Advanced Asymmetric Synthesis of Thiamphenicol and Florfenicol Intermediates for Commercial Scale Production
The pharmaceutical landscape for veterinary antibiotics is undergoing a significant transformation driven by the need for more efficient and environmentally sustainable manufacturing processes. Patent CN1743308A introduces a groundbreaking methodology for the synthesis of thiamphenicol and florfenicol, two critical broad-spectrum antibiotics widely used in animal health. Unlike conventional approaches that rely heavily on the resolution of racemic mixtures, this novel pathway leverages enzymatic catalysis to construct chiral centers with exceptional precision. By utilizing hydroxynitrile lyase (HNL) derived from specific botanical sources, the process achieves high enantiomeric excess without the substantial material waste associated with traditional splitting techniques. This technological leap not only enhances the economic viability of production but also aligns with modern green chemistry principles, making it a highly attractive option for large-scale commercial manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
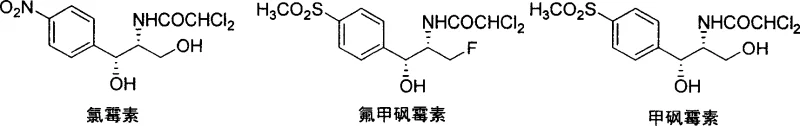
Historically, the industrial production of thiamphenicol and its fluorinated analog, florfenicol, has been plagued by inherent inefficiencies related to stereochemical control. The predominant legacy technology involves the synthesis of racemic substituted phenylserine derivatives, which subsequently require resolution to isolate the pharmacologically active D-(-)-isomer. This resolution process, often utilizing tartaric acid or similar chiral agents, inherently limits the maximum theoretical yield to 50%, as the unwanted L-isomer is typically discarded or requires complex recycling streams. Furthermore, the multiple steps involved in protecting and deprotecting functional groups during these traditional routes contribute to a lower overall process mass intensity (PMI) and generate significant volumes of chemical waste. For procurement managers and supply chain directors, these inefficiencies translate into higher raw material costs, volatile pricing due to yield fluctuations, and increased environmental compliance burdens that can disrupt continuous production schedules.
The Novel Approach
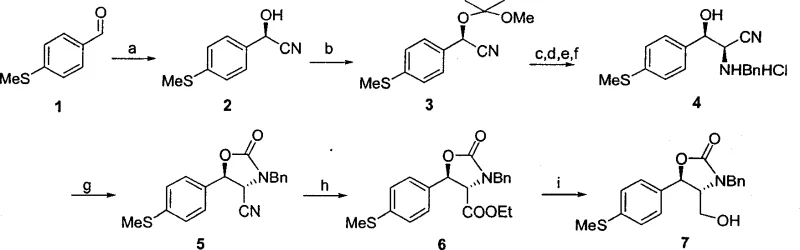
In stark contrast, the methodology disclosed in CN1743308A circumvents these bottlenecks by establishing chirality at the very beginning of the synthesis through biocatalysis. The process initiates with the asymmetric hydrocyanation of p-methylthiobenzaldehyde, catalyzed by hydroxynitrile lyase sourced from Badam seeds, which delivers the chiral cyanohydrin intermediate with greater than 95% yield and over 96% enantiomeric excess. This high-fidelity introduction of the first chiral center allows for the subsequent diastereoselective formation of the second chiral center via imine substitution and cyclization, effectively locking in the desired stereochemistry without the need for resolution. The resulting oxazolidinone intermediate serves as a robust scaffold that protects the sensitive amino and hydroxyl functionalities during downstream transformations, such as sulfide oxidation and fluorination. This streamlined approach significantly reduces the number of unit operations and eliminates the wasteful discarding of isomers, offering a direct path to cost reduction in veterinary antibiotic intermediate manufacturing.
