Scaling Enantiopure Indole Carboxylic Acid Intermediates for Commercial Anti-Atherosclerotic Drug Production
The pharmaceutical industry's relentless pursuit of potent anti-atherosclerotic agents has placed a premium on the availability of high-purity chiral intermediates, specifically enantiomerically pure indole carboxylic acids and their reactive derivatives. Patent CN1330065A introduces a groundbreaking methodology that fundamentally shifts the paradigm from wasteful separation techniques to efficient, diastereoselective synthesis. This technology addresses the critical bottleneck of producing complex heterocyclic structures like cycloalkanol indole carboxylic acids and pyrimido[1,2-a]-indole carboxylic acids with exceptional stereochemical control. By leveraging a novel sequence involving chiral esterification followed by precise alpha-carbon substitution, manufacturers can now access these valuable building blocks with unprecedented efficiency. For R&D directors and procurement strategists, this represents a pivotal opportunity to secure a robust supply chain for next-generation cardiovascular therapeutics while drastically minimizing the environmental footprint associated with traditional resolution methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of enantiomerically pure cycloalkanol indole carboxylic acids relied heavily on the separation of racemic mixtures through diastereomeric resolution, often involving chromatographic separation or fractional crystallization. These conventional approaches are inherently inefficient, typically generating a 50% loss of the desired material in the form of the unwanted diastereomer, which is frequently difficult or impossible to recycle back into the process. Furthermore, the equipment requirements for large-scale chromatography are substantial, driving up capital expenditure and operational complexity. The reliance on expensive chiral resolving agents, which themselves often require complex synthetic routes to produce, further exacerbates the cost burden. This significant yield loss and high equipment dependency create a fragile supply chain that struggles to meet the rigorous demand schedules of modern pharmaceutical manufacturing, ultimately inflating the cost of goods sold for the final active pharmaceutical ingredient.
The Novel Approach
In stark contrast, the methodology disclosed in CN1330065A circumvents these inefficiencies by employing a diastereoselective alkylation strategy that favors the formation of the desired stereoisomer from the outset. The process begins with the esterification of toluene acetic acid using a chiral alcohol, setting the stage for highly selective subsequent transformations. Instead of discarding the minor diastereomer, the invention allows for the epimerization of the undesired isomer back into the desired configuration through appropriate choice of solvent and base, enabling its recovery via crystallization. This closed-loop approach effectively eliminates the theoretical 50% yield ceiling imposed by traditional resolution. The result is a streamlined workflow that delivers high-purity intermediates with significantly reduced waste generation, offering a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing and ensuring a more sustainable production lifecycle.
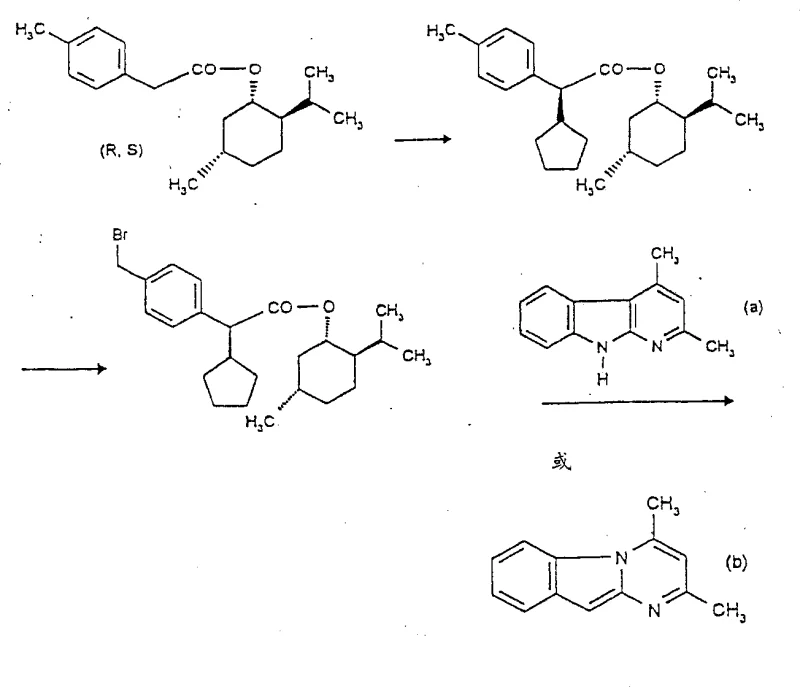
Mechanistic Insights into Diastereoselective Alkylation and Coupling
The core of this technological advancement lies in the precise control of stereochemistry during the alpha-carbon substitution phase. By utilizing a chiral auxiliary group, such as a menthyl or bornyl ester, the reaction environment becomes sterically biased, guiding the incoming alkyl halide to attack from a specific face of the enolate intermediate. This diastereoselective alkylation is conducted in inert solvents like dimethylformamide at controlled temperatures ranging from -20°C to +30°C, ensuring that the kinetic product dominates. The subsequent halogenation step, typically using agents like N-bromosuccinimide or bromine on the toluene methyl group, proceeds without racemization at the chiral center, preserving the optical integrity established in the earlier steps. This preservation of chirality is critical, as any erosion of enantiomeric excess at this stage would compromise the biological activity of the final anti-atherosclerotic drug candidate.
