Advanced Carbonylation Technology for High-Purity Phenylacetic Acid Derivatives in Pharmaceutical Manufacturing
Advanced Carbonylation Technology for High-Purity Phenylacetic Acid Derivatives in Pharmaceutical Manufacturing
The pharmaceutical industry's relentless pursuit of potent neurokinin-1 (NK-1) receptor antagonists has placed a premium on the efficient synthesis of their key structural motifs. Patent CN1235856C discloses a groundbreaking methodology for preparing phenylacetic acid derivatives of general Formula I, which serve as critical intermediates for therapeutically active compounds such as 2-(3,5-bis-trifluoromethyl-phenyl)-N-methyl-N-(6-morpholin-4-yl-4-o-tolyl-pyridin-3-yl)-isobutyramide. These molecules are pivotal in the development of treatments targeting substance P-mediated pathways. The disclosed technology represents a significant paradigm shift from traditional alkylation routes, offering a streamlined pathway that enhances both chemical purity and process economics. By leveraging a novel combination of Grignard chemistry and acid-catalyzed carbonylation, this process addresses long-standing challenges in impurity control and raw material costs.
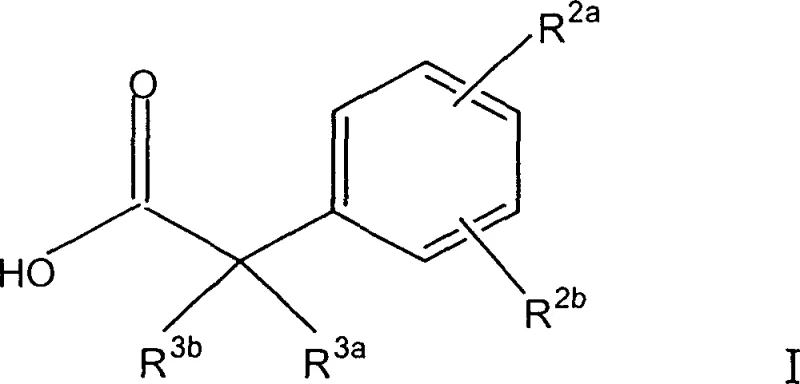
For R&D directors and process chemists, the structural versatility of Formula I is paramount. The substituents R2a and R2b can independently vary from hydrogen and halogens to lower alkoxy and cyano groups, while R3a and R3b allow for diverse alkyl or cycloalkyl modifications. This flexibility enables the rapid generation of analog libraries essential for structure-activity relationship (SAR) studies. However, the true value of this patent lies not just in the scope of compounds covered, but in the robustness of the synthetic route designed to access them, ensuring that high-purity standards required for clinical candidates can be met consistently.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha,alpha-disubstituted phenylacetic acids has relied heavily on the sequential alkylation of phenylacetic acid esters or acids. As illustrated by the starting materials in Formula IV found in prior art, this approach necessitates the use of expensive, pre-functionalized starting compounds. The fundamental flaw in this conventional strategy is the lack of selectivity during the second alkylation step. When attempting to introduce the second alkyl group to form the quaternary center, the reaction inevitably generates considerable amounts of mono-alkylated intermediates (Formula V) and over-alkylated by-products, including unwanted alkylation on the aromatic ring itself.
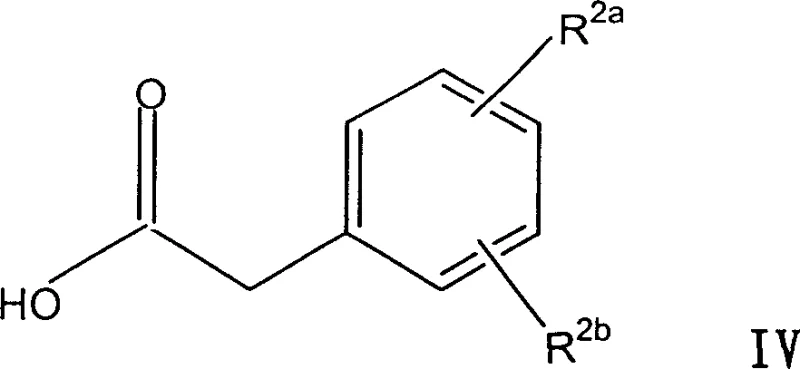
These by-products possess physical and chemical properties remarkably similar to the desired product, rendering standard purification techniques like crystallization largely ineffective. Consequently, manufacturers are often forced to employ resource-intensive chromatographic separations, which are impractical for large-scale production. Furthermore, the concentration of these impurities fluctuates wildly with minor changes in reaction conditions, leading to inconsistent batch quality. This unpredictability creates a significant bottleneck for supply chain managers who require reliable, reproducible manufacturing processes to meet regulatory deadlines and commercial demand without costly delays.
