Advanced Synthesis Of Prostaglandin F Derivatives For Commercial Scale-Up And High Purity Production
Advanced Synthesis Of Prostaglandin F Derivatives For Commercial Scale-Up And High Purity Production
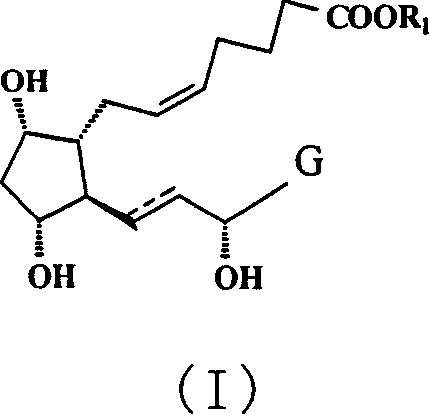
The pharmaceutical landscape for ophthalmic treatments continues to evolve, driven by the urgent need for effective therapies against glaucoma and intraocular hypertension. Patent CN1990450A introduces a groundbreaking methodology for the preparation of prostaglandin F type derivatives, specifically targeting the structural motif represented by Formula (I). This innovation addresses long-standing challenges in the synthesis of these complex molecules, offering a pathway that enhances both purity and process efficiency. The target compounds are critical active pharmaceutical ingredients (APIs) or intermediates used in the formulation of eye drops that manage intraocular pressure, a key factor in preventing vision loss associated with glaucoma. By leveraging a sophisticated dual-silylation strategy, this patent provides a robust framework for manufacturers aiming to scale production while maintaining stringent quality standards required for ophthalmic applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for prostaglandin analogues often suffer from significant inefficiencies, particularly regarding the purification of intermediates following carbon-carbon bond-forming reactions. A major bottleneck in conventional Wittig olefinations is the removal of phosphine oxide byproducts and unreacted phosphonium salts, which are highly polar and difficult to separate from the desired lipophilic product using standard extraction techniques. Furthermore, the presence of multiple hydroxyl groups in the prostaglandin backbone necessitates careful protection group strategies; failure to differentiate between these hydroxyls can lead to non-selective reactions, resulting in complex mixtures of regioisomers that drastically reduce overall yield. Conventional methods frequently rely on extensive column chromatography to achieve pharmaceutical-grade purity, a process that is not only costly and time-consuming but also notoriously difficult to translate from laboratory scale to industrial manufacturing due to solvent consumption and throughput limitations.
The Novel Approach
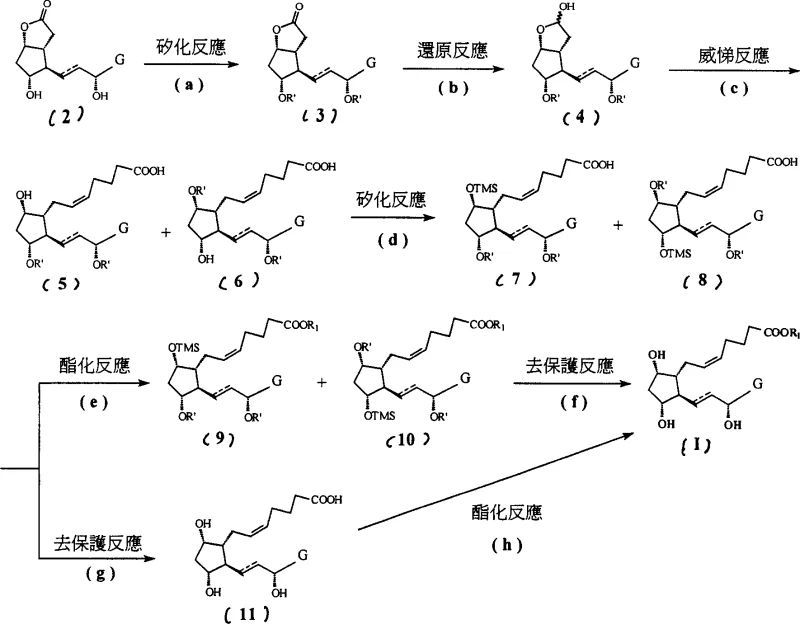
The methodology disclosed in CN1990450A overcomes these hurdles through a clever manipulation of silyl protecting groups with distinct steric and hydrolytic properties. The process initiates with the protection of specific hydroxyl groups using a bulky silylating agent, such as triethylchlorosilane (TES-Cl), which provides robust stability during subsequent reduction steps. Following the construction of the carbon skeleton via Wittig reaction, a second silylation is introduced using a smaller, more labile group like trimethylsilyl chloride (TMSCl). This differential protection strategy significantly alters the physicochemical properties of the intermediate, specifically increasing its lipophilicity. This modification is the key innovation, as it enables the efficient removal of polar phosphonium impurities through simple liquid-liquid extraction rather than chromatography. The result is a streamlined process that simplifies downstream processing and enhances the feasibility of large-scale production.