Mechanistic Insights into Enzymatic Hydrocyanation and Oxazolidinone Formation
The cornerstone of this innovative synthesis is the enzyme-catalyzed hydrocyanation reaction, which utilizes hydroxynitrile lyase (HNL) to facilitate the addition of hydrogen cyanide to the aldehyde substrate. The patent highlights a critical discovery that not all HNL sources are equal; specifically, enzymes extracted from Badam seeds demonstrated superior catalytic activity and stereoselectivity compared to those from almonds, loquats, or apples when used in organic solvent systems like diisopropyl ether (DIPE). The reaction mechanism involves the enzyme stabilizing the transition state of the cyanohydrin formation, ensuring that the nucleophilic attack occurs exclusively from one face of the planar carbonyl group. Optimization of the water content in the organic phase, maintained between 0.5% and 2% (v/v), is crucial for maintaining enzyme activity while preventing non-enzymatic background reactions that could erode enantiomeric purity. Following this, the chiral information is transferred to the second center through a sequence involving protection of the hydroxyl group, reduction of the nitrile to an imine using DIBAL-H, and subsequent nucleophilic addition of benzylamine and HCN. This cascade ensures that the relative stereochemistry is strictly controlled, yielding the trans-oxazolidinone structure which is thermodynamically stable and resistant to epimerization under the reaction conditions employed for subsequent steps.
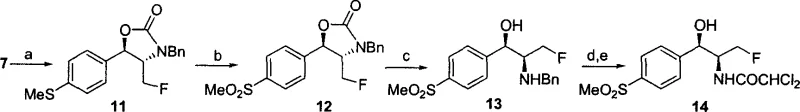
Following the construction of the chiral core, the synthesis proceeds through a series of chemically robust transformations designed to install the final functional groups required for biological activity. For thiamphenicol, the methylthio group is oxidized to the corresponding sulfone using meta-chloroperoxybenzoic acid (MCPBA), a reaction that proceeds with high selectivity without affecting the oxazolidinone ring. In the case of florfenicol, a dehydroxyfluorination step using DAST (diethylaminosulfur trifluoride) is introduced prior to oxidation to install the critical fluorine atom. A key mechanistic challenge addressed in the patent is the stability of the carbon-fluorine bond during the hydrolytic ring-opening of the oxazolidinone. Standard basic hydrolysis conditions were found to cause defluorination; however, the inventors discovered that using concentrated sulfuric acid (8N) at elevated temperatures (140°C) allows for successful ring opening while retaining the fluorine substituent. Finally, catalytic hydrogenation removes the benzyl protecting group, and acetylation with ethyl dichloroacetate installs the dichloroacetyl moiety, completing the synthesis of the active pharmaceutical ingredient with high purity and consistent stereochemical integrity.
How to Synthesize Thiamphenicol and Florfenicol Efficiently
The synthesis of these high-value veterinary antibiotics requires precise control over reaction parameters to maintain the integrity of the chiral centers and functional groups. The process begins with the preparation of the biocatalyst, where Badam seeds are degreased and processed to extract the active hydroxynitrile lyase, which is then employed in a biphasic or organic solvent system to generate the initial cyanohydrin. Subsequent steps involve standard organic transformations such as silyl or acetal protection, metal-hydride reductions, and cyclization using carbonyldiimidazole to form the key oxazolidinone intermediate. The detailed operational protocols, including specific molar ratios, temperature ranges, and workup procedures necessary to achieve the reported yields and purities, are critical for successful technology transfer. For R&D teams looking to implement this route, adherence to the specified conditions for the fluorination and hydrolysis steps is particularly vital to avoid degradation of the final product. The standardized synthesis steps outlined below provide a framework for scaling this process from laboratory to commercial production.
- Perform enzyme-catalyzed hydrocyanation of p-methylthiobenzaldehyde using Hydroxynitrile Lyase (HNL) from Badam seeds to generate chiral cyanohydrin with high enantiomeric excess.
- Protect the hydroxyl group and convert the nitrile to an imine, followed by benzylamine substitution and subsequent HCN addition to establish the second chiral center.
- Form the oxazolidinone ring using carbonyldiimidazole, reduce the ester to alcohol, and finally oxidize the sulfide to sulfone followed by acetylation to yield the target antibiotic.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this enzymatic asymmetric synthesis route offers profound strategic advantages over traditional resolution-based manufacturing. The primary benefit lies in the drastic improvement of atom economy; by eliminating the resolution step, the process theoretically doubles the yield of usable chiral material from the starting aldehyde, thereby significantly reducing the raw material cost per kilogram of the final active ingredient. This efficiency gain is not merely a laboratory metric but translates directly into a more competitive cost structure, allowing suppliers to offer more stable pricing even in the face of fluctuating raw material markets. Furthermore, the reduction in chemical waste and the avoidance of harsh resolution reagents simplify the effluent treatment process, lowering the environmental compliance costs and reducing the risk of production stoppages due to regulatory issues. The robustness of the oxazolidinone intermediate also enhances supply chain reliability, as it can be stored and transported with greater stability than some of the more labile intermediates found in older synthetic routes.
