Advanced Direct Epoxidation Technology for High-Purity Antifungal Intermediates and Commercial Scale-Up
Introduction to Breakthrough Epoxidation Technology
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing complex heterocyclic intermediates, particularly those serving as precursors for potent antifungal agents. Patent CN1662518A introduces a transformative methodology for the preparation of epoxy triazole derivatives, addressing long-standing inefficiencies in the construction of the oxirane ring adjacent to chiral centers. This technology fundamentally shifts the paradigm from traditional multi-step protection strategies to a streamlined direct epoxidation approach. By leveraging the reactivity of trimethyloxosulfonium salts with unprotected alpha-hydroxy ketones, the process achieves superior diastereoselectivity and operational simplicity. For R&D directors and procurement specialists, this represents a critical opportunity to optimize supply chains for high-value pharmaceutical intermediates. The ability to bypass unstable intermediate states not only enhances yield but also ensures consistent optical purity, a parameter vital for regulatory compliance in active pharmaceutical ingredient (API) manufacturing.
This technical insight report analyzes the mechanistic advantages and commercial implications of the disclosed process. The core innovation lies in the surprising stability and reactivity of the unprotected hydroxy-ketone substrate under specific basic conditions, challenging previous assumptions that such substrates were too chemically unstable for direct transformation. This discovery allows for the economic production of high-quality epoxy triazole derivatives, which are indispensable building blocks in the synthesis of next-generation triazole antifungals. As global demand for effective antifungal therapies rises, the capacity to produce these intermediates with reduced environmental footprint and lower cost of goods sold becomes a strategic asset for any forward-thinking chemical enterprise.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
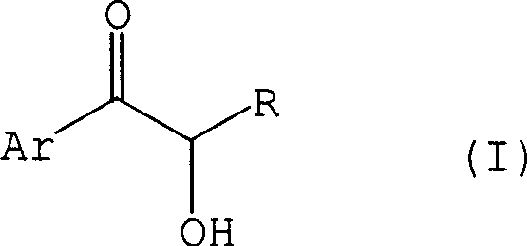
Historically, the synthesis of epoxy triazole derivatives relied heavily on the use of protecting groups to mask the reactivity of the alpha-hydroxyl functionality during the epoxidation step. As illustrated in the reaction schemes of prior art, compounds such as formula (VII) required the introduction of a tetrahydropyranyl (THP) group to prevent side reactions and ensure stereocontrol. However, this conventional approach is fraught with industrial disadvantages. The introduction of the protecting group necessitates the use of equimolar amounts of THP derivatives, adding significant material costs and generating stoichiometric waste. Furthermore, the protection step itself adds a distinct unit operation to the manufacturing timeline, increasing the overall lead time and complicating the process flow. Perhaps most critically, the conventional method suffers from poor diastereoselectivity, often yielding approximately 20% of unwanted stereoisomers that exist as diastereomers. These impurities are difficult to separate and cannot be utilized as intermediates for the final triazole compounds, effectively reducing the overall mass efficiency of the process.
The Novel Approach
In stark contrast, the novel approach disclosed in CN1662518A eliminates the need for hydroxyl protection entirely. The inventors discovered that alpha-hydroxy-ketone derivatives, previously deemed too unstable for direct epoxidation without hydrocarbyl protection, could be successfully converted into epoxy alcohols using trimethyloxosulfonium salts in the presence of a base. This breakthrough simplifies the synthetic route by removing both the protection and deprotection steps, thereby drastically reducing the number of operational stages. The result is a process that is not only shorter but also significantly more selective. Experimental data within the patent indicates that this direct method can achieve diastereomeric ratios as high as 12:1 or even 20:1, vastly outperforming the 4:1 ratio typical of the protected route. This improvement in selectivity translates directly to higher purity profiles and reduced downstream purification costs, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations seeking to maximize throughput.