Mechanistic Insights into Differential Silylation and Wittig Olefination
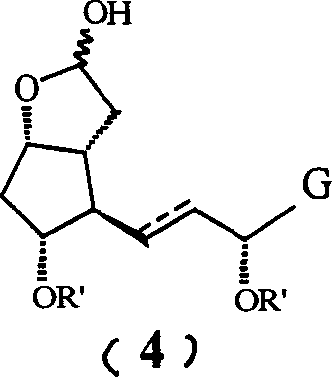
The core of this synthetic advancement lies in the orthogonal protection strategy employed throughout the reaction sequence. Initially, the starting material, typically a bicyclic lactone derivative, undergoes selective silylation where the more accessible or reactive hydroxyl groups are protected with triethylsilyl (TES) groups. This choice is deliberate; the TES group is sterically larger and more resistant to hydrolysis than a trimethylsilyl (TMS) group, ensuring that these positions remain protected during the vigorous conditions of the subsequent reduction and olefination steps. The reduction of the lactone carbonyl to a lactol using diisobutylaluminium hydride (DIBAL-H) is a critical transformation that must be conducted at cryogenic temperatures, specifically between -60°C and -70°C. This low-temperature control is essential to prevent over-reduction or the opening of the lactone ring, preserving the integrity of the cyclopentane core which is vital for the biological activity of the final prostaglandin derivative.
Following the Wittig reaction, which installs the crucial omega-chain containing the carboxylic acid precursor, the intermediate possesses free hydroxyl groups that would otherwise complicate purification. The introduction of the TMS group at this stage serves a dual purpose: it protects the hydroxyl functionality for future deprotection and, more importantly, masks the polarity of the molecule. The resulting bis-silylated intermediate exhibits sufficient lipophilicity to partition into organic solvents, leaving behind the highly polar phosphonium salts and acidic byproducts in the aqueous phase. This mechanistic nuance allows for a 'clean-up' of the reaction mixture prior to the final esterification and global deprotection steps. The final deprotection is achieved under mild acidic conditions, removing both TES and TMS groups simultaneously to reveal the native hydroxyl architecture of the prostaglandin F derivative, ensuring the final product matches the required stereochemistry and functional group arrangement for therapeutic efficacy.
How to Synthesize Prostaglandin F Derivatives Efficiently
The synthesis of these high-value ophthalmic intermediates requires precise adherence to the reaction parameters outlined in the patent to ensure reproducibility and safety. The process involves a sequence of protection, reduction, chain extension, and deprotection steps that must be carefully monitored. Operators must pay close attention to the stoichiometry of silylating agents and the temperature profiles during the exothermic reduction phase. The following guide summarizes the critical operational phases derived from the patent data, providing a roadmap for technical teams looking to implement this route. For the detailed standardized operating procedures, including specific reagent grades and workup protocols, please refer to the structured guide below.
- Perform initial protection of compound (2) using a bulky silylating agent like triethylchlorosilane to form compound (3), preventing side reactions during reduction.