- Cost Reduction in Manufacturing: The elimination of the racemic resolution step fundamentally alters the cost dynamics of production by removing the 50% yield penalty associated with discarding the unwanted isomer. This means that for every ton of starting aldehyde purchased, nearly double the amount of active pharmaceutical ingredient can be produced compared to conventional methods. Additionally, the use of enzymatic catalysis often allows for milder reaction conditions, which can lead to reduced energy consumption for heating and cooling, further driving down operational expenditures. The streamlined sequence also reduces the total number of isolation and purification steps, minimizing solvent usage and labor costs associated with batch processing. These cumulative efficiencies result in a substantially lower cost of goods sold (COGS), providing a strong buffer against market volatility and enabling more aggressive pricing strategies in the competitive veterinary pharmaceutical sector.
- Enhanced Supply Chain Reliability: Reliance on enzymatic processes sourced from renewable botanical materials, such as Badam seeds, diversifies the supply base away from purely petrochemical-derived chiral auxiliaries which can be subject to supply constraints. The high selectivity of the enzymatic step reduces the formation of difficult-to-remove impurities, leading to more consistent batch-to-batch quality and reducing the likelihood of failed quality control tests that can delay shipments. Moreover, the stability of the key oxazolidinone intermediate allows for the decoupling of upstream and downstream manufacturing stages; the intermediate can be produced in large batches and stored, acting as a buffer against demand spikes or temporary disruptions in the final synthesis steps. This flexibility is crucial for maintaining continuous supply to global pharmaceutical customers who require just-in-time delivery to support their own formulation schedules.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reagents and conditions that are compatible with standard stainless steel reactor infrastructure found in most fine chemical plants. The avoidance of heavy metal catalysts in the key chirality-setting step simplifies the purification process and reduces the burden of heavy metal testing and removal, which is a stringent requirement for API manufacturing. From an environmental perspective, the higher overall yield and reduced solvent consumption contribute to a lower E-factor (mass of waste per mass of product), aligning with increasingly strict global environmental regulations. This green chemistry profile not only mitigates regulatory risk but also enhances the brand reputation of the manufacturer as a sustainable partner, which is becoming a key differentiator in procurement decisions for major multinational pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis route for thiamphenicol and florfenicol. These insights are derived directly from the experimental data and process descriptions provided in the patent literature, offering a clear understanding of the feasibility and benefits of this technology. Understanding these details is essential for technical teams evaluating the potential for process optimization and cost savings in their current supply chains.
Q: How does the enzymatic route improve yield compared to traditional resolution methods?
A: Traditional methods rely on resolving racemic mixtures, theoretically discarding 50% of the material. The enzymatic hydrocyanation described in CN1743308A achieves >95% yield and >96% ee directly, effectively doubling the theoretical atom economy regarding chiral centers.
Q: What are the critical conditions for the hydroxynitrile lyase (HNL) catalysis step?
A: The patent specifies using HNL from Badam seeds in organic solvents like DIPE or TBME with controlled water content (0.5-2% v/v). This specific enzyme source was found to be superior to almond or apple seed enzymes for this specific substrate.
Q: How is the fluorine atom introduced for Florfenicol synthesis in this route?
A: Fluorination is achieved using DAST (Diethylaminosulfur trifluoride) on the protected intermediate prior to sulfone oxidation. The patent notes that careful control of hydrolysis conditions (using 8N H2SO4 at 140°C) is required to prevent defluorination during ring opening.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Thiamphenicol and Florfenicol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the enzymatic asymmetric synthesis route described in CN1743308A for the production of high-purity veterinary antibiotics. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel chemistry are fully realized in an industrial setting. Our state-of-the-art facilities are equipped to handle the specific requirements of biocatalytic processes, including the handling of enzymatic reagents and the precise control of reaction parameters needed to maintain high enantiomeric excess. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of thiamphenicol or florfenicol intermediate meets the exacting standards required by global regulatory authorities. Our commitment to technical excellence ensures that clients receive a product that is not only cost-effective but also consistently reliable in terms of quality and performance.
We invite procurement leaders and technical directors to engage with our team to explore how this advanced synthesis route can optimize your supply chain and reduce overall manufacturing costs. By leveraging our expertise in process development and scale-up, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to this next-generation manufacturing platform. Together, we can drive efficiency and sustainability in the production of essential veterinary medicines, securing a robust supply for the global animal health market.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
