Advanced Manufacturing of High-Purity Epoxy Triazole Intermediates for Antifungal Agents
Introduction to Patent CN1298711C and Technological Breakthroughs
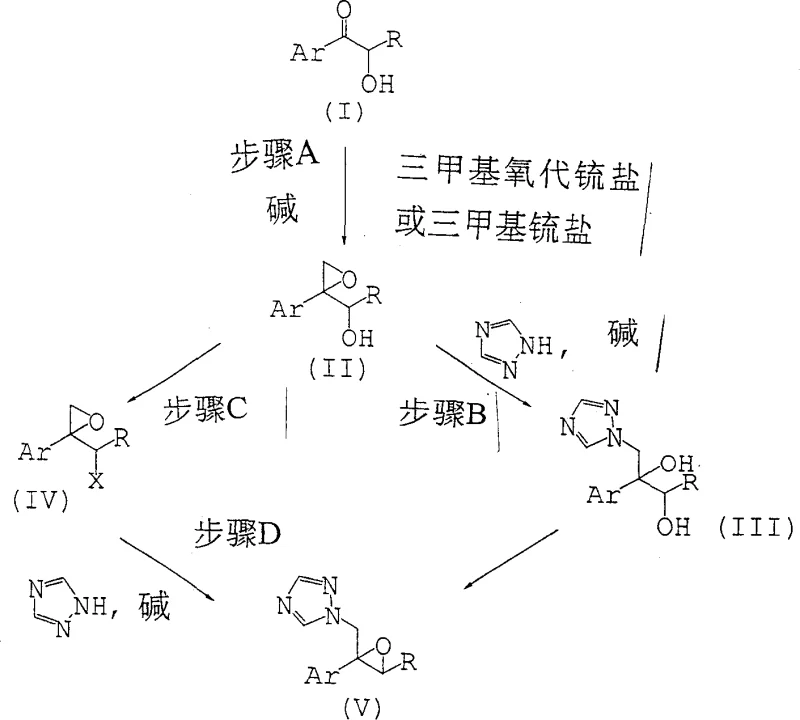
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing complex intermediates, particularly for broad-spectrum antifungal agents. Patent CN1298711C, titled Process for preparing epoxy triazole derivatives and intermediates used therefor, represents a significant paradigm shift in the manufacturing of these critical building blocks. Historically, the synthesis of epoxy triazole derivatives, which serve as pivotal precursors for triazole antifungal drugs, relied heavily on multi-step sequences involving the protection and subsequent deprotection of sensitive hydroxyl functionalities. This patent discloses a novel methodology that surprisingly allows for the direct epoxidation of alpha-hydroxy ketones without the need for prior hydroxyl protection. By utilizing trimethyloxosulfonium salts or trimethylsulfonium salts in the presence of a base, the process achieves high yields and exceptional diastereoselectivity. This technological advancement addresses long-standing challenges in process chemistry, offering a more atom-economical and operationally simple route that is highly attractive for industrial scale-up.
For R&D directors and process chemists, the implications of this invention are profound, as it simplifies the synthetic tree and reduces the potential for impurity generation associated with extra synthetic steps. The core innovation lies in the stability and reactivity of the unprotected alpha-hydroxy ketone substrate under the specific reaction conditions employed. Contrary to previous beliefs that such substrates were too unstable or unreactive for direct epoxidation, the data presented in the patent confirms that high-quality epoxy intermediates can be produced economically. This report analyzes the technical merits of this process, comparing it against conventional methodologies to highlight the tangible benefits for procurement and supply chain optimization. As a reliable pharmaceutical intermediate supplier, understanding these mechanistic nuances is essential for delivering cost-effective solutions to our global partners.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for epoxy triazole derivatives, as documented in prior art such as JP-A-4-356471 and JP-A-5-230038, typically necessitate the use of protecting groups to mask the hydroxyl functionality of the starting alpha-hydroxy ketone. The most common approach involves converting the hydroxyl group into a tetrahydropyranyl (THP) ether before subjecting the molecule to epoxidation conditions. While chemically feasible, this strategy introduces significant inefficiencies into the manufacturing process. The introduction of a protecting group requires stoichiometric amounts of protecting reagents, such as dihydropyran or THP derivatives, which adds directly to the raw material costs. Furthermore, the protection step itself is an additional unit operation that consumes time, solvent, and energy, thereby extending the overall production cycle time. Perhaps most critically, the conventional methods suffer from suboptimal stereocontrol, often generating approximately 20% of unwanted diastereomeric by-products. These stereoisomers are not only useless for the final antifungal application but also complicate the purification process, leading to yield losses and increased waste generation.