- Reduce the lactone carbonyl of compound (3) to a lactol using DIBAL-H at low temperatures (-60 to -70°C) to generate compound (4).
- Execute a Wittig reaction on compound (4) to introduce the upper side chain, followed by a second protection step using trimethylsilyl chloride to facilitate impurity removal.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the drastic simplification of the purification workflow. By enabling the removal of phosphonium impurities via extraction rather than chromatography, the process eliminates a major bottleneck that typically constrains batch throughput and increases solvent costs. This shift from chromatographic purification to extractive workup translates directly into reduced manufacturing cycles and lower operational expenditures. Furthermore, the use of commercially available silylating reagents like TES-Cl and TMSCl ensures that raw material sourcing remains stable and cost-effective, mitigating the risks associated with specialized or exotic catalysts that might face supply disruptions.
- Cost Reduction in Manufacturing: The elimination of large-scale column chromatography significantly reduces the consumption of silica gel and organic solvents, which are major cost drivers in fine chemical production. Additionally, the improved selectivity of the differential silylation strategy minimizes the formation of regioisomeric byproducts, thereby enhancing the overall yield of the desired intermediate. This increase in yield means that less starting material is required to produce the same amount of final API, effectively lowering the cost of goods sold (COGS). The robustness of the intermediates also reduces the likelihood of batch failures, further contributing to substantial cost savings over the lifecycle of the product.
- Enhanced Supply Chain Reliability: The reliance on standard reagents and straightforward unit operations makes this process highly resilient to supply chain volatility. Unlike routes dependent on sensitive transition metal catalysts that may require complex removal steps or specialized handling, this silylation-based approach utilizes stable chemicals with long shelf lives. This stability allows manufacturers to maintain strategic stockpiles of key intermediates without significant degradation concerns. Consequently, lead times for high-purity prostaglandin intermediates can be significantly shortened, ensuring a continuous supply for downstream formulation partners and reducing the risk of production stoppages due to material shortages.
- Scalability and Environmental Compliance: From an environmental and safety perspective, the reduction in solvent usage associated with the extractive workup aligns well with green chemistry principles and increasingly stringent regulatory standards. The process avoids the generation of large volumes of silica waste, simplifying waste management and disposal protocols. Moreover, the ability to scale the reaction without the geometric limitations of chromatography columns facilitates the transition from pilot plant to commercial production. This scalability ensures that the manufacturing capacity can be rapidly expanded to meet market demand for glaucoma medications, supporting long-term supply continuity for global pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these prostaglandin derivatives. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific production needs. For further technical clarification or custom synthesis requests, our team is available to provide detailed discussions.
Q: What is the primary therapeutic application of Prostaglandin F derivatives?
A: Prostaglandin F type derivatives are clinically indicated for treating glaucoma and intraocular hypertension, helping to reduce intraocular pressure and prevent optic nerve damage.
Q: How does the novel silylation strategy improve purification?
A: By using a combination of bulky (TES) and small (TMS) silyl groups, the process increases lipophilicity, allowing for the effective extraction and removal of highly polar phosphonium salt impurities without complex chromatography.
Q: What are the critical temperature controls in this synthesis?
A: Precise temperature control is vital, particularly during the DIBAL-H reduction step which requires -60 to -70°C, and the silylation steps which are optimally performed between 0 to 5°C to ensure selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Prostaglandin F Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving ophthalmic therapies. Our facility is equipped with state-of-the-art reactors and purification systems capable of handling complex multi-step syntheses like the one described in CN1990450A. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our rigorous QC labs enforce stringent purity specifications, guaranteeing that every batch of prostaglandin intermediate meets the exacting standards required for pharmaceutical registration and clinical use.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can be optimized for your specific project requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that evaluates the economic benefits of implementing this differential silylation strategy in your supply chain. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to being your trusted partner in the production of high-value pharmaceutical intermediates.
