Industrial Scale Production of 4-Aminotetrahydropyran via Catalytic Decomposition
The pharmaceutical and agrochemical industries continuously demand efficient, scalable routes for synthesizing complex heterocyclic amines, specifically 4-aminotetrahydropyran derivatives which serve as critical building blocks for various bioactive molecules. A pivotal advancement in this domain is detailed in patent CN1823051A, which discloses a robust manufacturing method for 4-aminotetrahydropyran compounds and their acid salts. Unlike traditional methods that rely on cumbersome reductive amination processes, this innovation utilizes a catalytic decomposition of 4-hydrazinotetrahydropyran intermediates. This approach not only streamlines the synthetic pathway but also addresses significant pain points regarding yield optimization and operational simplicity. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding this mechanistic shift is crucial for securing supply chains and reducing overall manufacturing costs in API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-aminotetrahydropyran has been plagued by inefficiencies inherent to reductive amination strategies. Conventional protocols typically involve the reaction of tetrahydropyran-4-one with ammonium acetate and sodium cyanoborohydride in ethanol. As documented in prior art such as Japanese Patent Application Laid-Open No. H11-510180, this method suffers from critically low yields, often reported around 12%, necessitating the use of far excess ammonia sources to drive the equilibrium. Furthermore, the reaction system is chemically complex, requiring rigorous purification steps to remove toxic cyanide byproducts and unreacted ketones. These factors contribute to substantial waste generation and increased processing time, making the conventional route economically unviable for large-scale commercial operations where cost reduction in API manufacturing is a primary objective.
The Novel Approach
In stark contrast, the novel methodology presented in the patent introduces a streamlined two-step sequence that bypasses the limitations of direct reductive amination. The process begins with the nucleophilic substitution of a 4-substituted-tetrahydropyran derivative with hydrazine to form a stable 4-hydrazinotetrahydropyran intermediate. This intermediate is then subjected to a catalytic decomposition reaction in the presence of hydrogen and a suitable catalyst. 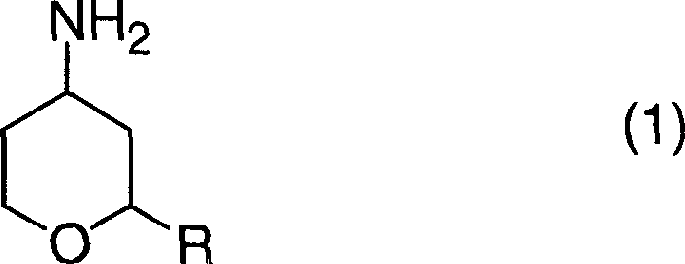 This transformation effectively cleaves the nitrogen-nitrogen bond to yield the target primary amine. By shifting the synthetic strategy to hydrazine decomposition, the process eliminates the need for hazardous reducing agents like sodium cyanoborohydride and allows for milder reaction conditions, thereby enhancing both safety profiles and overall throughput for high-purity pharmaceutical intermediates.
This transformation effectively cleaves the nitrogen-nitrogen bond to yield the target primary amine. By shifting the synthetic strategy to hydrazine decomposition, the process eliminates the need for hazardous reducing agents like sodium cyanoborohydride and allows for milder reaction conditions, thereby enhancing both safety profiles and overall throughput for high-purity pharmaceutical intermediates.
Mechanistic Insights into Catalytic Hydrazine Decomposition
The core of this technological breakthrough lies in the catalytic decomposition mechanism of the hydrazino group. The reaction proceeds efficiently in the presence of specific catalysts selected from Raney nickel, noble metals such as palladium or platinum on carbon, and metal oxides like copper(I) or copper(II) oxide. The mechanism involves the adsorption of the hydrazine intermediate onto the catalyst surface, followed by the cleavage of the N-N bond under a hydrogen atmosphere. Experimental data indicates that reaction temperatures between 50°C and 100°C and pressures ranging from 0.1 to 5 MPa are optimal for driving this conversion to completion. The choice of solvent also plays a pivotal role, with water and lower alcohols like methanol and ethanol proving most effective in solubilizing the reactants while maintaining catalyst activity. This precise control over reaction parameters ensures high conversion rates and minimizes the formation of undesired side products.
Furthermore, the patent highlights the versatility of the leaving group in the precursor step, which directly influences the purity of the final amine. The use of sulfonate leaving groups, such as mesylates or tosylates, facilitates a clean nucleophilic attack by hydrazine. 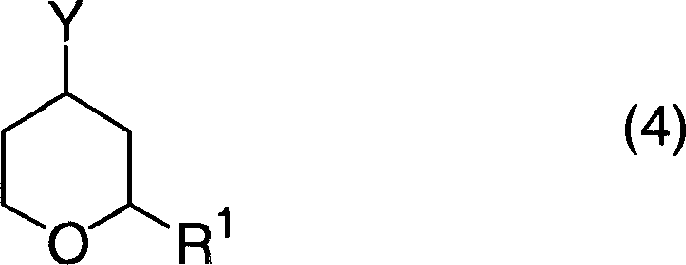 This precursor chemistry is particularly advantageous because 2-substituted tetrahydropyran-4-sulfonates can be synthesized efficiently from readily available 3-buten-1-ol and aldehydes. This upstream flexibility allows manufacturers to tailor the substitution pattern at the 2-position of the pyran ring without compromising the efficiency of the subsequent amination step. Consequently, impurity profiles are tightly controlled, as the robust nature of the sulfonate intermediate prevents premature degradation, ensuring that the final 4-aminotetrahydropyran product meets stringent quality specifications required for regulatory compliance in drug substance manufacturing.
