up instructions tailored to your facility's capabilities, please refer to the standardized synthesis guide below.
- Perform Buchwald-Hartwig amination to couple substituted anilines with aryl halides using Pd catalysts.
- Execute N-acylation with chloroacetyl chloride followed by Friedel-Crafts cyclization using AlCl3 at elevated temperatures.
- Hydrolyze the resulting oxindole lactam with aqueous sodium hydroxide and neutralize to isolate the free acid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route outlined in CN1377337A offers significant strategic benefits for procurement managers and supply chain directors focused on long-term stability and cost efficiency. The reliance on commodity chemicals such as aluminum chloride, sodium hydroxide, and chloroacetyl chloride means that raw material sourcing is not constrained by specialized or monopolistic suppliers. This abundance of starting materials drastically reduces the risk of supply disruptions caused by raw material shortages, ensuring a continuous flow of intermediates for downstream API production. Furthermore, the elimination of transition metal catalysts in the final hydrolysis step simplifies the purification process, removing the need for expensive and time-consuming heavy metal scavenging resins. This reduction in processing complexity translates directly into lower operational expenditures and a smaller environmental footprint, aligning with modern green chemistry initiatives.
- Cost Reduction in Manufacturing: The process achieves substantial cost savings by utilizing a convergent synthesis strategy where complex molecular architecture is built early in the sequence via the Friedel-Crafts cyclization. This avoids the need for late-stage functionalization which often suffers from lower yields and higher reagent costs. Additionally, the ability to perform the cyclization under melt conditions or in high-boiling solvents allows for high concentration reactions, maximizing reactor throughput and reducing solvent consumption volumes. The crystallization-driven purification of both the lactam intermediate and the final acid product minimizes the need for preparative chromatography, which is a major cost driver in fine chemical manufacturing. These factors collectively contribute to a highly economical production model.
- Enhanced Supply Chain Reliability: The synthetic pathway is designed with scalability in mind, utilizing robust reaction conditions that are tolerant to minor variations in temperature and mixing, which is crucial for transfer from pilot plant to commercial scale. The intermediates, particularly the oxindole lactams, are chemically stable solids that can be stored and transported without significant degradation, providing flexibility in inventory management. This stability allows manufacturers to build strategic stockpiles of key intermediates, buffering against potential fluctuations in demand or unexpected production delays. The use of standard equipment materials, such as glass-lined steel reactors capable of handling acidic and basic conditions, further ensures that the process can be adopted by a wide range of contract manufacturing organizations without requiring specialized infrastructure investments.
- Scalability and Environmental Compliance: The process demonstrates excellent scalability due to the straightforward nature of the unit operations involved, primarily heating, stirring, filtration, and crystallization. The waste streams generated are predominantly aqueous salts and organic solvents that can be readily treated or recycled using standard industrial wastewater treatment protocols. The avoidance of exotic reagents and the minimization of solvent usage in the cyclization step significantly reduce the volume of hazardous waste requiring disposal. This alignment with environmental compliance standards reduces regulatory burdens and associated disposal costs, making the process sustainable for long-term commercial production. The high atom economy of the cyclization step further supports sustainability goals by maximizing the incorporation of raw materials into the final product.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of these phenylacetic acid derivatives. The answers are derived directly from the experimental data and procedural descriptions found within the patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these specifics helps in evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: What are the key reaction conditions for the lactam hydrolysis step?
A: The hydrolysis is typically conducted using strong bases like 30% aqueous sodium hydroxide at elevated temperatures ranging from 50°C to 100°C, often in the presence of water-miscible organic solvents such as ethanol.
Q: How is the diphenylamine backbone constructed in this process?
A: The backbone is formed via palladium-catalyzed cross-coupling reactions, specifically utilizing catalysts like Pd(dba)2 with ligands such as BINAP or tri-tert-butylphosphine to couple aniline derivatives with aryl halides.
Q: What purification methods are recommended for the final phenylacetic acid products?
A: The final products are effectively purified through crystallization techniques, often achieved by cooling the reaction mixture to ambient temperature after acid neutralization, or by recrystallization from solvents like 2-propanol.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylacetic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development of next-generation therapeutics. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and efficient. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of phenylacetic acid derivatives meets the exacting standards required by global regulatory bodies. Our commitment to technical excellence allows us to navigate the complexities of Friedel-Crafts cyclizations and base-mediated hydrolyses with precision, delivering products that facilitate the rapid advancement of your drug development programs.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall cost of goods. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments tailored to your target molecules. Let us collaborate to secure a reliable supply of high-purity pharmaceutical intermediates that drive your success in the competitive marketplace.