The Novel Approach
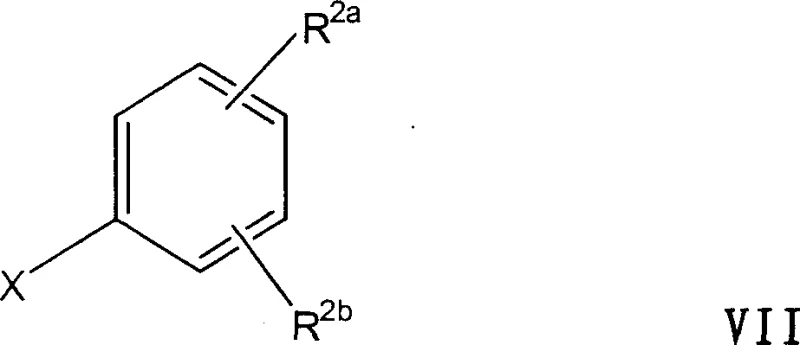
In stark contrast, the methodology described in CN1235856C circumvents these selectivity issues by reversing the order of bond construction. Instead of building the alkyl groups onto an existing acid framework, the process constructs the carbon skeleton first via a Grignard reaction between an aryl magnesium halide (Formula VII) and a ketone (Formula VIII). This addition reaction is inherently high-yielding and produces a tertiary alcohol intermediate (Formula IX) with exceptional cleanliness. The subsequent conversion of this alcohol to the target acid is achieved through a direct carbonylation step in the presence of a strong acid catalyst.
This strategic inversion of the synthetic logic eliminates the formation of the troublesome mono-alkylated and ring-alkylated impurities that plague the old method. Because the quaternary center is established during the Grignard addition—a reaction known for its reliability—the subsequent carbonylation merely functionalizes the existing alcohol without disturbing the carbon skeleton. This results in a reaction profile where the target compound is the predominant species, drastically simplifying downstream processing. For procurement teams, this translates to a process that is not only chemically superior but also economically advantageous due to the elimination of complex purification stages and the use of more readily available raw materials.
Mechanistic Insights into Grignard Addition and Acid-Catalyzed Carbonylation
The core of this innovative process lies in the seamless integration of two distinct chemical transformations. The first stage involves the formation of the Grignard reagent from an aryl halide where X is chlorine, bromine, or iodine. This reaction is typically conducted in ether solvents such as diethyl ether or tetrahydrofuran, or mixtures thereof with aromatic solvents like toluene. The reaction proceeds under normal pressure at temperatures ranging from 15°C to the reflux temperature of the mixture. The resulting organomagnesium species then attacks the carbonyl carbon of the ketone (Formula VIII), forming the tertiary alcohol intermediate (Formula IX). A key advantage here is that the purity of this alcohol intermediate is not critical; it can be as low as 70% and still yield the final acid with a purity of at least 97%, demonstrating the robustness of the subsequent step.
The second stage, the carbonylation of the tertiary alcohol, is where the true chemical ingenuity is displayed. This transformation requires the presence of a strong acid, preferably a fluorinated sulfonic acid such as trifluoromethanesulfonic acid (CF3SO3H) or fluorosulfonic acid (FSO3H). The reaction is carried out under a carbon monoxide atmosphere at pressures between 1×10^5 and 5×10^7 Pa, with optimal results observed between 2×10^6 and 6×10^6 Pa. The mechanism likely involves the protonation of the hydroxyl group to form a stable carbocation, which is then trapped by carbon monoxide to form an acylium ion, followed by hydrolysis to yield the carboxylic acid. The addition of small amounts of water (0.1 to 1 molar equivalent) or formic acid significantly enhances selectivity towards the desired acid product. This precise control over reaction conditions ensures that the delicate balance between carbocation formation and potential decomposition is maintained, leading to high yields without the need for transition metal catalysts.
How to Synthesize 2-(3,5-Bistrifluoromethylphenyl)-2-methyl-propionic Acid Efficiently
The synthesis of specific derivatives, such as 2-(3,5-bistrifluoromethylphenyl)-2-methyl-propionic acid, exemplifies the practical application of this technology. The process begins with the preparation of the Grignard reagent from 3,5-bis-trifluoromethylbromobenzene and magnesium turnings in diethyl ether, followed by the addition of acetone to generate the corresponding tertiary alcohol. This intermediate is then subjected to carbonylation in dichloromethane using trifluoromethanesulfonic acid as the catalyst under a CO pressure of 3×10^6 Pa. The detailed standardized synthesis steps for replicating this high-efficiency route are outlined in the guide below.
- Prepare a Grignard reagent from an aryl halide (Formula VII) in an ether solvent and react it with a ketone (Formula VIII) to form a tertiary alcohol intermediate (Formula IX).