The Novel Approach
In stark contrast to the cumbersome traditional workflows, the novel approach disclosed in CN1298711C eliminates the protection-deprotection sequence entirely. The inventors discovered that alpha-hydroxy ketones, specifically those substituted with halogenated phenyl groups like 2,4-difluorophenyl or 2,5-difluorophenyl, can undergo direct epoxidation using sulfur ylides generated in situ. This breakthrough allows the reaction to proceed smoothly even with the free hydroxyl group present, challenging the previous assumption that such functionality would interfere with the epoxidation reagent. By bypassing the protection steps, the process drastically reduces the number of synthetic operations, which translates to lower operational expenditures and a smaller environmental footprint. Moreover, the new method demonstrates superior diastereoselectivity, with experimental examples showing ratios of desired to undesired isomers ranging from 12:1 to as high as 25:1. This enhanced selectivity ensures a cleaner crude product profile, simplifying downstream isolation and crystallization steps. For manufacturing teams, this means a more robust process with higher throughput and reduced risk of batch failure due to complex impurity profiles.
Mechanistic Insights into Sulfur Ylide-Mediated Epoxidation
The core chemical transformation in this patented process is the Corey-Chaykovsky epoxidation, adapted here for unprotected substrates. The reaction initiates with the generation of a sulfur ylide, typically dimethylsulfoxonium methylide or dimethylsulfonium methylide, by treating a trimethyloxosulfonium salt or trimethylsulfonium salt with a strong base such as sodium hydride or potassium hydride. This ylide acts as a nucleophile, attacking the carbonyl carbon of the alpha-hydroxy ketone (Compound I). The presence of the adjacent free hydroxyl group could theoretically lead to side reactions or protonation of the ylide; however, the specific reaction conditions, including solvent choice (often DMSO or mixtures with THF) and temperature control, mitigate these risks. The betaine intermediate formed collapses to expel the sulfide species and close the epoxide ring, yielding the epoxy alcohol (Compound II). The high diastereoselectivity observed suggests that the free hydroxyl group may actually participate in directing the approach of the ylide through hydrogen bonding or chelation effects, although the exact transition state geometry remains a subject of detailed mechanistic study. This level of control is crucial for producing the specific stereoisomers required for biological activity in the final antifungal drug.
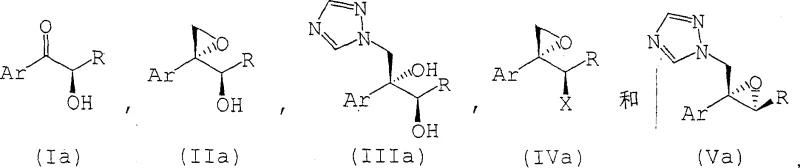
Following the formation of the epoxy alcohol, the process offers flexibility in the subsequent functionalization to introduce the triazole moiety. One pathway involves converting the secondary hydroxyl group of the epoxide into a good leaving group, such as a mesylate or tosylate (Compound IV), using sulfonyl halides or anhydrides in the presence of a tertiary amine base. This activated intermediate then undergoes nucleophilic substitution with 1,2,4-triazole. Alternatively, the patent describes conditions where the triazole ring can be introduced more directly. The choice of base in these subsequent steps is critical; bases like sodium hydride, potassium carbonate, or alkoxides are employed to deprotonate the triazole, generating the nucleophilic triazolyl anion. The reaction parameters, including temperature ranges from -20°C to 150°C depending on the specific step, are optimized to minimize ring-opening of the sensitive epoxide while ensuring complete conversion of the leaving group. Understanding these mechanistic details allows process engineers to fine-tune reaction conditions for maximum efficiency and safety during commercial scale-up, ensuring that the high purity specifications required for pharmaceutical intermediates are consistently met.
How to Synthesize Epoxy Triazole Derivatives Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction monitoring to maintain the high diastereoselectivity reported in the patent. The process begins with the preparation of the sulfur ylide under inert atmosphere, followed by the controlled addition of the alpha-hydroxy ketone substrate. Maintaining the correct stoichiometry between the ylide precursor and the base is essential to prevent excess base from causing decomposition or isomerization of the product. Following the epoxidation, the workup procedure typically involves aqueous quenching and extraction, often allowing the crude epoxy alcohol to be used directly in the next step without rigorous purification, further enhancing process efficiency. The subsequent activation and triazole coupling steps require precise temperature control to avoid epoxide ring opening by hydroxide or other nucleophiles present in the medium. Detailed standard operating procedures regarding solvent drying, reagent addition rates, and crystallization protocols are vital for reproducibility. For a comprehensive guide on the specific experimental conditions, reagent grades, and safety precautions necessary for executing this synthesis, please refer to the standardized technical documentation provided below.
- React an alpha-hydroxy ketone (Compound I) with a trimethyloxosulfonium salt in the presence of a base to form the epoxy alcohol (Compound II).
- Convert the hydroxyl group of Compound II into a leaving group (such as a sulfonate) to yield Compound IV, or react directly with triazole.
- Perform nucleophilic substitution with 1,2,4-triazole in the presence of a base to obtain the final epoxy triazole derivative (Compound V).