Mechanistic Insights into Sulfur Ylide-Mediated Epoxidation
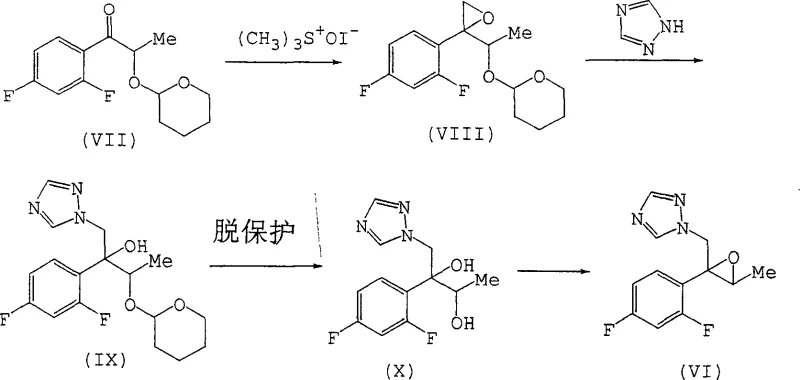
The core of this technological advancement relies on the precise generation and reaction of sulfur ylides. In the presence of a strong base such as potassium hydride or sodium hydride, trimethyloxosulfonium salts generate a reactive sulfur ylide species in situ. This ylide acts as a nucleophile, attacking the carbonyl carbon of the alpha-hydroxy ketone (Compound I). The presence of the free hydroxyl group, rather than hindering the reaction as previously thought, appears to participate in a coordinated transition state that favors the formation of the desired stereoisomer. The choice of solvent plays a pivotal role in this mechanism; polar aprotic solvents like dimethyl sulfoxide (DMSO) or mixtures with tetrahydrofuran (THF) stabilize the ylide and facilitate the necessary ion pairing. The reaction temperature is carefully controlled, typically ranging from -20°C to 60°C, to balance reaction kinetics with stereochemical integrity. This careful modulation of reaction parameters ensures that the epoxide ring closes with high fidelity, preserving the optical activity of the starting material.
From an impurity control perspective, the mechanism inherently suppresses the formation of diastereomeric by-products. In the conventional protected route, the bulkiness of the THP group can induce steric clashes that lead to non-selective ring closure. By removing this bulky group, the transition state becomes less congested, allowing for a more defined trajectory of the ylide attack. Furthermore, the process avoids the acidic conditions often required for deprotection, which can sometimes lead to epoxide ring opening or racemization. The resulting epoxy alcohol (Compound II) retains the optical purity of the starting ketone, as evidenced by the maintenance of 100% enantiomeric excess (e.e.) in various examples. This robustness against racemization is crucial for R&D teams focused on developing chiral drugs, where even minor deviations in optical purity can disqualify a batch from clinical use. The ability to scale this mechanism while maintaining such tight control over stereochemistry underscores its value for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Epoxy Triazole Derivatives Efficiently
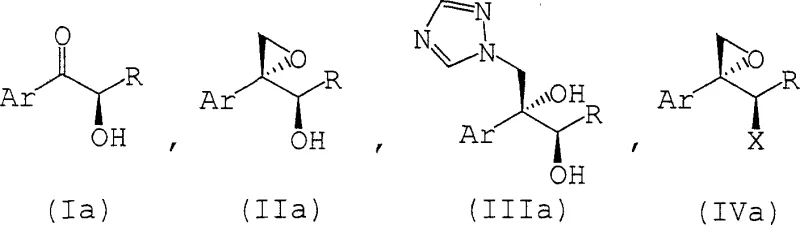
The synthesis of these high-value intermediates follows a logical progression designed for industrial robustness. The process begins with the preparation of the key epoxy alcohol intermediate via the direct epoxidation method described above. Once Compound (II) is secured, it serves as a versatile platform for further functionalization. The subsequent steps involve converting the remaining hydroxyl group into a good leaving group, such as a mesylate or tosylate, followed by nucleophilic substitution with 1,2,4-triazole. This sequence ensures that the triazole ring is installed with high regioselectivity. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, solvent choices, and temperature profiles required to replicate the high yields and selectivity reported in the patent data. Adhering to these parameters is essential for achieving the commercial viability of the route.
- React compound (I), an alpha-hydroxy ketone, with a trimethyloxosulfonium salt or trimethylsulfonium salt in the presence of a base such as potassium hydride or sodium hydride to form the epoxy alcohol compound (II).
- Convert the hydroxyl group of compound (II) into a leaving group, such as a sulfonate ester, to yield compound (IV) using a sulfonyl halide and a tertiary amine base.
- React compound (IV) with 1,2,4-triazole in the presence of a base to substitute the leaving group, yielding the final high-quality epoxy triazole derivative (V).