Furthermore, the coupling reaction with the indole or azaindole nucleus is designed to proceed under mild basic conditions that prevent epimerization at the sensitive alpha-position of the carboxylic acid function. The patent details the use of specific bases like potassium tert-butoxide or organic amines such as DBU to facilitate this nucleophilic substitution efficiently. The final hydrolysis or activation steps to generate the free acid or acid chloride are equally optimized to maintain stereochemical purity. For technical teams evaluating process feasibility, this mechanistic robustness means fewer purification cycles are required, directly translating to higher throughput and consistent quality. The ability to crystallize the intermediate diastereomers directly from the reaction mixture simplifies downstream processing, removing the need for resource-intensive chromatographic columns and aligning perfectly with the requirements for commercial scale-up of complex pharmaceutical intermediates.
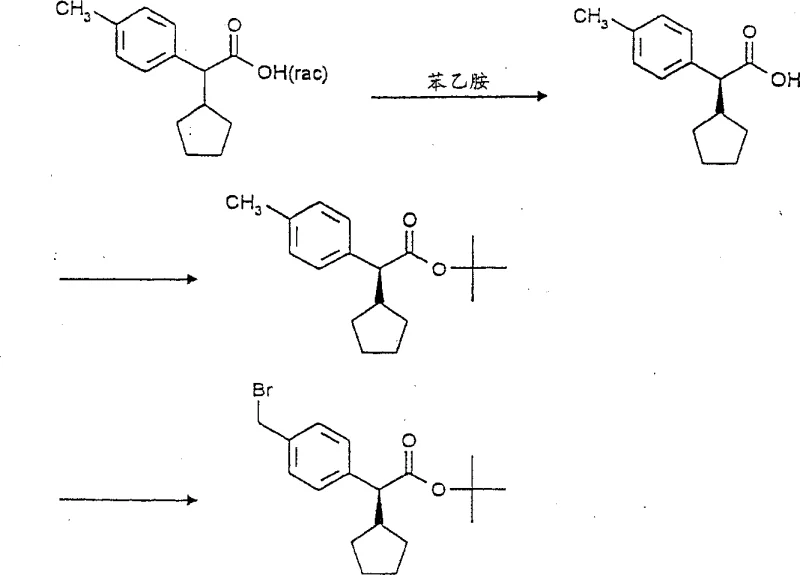
How to Synthesize Enantiopure Indole Carboxylic Acids Efficiently
Implementing this synthesis route requires a disciplined approach to reaction conditions and reagent quality to fully realize the benefits of the diastereoselective pathway. The process is anchored by the initial formation of the chiral ester, which dictates the stereochemical outcome of the entire sequence. Operators must ensure strict temperature control during the alkylation phase to maximize the diastereomeric ratio before crystallization. Following the isolation of the alkylated intermediate, the halogenation step must be monitored closely to prevent over-halogenation while ensuring complete conversion of the benzylic position. The detailed standardized synthesis steps见下方的指南 outline the specific molar ratios, solvent choices, and workup procedures necessary to achieve the high yields and purity levels reported in the patent examples. Adhering to these parameters is essential for replicating the success of the laboratory scale in a pilot or production environment.