- Subject the tertiary alcohol intermediate to carbonylation in the presence of a strong fluorinated sulfonic acid (e.g., triflic acid) and carbon monoxide.
- Maintain reaction pressure between 1x10^6 and 1x10^7 Pa and temperature between -20°C and 60°C to ensure high selectivity and yield without complex purification.
Commercial Advantages for Procurement and Supply Chain Teams
For stakeholders focused on the bottom line and supply continuity, the shift from conventional alkylation to this carbonylation-based route offers profound strategic benefits. The most immediate impact is seen in the raw material landscape. The conventional method relies on Formula IV starting materials, which are often specialized, expensive, and subject to limited supplier availability. In contrast, the new method utilizes aryl halides and simple ketones, which are commodity chemicals produced on a massive global scale. This substitution fundamentally alters the cost structure of the synthesis, decoupling production from niche supply chains and anchoring it in robust, high-volume markets.
- Cost Reduction in Manufacturing: The economic advantages extend beyond raw material pricing to the operational expenses of the manufacturing plant. By eliminating the formation of difficult-to-remove impurities, the new process removes the need for expensive and time-consuming chromatographic purification steps. The target acid can often be isolated simply by crystallization or extraction, significantly reducing solvent consumption and waste disposal costs. Furthermore, the absence of transition metal catalysts in the carbonylation step means there is no need for costly heavy metal scavenging processes, which are mandatory for pharmaceutical intermediates to meet strict residual metal specifications. This streamlining of the workflow leads to substantial cost savings per kilogram of produced material.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability of starting materials and the predictability of the manufacturing process. Because the new route relies on widely available aryl halides and ketones, the risk of supply disruption due to single-source dependency is drastically minimized. Additionally, the robustness of the reaction—tolerating lower purity intermediates without compromising final product quality—means that variations in raw material grades from different suppliers can be accommodated without re-validating the entire process. This flexibility allows procurement managers to negotiate better terms and switch suppliers more easily, ensuring uninterrupted production schedules even in volatile market conditions.
- Scalability and Environmental Compliance: From a scale-up perspective, the carbonylation step is highly amenable to continuous processing. The patent explicitly mentions that the reaction can be conducted in semi-batch or continuous flow reactors, which are ideal for ton-scale production. Continuous processing offers superior heat and mass transfer control, enhancing safety when handling carbon monoxide under pressure. Moreover, the simplified workup and reduced solvent usage contribute to a smaller environmental footprint. The ability to run the reaction at moderate temperatures (-20°C to 60°C) and the use of recyclable solvents like dichloromethane further align the process with modern green chemistry principles, facilitating easier regulatory approval and environmental compliance in stringent jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this carbonylation technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing a clear understanding of why this method is preferred for industrial applications.
Q: What are the primary limitations of conventional alkylation methods for phenylacetic acids?
A: Conventional methods involving double alkylation of phenylacetic acid esters often suffer from the formation of significant amounts of mono-alkylated intermediates and over-alkylated by-products. These impurities are structurally similar to the target molecule, making them extremely difficult to remove via standard crystallization, which drastically reduces overall yield and increases purification costs.
Q: Why is the carbonylation of tertiary alcohols considered superior for this synthesis?
A: The carbonylation route bypasses the selectivity issues of alkylation entirely. By constructing the carbon skeleton via Grignard addition first, the subsequent carbonylation step introduces the carboxylic acid group with high regioselectivity. This results in a cleaner reaction profile with fewer side products, allowing for simpler isolation procedures and higher final purity suitable for pharmaceutical applications.
Q: Is this carbonylation process suitable for large-scale industrial production?
A: Yes, the process is highly scalable. The patent explicitly notes that the carbonylation step can be conducted in semi-batch or continuous flow reactors. Furthermore, the starting materials (aryl halides and ketones) are generally more commercially available and cost-effective than the specialized alkylated phenylacetic acid precursors required by older methods, facilitating robust supply chain continuity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Phenylacetic Acid Derivatives Supplier
The technological breakthroughs detailed in CN1235856C underscore the complexity and sophistication required to manufacture high-value pharmaceutical intermediates efficiently. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring such advanced chemistries to life. Our facility is equipped with state-of-the-art high-pressure reactors capable of safely handling carbonylation reactions, alongside rigorous QC labs that ensure stringent purity specifications are met for every batch. We understand that the transition from lab-scale innovation to commercial reality requires a partner who can navigate both the chemical and logistical challenges of process scale-up.
We invite you to leverage our expertise to optimize your supply chain for NK-1 antagonist intermediates. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how adopting this carbonylation route can improve your margins. Please contact our technical procurement team today to request specific COA data for our phenylacetic acid derivatives and to discuss route feasibility assessments for your upcoming projects. Let us help you secure a competitive advantage through superior chemistry and reliable supply.