This precursor chemistry is particularly advantageous because 2-substituted tetrahydropyran-4-sulfonates can be synthesized efficiently from readily available 3-buten-1-ol and aldehydes. This upstream flexibility allows manufacturers to tailor the substitution pattern at the 2-position of the pyran ring without compromising the efficiency of the subsequent amination step. Consequently, impurity profiles are tightly controlled, as the robust nature of the sulfonate intermediate prevents premature degradation, ensuring that the final 4-aminotetrahydropyran product meets stringent quality specifications required for regulatory compliance in drug substance manufacturing.
How to Synthesize 4-Aminotetrahydropyran Efficiently
Implementing this synthesis route requires careful attention to the preparation of the hydrazine intermediate and the subsequent catalytic conditions. The process is designed to be telescoped or performed in discrete steps depending on the facility's capabilities. The initial substitution reaction generates the hydrazino compound in high purity, which can be isolated or used directly in the next stage. Detailed standard operating procedures for temperature ramping, catalyst loading, and workup protocols are essential to replicate the high yields reported in the patent examples. Following these standardized steps ensures reproducibility and safety during the scale-up phase.
- React 4-substituted-tetrahydropyran derivatives with hydrazine hydrate in alcohol solvents at 70-80°C to form 4-hydrazinotetrahydropyran intermediates.
- Subject the crude hydrazine intermediate to decomposition using Raney nickel or noble metal catalysts under hydrogen atmosphere at 50-100°C.
- Isolate the final 4-aminotetrahydropyran product through filtration, concentration, and salt formation with acids like hydrochloric acid.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic decomposition technology offers transformative benefits beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the supply chain for raw materials. By eliminating the dependency on specialized and hazardous reducing agents like sodium cyanoborohydride, manufacturers can source common, commodity-grade chemicals such as hydrazine hydrate and Raney nickel. This shift significantly reduces raw material costs and mitigates the regulatory burdens associated with handling toxic cyanide species. Moreover, the simplified workup procedure, which often involves simple filtration and concentration rather than complex chromatographic separations, leads to substantial cost savings in processing time and solvent consumption, directly impacting the bottom line of chemical manufacturing budgets.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric reducing agents and the ability to use cost-effective catalysts like Raney nickel drastically lowers the variable cost per kilogram of the final product. Additionally, the higher yields achieved through this method mean less starting material is wasted, further optimizing the cost structure. The process operates under relatively mild conditions, reducing energy consumption for heating and cooling compared to more vigorous traditional methods. These cumulative efficiencies result in a highly competitive pricing structure for the final 4-aminotetrahydropyran intermediates without compromising on quality standards.
- Enhanced Supply Chain Reliability: Utilizing widely available catalysts and solvents ensures that production is not bottlenecked by the scarcity of niche reagents. The robustness of the reaction conditions allows for flexible scheduling and reduced risk of batch failures due to sensitive reagent degradation. This reliability is critical for maintaining continuous supply to downstream API manufacturers who depend on just-in-time delivery models. By securing a manufacturing route that relies on stable, commoditized inputs, companies can better forecast production timelines and reduce lead time for high-purity intermediates, ensuring consistent availability for global markets.
- Scalability and Environmental Compliance: The process has been successfully demonstrated on a 20-liter scale in the patent literature, indicating strong potential for metric-ton scale-up. The use of aqueous or alcoholic solvents aligns well with green chemistry principles, reducing the environmental footprint associated with halogenated solvent disposal. Furthermore, the absence of cyanide waste streams simplifies wastewater treatment and regulatory compliance, making this route more sustainable and easier to permit in strict environmental jurisdictions. This scalability ensures that the technology can meet growing market demands for complex heterocyclic amines in the pharmaceutical and agrochemical sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details helps stakeholders make informed decisions regarding technology transfer and vendor selection.
Q: What are the advantages of the hydrazine decomposition route over reductive amination?
A: The hydrazine decomposition route avoids the use of toxic cyanoborohydride reducing agents and excessive ammonia sources, resulting in simpler workup procedures and significantly higher isolated yields compared to traditional reductive amination methods which often suffer from yields as low as 12%.
Q: Which catalysts are most effective for this decomposition reaction?
A: The patent specifies that Raney nickel, noble metal catalysts such as palladium on carbon (Pd/C) or platinum on carbon (Pt/C), and metal oxides like copper oxide are highly effective. Raney nickel is particularly noted for its cost-effectiveness in large-scale applications.
Q: Can this process be scaled for commercial production?
A: Yes, the process has been demonstrated on a 20-liter scale in the patent examples, utilizing standard reactor equipment and mild reaction conditions (50-100°C, 0.1-5 MPa), indicating strong feasibility for metric-ton scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Aminotetrahydropyran Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of 4-aminotetrahydropyran meets the highest international standards. Our commitment to excellence extends beyond mere supply; we act as a strategic partner dedicated to optimizing your supply chain through advanced manufacturing technologies.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic decomposition route can benefit your specific projects. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this superior manufacturing method. We encourage potential partners to contact us for specific COA data and route feasibility assessments tailored to your unique requirements. Let us collaborate to enhance the efficiency and reliability of your pharmaceutical intermediate supply chain today.