- Esterify toluene acetic acid with a chiral alcohol to form a chiral ester precursor.
- Perform diastereoselective alkylation on the alpha-carbon followed by halogenation on the toluene group.
- React the halogenated intermediate with indole or azaindole derivatives to form the final enantiopure carboxylic acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers tangible strategic advantages beyond mere technical elegance. The elimination of chromatographic separation steps significantly reduces the consumption of silica gel and organic solvents, leading to substantial cost savings in raw materials and waste disposal. Moreover, the ability to recycle mother liquors through epimerization means that the effective yield of the process is decoupled from the theoretical limits of resolution, providing a buffer against raw material price volatility. This efficiency translates directly into a more competitive pricing structure for the final intermediate, allowing pharmaceutical companies to optimize their cost of goods sold without compromising on quality. The simplified equipment requirements also mean that production can be scaled up more rapidly, reducing lead time for high-purity chiral intermediates and ensuring continuity of supply even during periods of high market demand.
- Cost Reduction in Manufacturing: The process eliminates the need for expensive chiral chromatography and reduces the consumption of resolving agents by enabling the recycling of unwanted diastereomers. This fundamental shift in material efficiency removes the 50% yield penalty associated with traditional resolution, drastically lowering the cost per kilogram of the active intermediate. By avoiding complex separation technologies, capital investment is minimized, and operational expenses related to solvent recovery and waste treatment are significantly curtailed, delivering a leaner and more profitable manufacturing model.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like toluene acetic acid and common chiral alcohols ensures that the supply chain is not vulnerable to the bottlenecks often associated with exotic reagents. The robustness of the crystallization-based purification steps means that production is less susceptible to equipment failures or column breakthroughs that can halt chromatographic processes. This stability allows for more accurate forecasting and inventory management, ensuring that downstream drug formulation teams receive their materials on schedule, thereby safeguarding the overall project timeline for new drug approvals.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing standard reactor types and avoiding high-pressure or cryogenic conditions that are difficult to manage on a multi-ton scale. The reduction in solvent usage and the ability to recycle byproducts align with increasingly stringent environmental regulations regarding hazardous waste discharge. This eco-friendly profile not only reduces regulatory risk but also enhances the corporate sustainability metrics of the manufacturing partner, making it an attractive option for global pharmaceutical companies committed to green chemistry principles.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific beneficial effects and background technical pain points outlined in the patent documentation. Understanding these nuances is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the objective capabilities of the method as described in CN1330065A, providing a clear picture of what can be expected in terms of performance and operational requirements.
Q: How does this process improve yield compared to conventional chromatographic separation?
A: Conventional methods often lose 50% of material as the wrong diastereomer. This patent utilizes epimerization to recycle mother liquor, significantly boosting overall yield and reducing waste.
Q: What ensures the enantiomeric purity of the final intermediate?
A: The process employs diastereoselective alkylation using chiral esters and specific crystallization conditions, achieving high ee values without needing complex chiral chromatography.
Q: Is this synthesis route scalable for industrial production?
A: Yes, the method avoids equipment-intensive chromatography and uses standard crystallization and halogenation steps, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enantiopure Indole Carboxylic Acids Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent to production requires more than just chemical knowledge; it demands engineering excellence and a commitment to quality. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate diastereoselective steps of this process are executed with precision. Our facilities are equipped with stringent purity specifications and rigorous QC labs capable of verifying enantiomeric excess and impurity profiles at every stage of the synthesis. We understand that for anti-atherosclerotic drugs, the consistency of the intermediate is paramount, and our quality management systems are designed to deliver that reliability consistently.
We invite you to collaborate with us to leverage this advanced technology for your pipeline. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that quantifies the potential efficiencies of adopting this route for your specific project needs. Please contact us to request specific COA data and route feasibility assessments tailored to your volume requirements. By partnering with us, you gain access to a supply chain that is not only cost-effective but also technically superior, positioning your organization for success in the competitive landscape of cardiovascular therapeutics.