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this direct epoxidation technology offers tangible strategic benefits beyond mere technical elegance. The most immediate impact is seen in the reduction of raw material complexity. By eliminating the requirement for tetrahydropyranyl derivatives and the associated deprotection reagents, the bill of materials is simplified, reducing the risk of supply disruptions for niche protecting group chemicals. This simplification also leads to substantial cost savings in manufacturing, as fewer reagents need to be sourced, stored, and handled. The removal of two entire synthetic steps (protection and deprotection) significantly shortens the production cycle time, allowing for faster turnover of inventory and improved responsiveness to market demand fluctuations. This agility is a key component of cost reduction in pharmaceutical intermediate manufacturing, where time-to-market is often as critical as the price per kilogram.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps removes the associated costs of reagents, solvents, and energy consumption for those specific unit operations. Without the need to purchase equimolar protecting groups or spend resources on their removal, the overall cost of goods sold is significantly optimized. Additionally, the improved diastereoselectivity reduces the loss of material to unusable stereoisomers, effectively increasing the yield of usable product from the same amount of starting material. This material efficiency translates directly into lower variable costs per unit of output, enhancing the margin profile for the final API.
- Enhanced Supply Chain Reliability: Relying on fewer synthetic steps inherently reduces the points of failure in the supply chain. Each chemical transformation carries a risk of batch failure or delay; by compressing the synthesis from five or six steps down to three or four, the probability of successful batch completion increases. Furthermore, the reagents used in this novel process, such as trimethyloxosulfonium salts and common bases like sodium hydride, are widely available commodity chemicals. This contrasts with specialized protecting group reagents that may have limited suppliers. Consequently, reducing lead time for high-purity pharmaceutical intermediates becomes achievable through a more resilient and diversified sourcing strategy.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents like toluene and DMSO that are manageable in large-scale reactors with appropriate safety controls. The reduction in step count also means a reduction in total waste generation, aligning with green chemistry principles. Less solvent is consumed overall, and fewer aqueous washes are required for workup, decreasing the load on wastewater treatment facilities. This environmental efficiency not only lowers disposal costs but also ensures compliance with increasingly stringent environmental regulations, securing the long-term viability of the manufacturing site.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this epoxidation technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing production portfolios. The focus remains on practical applicability, purity outcomes, and the specific conditions required to unlock the full potential of the direct epoxidation method.
Q: How does this new process improve diastereoselectivity compared to conventional methods?
A: The patented process eliminates the tetrahydropyranyl protection step, which traditionally resulted in approximately 20% by-produced stereoisomers. By directly epoxidizing the unprotected alpha-hydroxy ketone under optimized basic conditions, the diastereoselectivity is significantly improved, often achieving ratios of 12:1 or higher, thereby reducing the burden of downstream purification.
Q: What are the primary cost drivers eliminated in this synthesis route?
A: The primary cost reduction comes from removing the need for equimolar amounts of tetrahydropyranyl derivatives for protection and the subsequent reagents required for deprotection. Additionally, the reduction in unit operations decreases solvent consumption, energy usage for heating and cooling cycles, and labor costs associated with additional isolation steps.
Q: Is this process suitable for large-scale commercial production of antifungal intermediates?
A: Yes, the process utilizes industrially common solvents such as dimethyl sulfoxide, toluene, and tetrahydrofuran, and employs robust reagents like trimethyloxosulfonium salts. The elimination of sensitive protection groups enhances process stability, making it highly scalable from kilogram to multi-ton annual production capacities while maintaining stringent purity specifications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Epoxy Triazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving antifungal medications. Our technical team has thoroughly analyzed the methodologies described in CN1662518A and possesses the expertise to implement this direct epoxidation technology at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to manufacturing plant is seamless. Our facilities are equipped with rigorous QC labs capable of monitoring the stringent purity specifications required for chiral intermediates, including advanced HPLC methods for diastereomeric ratio analysis. We are committed to delivering high-purity epoxy triazole derivatives that meet the exacting standards of the global pharmaceutical industry.
We invite potential partners to engage with our technical procurement team to discuss how this innovative process can be tailored to your specific supply chain needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this unprotected route. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials against your internal benchmarks. Together, we can drive down costs and accelerate the availability of essential antifungal therapies to patients worldwide, leveraging our position as a trusted partner in fine chemical manufacturing.