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this protection-free synthesis route offers substantial advantages for cost reduction in pharmaceutical intermediate manufacturing. The most immediate impact is the elimination of costs associated with protecting group chemistry. By removing the need for tetrahydropyranyl derivatives and the reagents required for their installation and removal, the bill of materials is significantly streamlined. This reduction in raw material complexity not only lowers the direct material costs but also reduces the inventory burden on the supply chain. Furthermore, the reduction in the number of synthetic steps leads to a shorter overall manufacturing cycle time. Fewer unit operations mean less equipment occupancy time, lower utility consumption for heating and cooling, and reduced labor requirements per kilogram of finished product. These operational efficiencies compound to deliver a more competitive pricing structure for the final epoxy triazole derivative, making it an economically attractive option for large-scale production campaigns.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the atom economy gained by skipping the protection and deprotection stages. In traditional synthesis, these steps can account for a significant portion of the total processing cost due to the additional reagents, solvents, and waste disposal fees involved. By executing the epoxidation directly on the unprotected alpha-hydroxy ketone, the process avoids the purchase of expensive protecting group reagents and the acids or catalysts needed for their removal. Additionally, the improved diastereoselectivity reduces the loss of material during purification, effectively increasing the overall yield of the usable isomer. This higher effective yield means that less starting material is required to produce the same amount of active intermediate, further driving down the cost of goods sold (COGS) and improving the margin profile for the final drug product.
- Enhanced Supply Chain Reliability: Simplifying the synthetic route inherently enhances supply chain resilience. A process with fewer steps has fewer potential points of failure, reducing the risk of batch delays or failures that can disrupt supply continuity. The reagents used in this novel method, such as trimethyloxosulfonium salts and common inorganic bases, are commodity chemicals with stable and reliable global supply chains. This contrasts with specialized protecting group reagents which may have longer lead times or limited supplier bases. Moreover, the robustness of the reaction conditions allows for greater flexibility in manufacturing scheduling. The ability to telescope steps or use crude intermediates without extensive purification accelerates the throughput, enabling manufacturers to respond more quickly to fluctuations in market demand. This agility is crucial for maintaining consistent supply to downstream API manufacturers, especially in the event of urgent capacity requirements.
- Scalability and Environmental Compliance: The environmental benefits of this process align well with modern green chemistry principles and regulatory expectations. By eliminating two entire synthetic steps (protection and deprotection), the process generates significantly less chemical waste, including spent solvents and aqueous waste streams from workups. This reduction in waste volume lowers the costs associated with waste treatment and disposal, which are increasingly stringent and expensive in many jurisdictions. The improved selectivity also means less chromatographic purification is needed, reducing the consumption of silica gel and elution solvents. From a scalability standpoint, the reaction conditions are amenable to large-scale batch processing, with exotherms that can be managed through controlled addition rates. The use of common solvents like DMSO, THF, and toluene facilitates solvent recovery and recycling programs, further minimizing the environmental footprint and ensuring compliance with evolving environmental, health, and safety (EHS) standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this epoxy triazole synthesis technology. These insights are derived directly from the experimental data and claims within patent CN1298711C, providing clarity on the feasibility and advantages of the method. Understanding these details helps stakeholders evaluate the suitability of this route for their specific development programs. The answers focus on the practical aspects of reaction performance, impurity control, and the strategic benefits of adopting this streamlined approach over legacy methods.
Q: Why is the direct epoxidation of unprotected alpha-hydroxy ketones considered difficult?
A: Traditionally, the free hydroxyl group in alpha-hydroxy ketones was believed to interfere with epoxidation reagents or cause instability, necessitating protection steps like tetrahydropyranyl (THP) groups. However, this patent demonstrates that under specific basic conditions with sulfur ylides, the reaction proceeds efficiently with high diastereoselectivity.
Q: How does this process improve diastereoselectivity compared to conventional methods?
A: Conventional methods using protected substrates often yield significant amounts of unwanted stereoisomers (up to 20%). The novel direct epoxidation method described in CN1298711C achieves significantly higher diastereomeric ratios, such as 12:1 or even 25:1, reducing the burden of downstream purification.
Q: What are the primary cost drivers eliminated in this new synthesis route?
A: The primary cost savings come from eliminating the reagents and unit operations associated with hydroxyl protection and deprotection. This removes the need for expensive protecting group derivatives and the additional reaction steps required to install and remove them, streamlining the overall manufacturing timeline.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epoxy Triazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value pharmaceutical intermediates. Our technical team has thoroughly analyzed the innovations presented in CN1298711C and possesses the expertise to translate this laboratory-scale breakthrough into robust commercial manufacturing processes. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this protection-free epoxidation strategy are fully realized at an industrial level. Our facilities are equipped with state-of-the-art reactors capable of handling the specific solvent systems and temperature profiles required for sulfur ylide chemistry, while our stringent purity specifications and rigorous QC labs guarantee that every batch meets the exacting standards required for antifungal drug synthesis. We are committed to delivering high-purity epoxy triazole derivatives that support your R&D and commercial manufacturing needs with consistency and reliability.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Our technical procurement team is ready to discuss how we can optimize the production of these intermediates to reduce your overall project costs and timelines. Please contact us today to request specific COA data, route feasibility assessments, and a comprehensive proposal for the commercial supply of epoxy triazole derivatives. Let us help you secure a sustainable and cost-effective source for this vital component of your antifungal portfolio.
